Review Article
The Role of Macrophages in Angiogenesis
1Department of Natural Science, Middlesex University, Hendon, London, United Kingdom.
2Faculty of Medicine, Department of Pathology, Tanta University, Tanta, Egypt.
3Department of Histopathology, King’s College Hospital, Denmark Hill, London, United Kingdom.
*Corresponding Author: Abdel-Ghani Selim, Department of Histopathology, King’s College Hospital, Denmark Hill, London, United Kingdom.
Citation: Elayat G, Selim AG. (2026). The Role of Macrophages in Angiogenesis, Journal of Clinical Research and Clinical Trials, BioRes Scientia Publishers. 5(1):1-5. DOI: 10.59657/2837-7184.brs.26.065
Copyright: © 2026 Abdel-Ghani Selim, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: February 02, 2026 | Accepted: March 13, 2026 | Published: March 19, 2026
Abstract
Angiogenesis, the formation of new blood vessels from existing vasculature, is fundamental to both normal tissue homeostasis and pathological processes such as tumor growth and chronic inflammation. Macrophages, a highly plastic component of the immune system, are key regulators of angiogenesis. This review explores the evolving understanding of macrophage polarization beyond the classical M1/M2 paradigm, emphasizing their phenotypic diversity and plasticity in response to environmental cues. We detail the mechanisms by which macrophages orchestrate angiogenesis, including the secretion of pro-angiogenic mediators (e.g., VEGF, ANGPT2), extracellular matrix remodeling via matrix metalloproteinases, and immune modulation through cytokines like IL-10 and TGF-β. We further highlight critical signaling pathways—HIF-1α and STAT3—that govern macrophage-mediated angiogenesis in both physiological (e.g., wound healing, development) and pathological (e.g., cancer, diabetic retinopathy) contexts. The therapeutic implications of targeting macrophages to modulate angiogenesis in oncology and chronic inflammatory diseases are also discussed. A comprehensive understanding of the regulatory networks linking macrophage biology and angiogenesis is essential for the development of next-generation therapies targeting aberrant vascularization.
Keywords: angiogenesis; macrophages; extracellular matrix; HIF pathway; therapeutic approaches; STAT3; VEGF
Introduction
Angiogenesis, the formation of new blood vessels from pre-existing vasculature, is essential for tissue development, wound healing, and reproduction. However, dysregulated angiogenesis is also a hallmark of numerous pathological conditions, including cancer, diabetic retinopathy, and rheumatoid arthritis. Among the immune cells implicated in this process, macrophages have emerged as central regulators, influencing every phase of angiogenesis through a wide array of molecular and cellular mechanisms.
Macrophage Polarisation and Plasticity
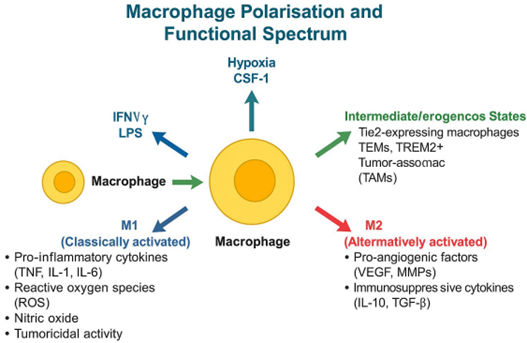
Macrophages exhibit remarkable plasticity, adapting their functional states in response to diverse microenvironmental cues. The classical M1/M2 dichotomy, though historically useful, oversimplifies the dynamic and context-specific spectrum of macrophage activation. Contemporary understanding recognizes a continuum of macrophage states shaped by stimuli such as pathogen-associated molecular patterns (PAMPs), cytokines, tissue signals, and metabolic cues [1].
Classically activated macrophages (M1) are typically induced by IFN-γ and Toll-like receptor ligands (e.g., LPS), leading to a pro-inflammatory phenotype. These macrophages produce nitric oxide (NO), reactive oxygen species (ROS), and pro-inflammatory cytokines (e.g., TNF-α, IL-1β, IL-6), playing key roles in microbial defense and tumor surveillance. However, excessive M1 activity can drive tissue damage and chronic inflammation [2].
Alternatively activated macrophages (M2) arise in response to IL-4, IL-13, and glucocorticoids. They are associated with tissue repair, immune regulation, and angiogenesis through the secretion of VEGF, IL-10, TGF-β, and MMPs. M2 macrophages promote vessel stability and endothelial cell proliferation but can also facilitate tumor progression and fibrotic diseases [3].
Recent evidence underscores the existence of hybrid or intermediate phenotypes. For instance, tumor-associated macrophages (TAMs) often co-express M1 and M2 markers, influenced by hypoxia, metabolic reprogramming, and tumor-derived factors [4]. Other subsets such as TREM2+, CD206+, and Tie2-expressing macrophages (TEMs) have been identified with potent angiogenic and immunomodulatory functions, especially in cancer and chronic inflammation [5,6].
Understanding this phenotypic diversity is critical, as targeting macrophage subtypes offers potential for modulating angiogenesis in both therapeutic and pathological contexts.
Figure 1: Macrophage Polarisation and Functional Spectrum.
Regulatory Pathways in Macrophage-Mediated Angiogenesis
Hypoxia-Inducible Factor (HIF) Pathway
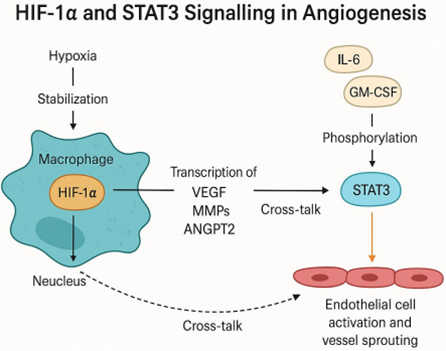
Hypoxia is a critical trigger for angiogenesis, particularly in tumors and healing wounds. Macrophages adapt to hypoxic environments by stabilising hypoxia-inducible factor 1-alpha (HIF-1α), a master regulator of oxygen-sensitive gene expression. Under normoxic conditions, HIF-1α is rapidly degraded via the von Hippel-Lindau (VHL) pathway. However, hypoxia inhibits this degradation, allowing HIF-1α to accumulate, dimerise with HIF-1β, and activate transcription of angiogenic genes including VEGF, ANGPT2, and MMP9 [7].
HIF-1α drives macrophage polarisation toward a pro-angiogenic M2-like phenotype, enhancing their capacity to secrete VEGF and facilitate extracellular matrix remodeling. In tumors, hypoxia-stabilised HIF-1α in TAMs fosters neovascularisation and immune evasion. Furthermore, HIF-2α, which is preferentially expressed in tissue-resident macrophages, also supports angiogenesis through overlapping but distinct gene targets [8].
Studies have shown that macrophage-specific deletion of HIF-1α impairs angiogenesis in models of wound healing and cancer, affirming its central role in coordinating macrophagedriven vascular growth [9].
STAT3 Pathway
The Signal Transducer and Activator of Transcription 3 (STAT3) pathway is another pivotal regulator of macrophage function in angiogenesis. It is activated by cytokines such as IL-6, IL-10, and growth factors like GM-CSF and EGF, all of which are enriched in tumor and chronically inflamed tissues. Once phosphorylated, STAT3 dimerises and translocate to the nucleus to initiate transcription of genes associated with M2 polarisation and angiogenesis, including VEGF, TGF-β, and Arg1 [10].
Persistent STAT3 signaling in TAMs sustains their immunosuppressive and pro-angiogenic phenotype. It also facilitates crosstalk with endothelial cells, promoting tip cell formation and vascular sprouting. Inhibition of STAT3 has been shown to reduce pathological angiogenesis in preclinical models of cancer, rheumatoid arthritis, and ocular neovascular diseases [11].
Moreover, the STAT3-HIF-1α axis represents a key convergence point where inflammatory and hypoxic signals cooperate to enhance the angiogenic function of macrophages. Targeting this axis may provide a dual approach to suppress aberrant neovascularisation and reshape the immune microenvironment [12].
Figure 2: HIF-1α and STAT3 Signaling in Macrophage-Mediated Angiogenesis.
Tumour-Associated Macrophages and Pathological Angiogenesis
Tumour-associated macrophages (TAMs) represent a major leukocyte population within the tumour microenvironment and play a central role in promoting tumour angiogenesis. TAMs are typically skewed toward an M2-like phenotype, exhibiting pro-angiogenic, immunosuppressive, and tissue-remodeling functions. This phenotype is maintained by tumour-derived signals such as IL-10, TGF-β, CSF-1, and VEGF, as well as hypoxia-induced pathways like HIF-1α and STAT3 signaling [13].
TAMs support angiogenesis through multiple mechanisms: secretion of angiogenic factors including VEGF, PDGF, bFGF, and ANGPT2; extracellular matrix (ECM) remodeling via production of MMP2, MMP9, and cathepsins; and immunosuppression through IL-10 and TGF-β [14–16]. Tie2-expressing macrophages (TEMs) localise near vasculature and directly coordinate neovascular sprouting through angiopoietin–Tie2 signaling [17].
Clinically, a high density of TAMs correlates with poor prognosis in cancers such as breast, ovarian, and glioblastoma. Therapeutic strategies targeting TAMs aim to deplete or inhibit recruitment (e.g., CSF-1R or CCL2 blockade), reprogram TAMs toward M1-like phenotypes (e.g., CD40 or TLR agonists), or suppress angiogenic outputs (e.g., via VEGF or STAT3 inhibition) [18,19].
Therapeutic Implications
Targeting Tumour-Associated Macrophages in Cancer Therapy
Given the pivotal role of TAMs in sustaining tumour angiogenesis, immune suppression, and metastasis, therapeutic interventions have increasingly focused on modulating macrophage activity. These include CSF-1R blockade (e.g., pexidartinib), CCL2/CCR2 inhibition, and functional reprogramming using CD40 agonists, TLR ligands, or PI3Kγ inhibitors [20-23].
Combining TAM-targeting approaches with immune checkpoint inhibitors or antiangiogenic therapies enhances efficacy and improves vascular normalisation [24].
Modulating Macrophages in Chronic Inflammatory Diseases
In conditions such as rheumatoid arthritis and diabetic retinopathy, macrophages contribute to chronic inflammation and pathological angiogenesis. Anti-VEGF therapies, STAT3 inhibitors, and CXCL8/IL-8 blockade are promising strategies for restoring immune and vascular homeostasis in these settings [25-27].
Conclusion
Macrophages play a central and multifaceted role in angiogenesis, acting as orchestrators of vascular growth through secretion of pro-angiogenic factors, immune regulation, and extracellular matrix remodeling. Their functional versatility is governed by dynamic environmental cues, resulting in a spectrum of activation states beyond the simplistic M1/M2 model. In pathological settings such as cancer and chronic inflammation, macrophages drive aberrant angiogenesis that contributes to disease progression. Critical signaling pathways-including HIF-1α and STAT3-integrate hypoxic and inflammatory stimuli to reinforce macrophage-driven vascularisation. Therapeutically, targeting macrophage recruitment, polarisation, and angiogenic signaling offers a promising strategy to restore vascular homeostasis or suppress tumour growth. Continued advances in our understanding of macrophage plasticity and their regulatory circuits will be key to realising the full therapeutic potential of angiogenesis modulation.
Declarations
Conflicts of Interest
The authors have no conflicts of interest to declare.
Ethical Approval
Not applicable.
Funding
Not applicable.
Data Availability
Not applicable.
References
- Murray PJ. (2017). Macrophage polarization. Annu Rev Physiol. 79:541-566.
Publisher | Google Scholor - Mantovani A, Marchesi F, Malesci A, Laghi L, Allavena P. (2017). Tumour-associated macrophages as treatment targets in oncology. Nat Rev Clin Oncol. 14(7):399-416.
Publisher | Google Scholor - Italiani P, Boraschi D. (2014). From monocytes to M1/M2 macrophages: Phenotypical vs. functional differentiation. Front Immunol. 5:514.
Publisher | Google Scholor - Cassetta L, Pollard JW. (2018). Targeting macrophages: Therapeutic approaches in cancer. Nat Rev Drug Discov. 17(12):887-904.
Publisher | Google Scholor - De Palma M, Lewis CE. (2013). Macrophage regulation of tumor responses to anticancer therapies. Cancer Cell. 23(3):277-286.
Publisher | Google Scholor - Zhang Q, Liu L, Gong C, Shi H, Zeng Y, et al. (2012). Prognostic significance of tumor[1]associated macrophages in solid tumor: A meta-analysis of the literature. PLoS One. 7(12):e50946.
Publisher | Google Scholor - Nizet V, Johnson RS. (2009). Interdependence of hypoxic and innate immune responses. Nat Rev Immunol. 9(9):609-617.
Publisher | Google Scholor - Imtiyaz HZ, Simon MC. (2010). Hypoxia-inducible factors as essential regulators of inflammation. Curr Top Microbiol Immunol. 345:105-120.
Publisher | Google Scholor - Zhang W, Petrovic JM, Callahan M, Dean DA, Huang J, et al. (2014). HIF-1α-dependent angiogenic capacity of M2 macrophages. J Immunol. 193(2):867-875.
Publisher | Google Scholor - Yu H, Pardoll D, Jove R. (2009). STATs in cancer inflammation and immunity: A leading role for STAT3. Nat Rev Cancer. 9(11):798-809.
Publisher | Google Scholor - Kortylewski M, Kujawski M, Wang T, Wei S, Zhang S, et al. (20050. Inhibiting STAT3 signaling in the hematopoietic system elicits multicomponent antitumor immunity. Nat Med. 11(12):1314-1321.
Publisher | Google Scholor - Qin HR, Kim HJ, Kim JY, Hurt EM, Klarmann GJ, et al. (2008). Activation of STAT3 through a phospho-Tyr705-independent pathway promotes angiogenesis in hypoxia. Mol Cancer Res. 6(4):546-554.
Publisher | Google Scholor - Ruffell B, Affara NI, Coussens LM. (2012). Differential macrophage programming in the tumour microenvironment. Trends Immunol. 33(3):119-126.
Publisher | Google Scholor - Lewis CE, Pollard JW. (2006). Distinct role of macrophages in different tumour microenvironments. Cancer Res. 66(2):605-612.
Publisher | Google Scholor - Wyckoff J, Wang W, Lin EY, Wang Y, Pixley F, et al. (2004). A paracrine loop between tumour cells and macrophages is required for tumour cell migration in mammary tumors. Cancer Res. 64(19):7022-7029.
Publisher | Google Scholor - Gabrilovich DI, Nagaraj S. (2009). Myeloid-derived suppressor cells as regulators of the immune system. Nat Rev Immunol. 9(3):162-174.
Publisher | Google Scholor - De Palma M, Venneri MA, Galli R, Sergi L, Politi LS, et al. (2007). Tie2-expressing monocytes: regulation of tumor angiogenesis and therapeutic implications. Trends Immunol. 28(12):519-524.
Publisher | Google Scholor - Zhang QW, Liu L, Gong CY, Shi HS, Zeng YH, et al. (2012). Prognostic significance of tumour-associated macrophages in solid tumour: A meta-analysis of the literature. PLoS One. 7(12):e50946.
Publisher | Google Scholor - Ries CH, Cannarile MA, Hoves S, Benz J, Wartha K, et al. (2014). Targeting tumour associated macrophages with anti-CSF-1R antibody reveals a strategy for cancer therapy. Cancer Cell. 25(6):846-859.
Publisher | Google Scholor - Cannarile MA, Weisser M, Jacob W, Jegg AM, Ries CH, et al. (2017). Colony-stimulating factor 1 receptor (CSF1R) inhibitors in cancer therapy. J Immunother Cancer. 5(1):53.
Publisher | Google Scholor - Qian BZ, Pollard JW. (2010). Macrophage diversity enhances tumor progression and metastasis. Cell. 141(1):39-51.
Publisher | Google Scholor - Beatty GL, Chiorean EG, Fishman MP, Saboury B, Teitelbaum UR, et al. (2011). CD40 agonists alter tumour stroma and show efficacy against pancreatic carcinoma in mice and humans. Science. 331(6024):1612-1616.
Publisher | Google Scholor - De Henau O, Rausch M, Winkler D, Campesato LF, Liu C, et al. (2016). Overcoming resistance to checkpoint blockade therapy by targeting PI3Kγ in myeloid cells. Nature. 539(7629):443-447.
Publisher | Google Scholor - Mantovani A, Marchesi F, Malesci A, Laghi L, Allavena P. (2017). Tumour-associated macrophages as treatment targets in oncology. Nat Rev Clin Oncol. 14(7):399-416.
Publisher | Google Scholor - Ferrara N, Adamis AP. (2016). Ten years of anti-vascular endothelial growth factor therapy. Nat Rev Drug Discov. 15(6):385-403.
Publisher | Google Scholor - Semenza GL. (2019). Pharmacologic targeting of hypoxia-inducible factors. Annu Rev Pharmacol Toxicol. 59:379-403.
Publisher | Google Scholor - Li A, Dubey S, Varney ML, Dave BJ, Singh RK. (2003). IL-8 directly enhanced endothelial cell survival, proliferation, and matrix metalloproteinases production and regulated angiogenesis. J Immunol. 170(6):3369-3376.
Publisher | Google Scholor