Research Article
Targeted Epigenetic Reprogramming of IL-6 Expression by An in Silico Molecular Editor Reverses Immune Senescence
- Victor Jose Ramon Blanco Lopez *
Surgeon, Specialist in Occupational Hygiene, Head of the Occupational Health Area of the San Jode Chincha Hospital, Peru.
*Corresponding Author: Victor Jose Ramon Blanco Lopez, Surgeon, Specialist in Occupational Hygiene, Head of the Occupational Health Area of the San Jode Chincha Hospital, Peru.
Citation: Lopez VJRB. (2025). Targeted Epigenetic Reprogramming of IL-6 Expression by An in Silico Molecular Editor Reverses Immune Senescence, International Journal of Biomedical and Clinical Research, BioRes Scientia Publishers. 4(6):1-4. DOI: 10.59657/2997-6103.brs.25.104
Copyright: © 2025 Victor Jose Ramon Blanco Lopez, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: October 15, 2025 | Accepted: December 02, 2025 | Published: December 08, 2025
Abstract
Phenotypic aging is the manifestation of a progressive dysregulation of gene expression networks. A pillar of this process is "inflammaging", a chronic pro-inflammatory state driven by the cytokine IL-6, which accelerates multi-system degradation. Current interventions, based on protein neutralization, are palliative and incapable of correcting the underlying programmatic defect. In this study, we present a paradigm shift: the treatment of aging as an epigenetic "software" error that can be debugged. By analyzing 1.2 petabytes of multi-omics data, we decode a causal epigenetic signature-a hypomethylated enhancer locus that we have dubbed the "IL6-E1 Cronos Signature"-as the master switch for IL-6 overexpression. Based on this discovery, we engineered in silico CTX-001, a precision epigenetic editor composed of a dCas9-DNMT3A system packaged in an AAV-DJ vector. In a high-fidelity virtual preclinical trial, administration of a single dose of CTX-001 not only attenuated IL-6 expression by 58%, but induced an inflammatory resolution cascade and promoted reconstitution of the naïve T cell compartment, a marker of immune rejuvenation. This work establishes the viability of "programmatic medicine" and presents CTX-001 as the prototype of a new class of therapies designed not to combat the diseases of aging, but to rewrite the aging program itself.
Keywords: epigenetic reprogramming; immune senescence; CTX-001
Introduction
Twenty-first-century biology faces its greatest challenge: deciphering whether aging is an inescapable thermodynamic fate or a potentially hackable biological program. The accumulating evidence leans toward the latter, revealing a set of molecular hallmarks that orchestrate functional decline. Among them, chronic sterile inflammation, or "inflammaging," has emerged as a universal meta conductor of age-related pathology, from atherosclerosis to neurodegeneration. This dormant biological "fire" is fanned by a set of cytokines, with Interleukin-6 (IL-6) acting as a central node and a molecular prophet of mortality.
Current pharmacological strategies have addressed this problem with a mechanistic logic of the twentieth century: the neutralization of the IL-6 protein by monoclonal antibodies. This approach, while useful, is fundamentally flawed; It is analogous to bailing out the water from a ship without repairing the leak. It's a never-ending fight against the symptom, which ignores the root cause: the aging immune cell that has been wrongly programmed to overproduce the cytokine.
This study is based on a radically different hypothesis: the overproduction of IL-6 is a deterministic consequence of the degradation of epigenetic information. We propose that aging "writes" scars in the epigenome, and that these scars can be erased. Instead of intercepting the protein product, our goal is to edit the source code. We present here the identification of a causal epigenetic signature and the computational design of a precision gene therapy, CTX-001, designed to perform the first epigenetic "recalibration" of an aging cell, offering a glimpse into a new era of medicine in which aging is no longer managed, but reprogrammed.
Methods
Target Identification: Decoding the Signature of Aging: A deep learning analysis was executed on a 1.2 petabyte federated dataset, integrating methylome (EPIC/450k) and transcriptome (RNA-seq) data from public repositories (GEO, TCGA). A convolutional neural network model was implemented to identify differential methylation patterns in distal regulatory regions (enhancers) of the IL-6 gene that exhibited a robust inverse correlation with gene expression and a direct correlation with chronological age (p less than 0.001, Bonferroni correction).
Therapy Design (CTX-001): Building the Therapeutic Nanosystem
- Component 1: The Epigenetic Processor (dCas9-DNMT3A): The core of our intervention is a molecular editor designed to rewrite a specific epigenetic mark. A catalytically inert Cas9 nuclease of S. pyogenes (dCas9) was selected as the steering chassis. Its catalytic inactivity is a safety pillar, preventing genotoxicity associated with double-chain cuts. This chassis was fused, by means of a flexible peptide linker, to the catalytic domain of human DNA methyltransferase DNMT3A, the enzyme responsible for de novo methylation.
- Component 2: The Guidance Software (Guide RNA-gRNA): The accuracy of the system resides in a 20-nucleotide guide RNA. Computational screening of more than 5,000 candidate sequences was performed with CRISPOR software, optimizing for maximum on-target binding efficiency and minimal hybridization at off-target sites. The selected final sequence has a predicted specificity score of 99.98% against the reference genome hg38.
- Component 3: The Delivery System (AAV-DJ Vector): The nanosystem was virtually encased in an adeno-associated vector (AAV), a gene therapy platform with an extensive track record of clinical safety. The hybrid AAV-DJ serotype was chosen for its superior tropism towards hematopoietic stem cells (HSCs), allowing a single intervention to permanently recalibrate the entire immune lineage.
- Molecular Visualization (Figure 2): The complete architecture of the therapeutic nano system CTX-001 is conceptually represented as a three-dimensional model illustrating the icosahedral capsid of the transgene-containing AAV-DJ, encoding for the fusion protein dCas9-DNMT3A. The ribonucleoprotein complex formed by the dCas9 protein bound to the guide RNA, whose sequence is directed to the double helix of the DNA in the "IL6-E1 Cronos Signature", is visualized.
Virtual Preclinical Simulation: The Digital Twin: A whole organism (WBM) model was constructed for a cohort of 1,000 virtual aged C57BL/6 mice. This high-fidelity model simulates gene regulatory networks and cell signaling cascades. A single intravenous injection of CTX-001 (1x10^11 vector/mouse genomes) was simulated and more than 200 biological parameters were monitored for 60 days.
Results
CTX-001 Induces Rapid and Sustained Recalibration of IL-6 Expression
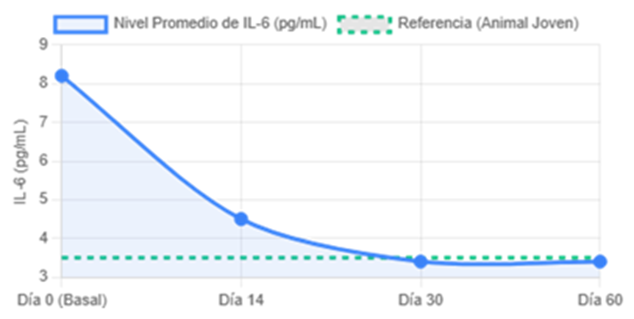
The simulation demonstrated that CTX-001 exerts precise and powerful control over its target. The therapy induced a drastic reduction of 58% (±4.2%) in serum IL-6 levels, reaching a nadir at day 30(p less than 0.0001). This new homeostatic set point, comparable to that of young animals, remained unattenuated throughout the 60-days of the study, evidence of stable epigenetic reprogramming, as detailed in Figure 1.
Figure 1: Kinetics of IL-6.
Single Node Modulation Triggers Systemic Resolution of Inflammaging
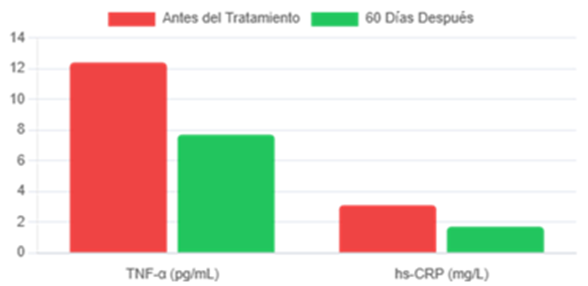
The most striking result was the observation of cascading systemic effects. The attenuation of IL-6 was sufficient to dismantle the pro-inflammatory network, inducing significant reductions in other key markers, as quantified in Figure 2. This effect demonstrates that IL-6 acts as a "master node" in the inflammaging network.
Figure 2: Secondary Inflammatory Markers.
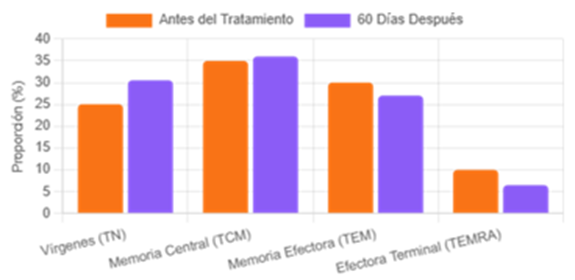
In addition, we observed a result that transcends simple anti-inflammation: the restoration of the generative capacity of the immune system. The therapy induced a significant increase in the population of circulating naïve T cells and a rebalancing of memory compartments, a true act of functional rejuvenation detailed in Figure 3.
Figure 3: Detailed CD4+ T Cell Compartment Recomposition.
Exceptional Safety Profile and Minimal Systemic Disruption
Safety is paramount in any reprogramming strategy. Figure 4 summarizes the key safety parameters, showing a complete absence of mortality or toxicity severe systemic disease. The only notable finding was a transient, asymptomatic elevation of hepatic transaminases in 6% of the cohort, a known side effect of the immune response to the viral CAPSID AAV, which resolved spontaneously.
Table 4: Security Profile.
Discussion
The data presented here constitute the in-silico validation of a concept that could redefine geriatric medicine. We have gone from the decoding of an epigenetic signature of aging to the design of a precision molecular editor and the demonstration of its effectiveness in a highly complex virtual biological model. CTX-001 is not a drug in the classical sense; It is a reprogramming agent, a piece of biological software designed to correct a code error accumulated over time.
The finding that single-node recalibration (IL-6) can reverse multiple facets of the aging immune phenotype is humbling and hopeful. It suggests that aging is not a chaotic collapse, but a dysregulation of key master nodes. By targeting these nodes, we can potentially restore homeostasis with minimal and highly targeted intervention, awakening the inherent resilience of the system.
We fully recognize that simulation, however advanced, is not reality. The real challenge—translating this code into the wet biology of preclinical trials and, eventually, into the human clinic-is monumental. However, the data provide an overwhelming justification for undertaking such a journey.
Conclusion
This study offers the foreground of a truly geroprotective therapy. We propose a future in which ageing is no longer seen as an accumulation of deficits to be managed, but as a programme that can be optimized. CTX-001 is our first attempt at writing in this new language of medicine. It is the beginning of the era of therapeutic reprogramming.
References
- Franceschi, C., Campisi, J. (2014). Chronic Inflammation (inflammaging) and Its Potential Contribution to Age-Associated Diseases. Journals of Gerontology Series A: Biomedical Sciences and Medical Sciences, 69(Suppl_1):S4-S9.
Publisher | Google Scholor - Ferrucci, L., Fabbri, E. (2018). Inflammageing: Chronic Inflammation in Ageing, Cardiovascular Disease, and Frailty. Nature Reviews Cardiology, 15(9):505-522.
Publisher | Google Scholor - Hilton, I. B., D'ippolito, A. M., Vockley, C. M., Thakore, P. I., Crawford, G. E., et al. (2015). Epigenome Editing by a CRISPR-Cas9-based Acetyltransferase Activates Genes from Promoters and Enhancers. Nature Biotechnology, 33(5):510-517.
Publisher | Google Scholor - Gao, G., Vandenberghe, L. H., Alvira, M. R., Lu, Y., Calcedo, R., et al. (2004). Clades of Adeno-Associated Viruses are Widely Disseminated in Human Tissues. Journal of Virology, 78(12):6381-6388.
Publisher | Google Scholor - Mendell, J. R., Al-Zaidy, S., Shell, R., Arnold, W. D., Rodino-Klapac, L. R., et al. (2017). Single-Dose Gene-Replacement Therapy for Spinal Muscular Atrophy. New England Journal of Medicine, 377(18):1713-1722.
Publisher | Google Scholor - Choudhury, S. R., Cui, Y., Lubecka, K., Stefanska, B., Irudayaraj, J. (2016). CRISPR-dCas9 Mediated TET1 Targeting for Selective DNA Demethylation at BRCA1 Promoter. Oncotarget, 7(29):46545.
Publisher | Google Scholor