Research Article
Post-Surgical Evaluation of Quality of Life in Patients with Oral Cancer: An Intervention Study from India
1Department of Molecular Oncology, Atal Bihari Vajpayee Regional Cancer Centre, Agartala, Tripura, India.
2Department of Nursing, Desh Bhagat University, Gobindgarh, Punjab, India.
3Department of Microbiology & Public Health, ICMR-Regional Medical Research Centre, Bhubaneswar, Odisha, India.
*Corresponding Author: Ankita Debnath, Department of Nursing, Desh Bhagat University, Gobindgarh, Punjab, India.
Citation: Nath S, Falwaria K, Debbarma S, Pattnaik M, Debnath A. (2025). Post-Surgical Evaluation of Quality of Life in Patients with Oral Cancer: An Intervention Study from India, Journal of BioMed Research and Reports, BioRes Scientia Publishers. 7(5):1-9. DOI: 10.59657/2837-4681.brs.25.152
Copyright: © 2025 Ankita Debnath, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: March 28, 2025 | Accepted: April 11, 2025 | Published: April 18, 2025
Abstract
Introduction: The emotional, physical, and functional well-being of patients with oral cancer is adversely impacted after surgery. These traumatic experiences may leave the patients with severe life scars and a sharp decline in their quality of life. The study aimed to evaluate the impacts of comprehensive nursing intervention on postoperative quality of life among patients with oral cancer.
Methods: An interventional study was conducted on 190 postoperative patients with oral cancer. A validated quality of life questionnaire from the European Organization for the Research and Treatment of Cancer was used to evaluate the patients' quality of life.
Results: Among the demographic variables, there was a statistically significant difference in terms of cancer stage (p<0.01) and tumor metastasis (p=0.01). The experimental group experienced a significant increase in their quality of life (p<0.01) compared to the control group.
Conclusion: Such comprehensive nursing interventions would improve quality of life by reducing and controlling the associated emotional, physical, and functional well-being. It is recommended that these kinds of interventions be used to assist patients in managing the temporary deterioration that occurs after surgery.
Keywords: postoperative oral cancer; quality of life; comprehensive nursing intervention
Introduction
Oral squamous cell carcinoma (OSCC) is the 17th most frequent cancer worldwide, accounting for over 90% of all malignant neoplasms of the oral cavity. It is the most prevalent cancer among men in India, but it is also the third most common cancer overall when considering both genders together. It also comes in third among all the cancer kinds that cause death in the country (Nethan et al., 2022). Small, unusual, unexplained growths or sores in the mouth—which include the lips, cheeks, sinuses, tongue, hard and soft palates, and the area from the base of the mouth to the oropharynx - are indicative of oral cancer. India reports over 52,000 deaths and 77,000 new cases every year or about one-fourth of all cases worldwide (Borse et al., 2020).
Treatment for oral cancer varies depending on the cancer's stage, as identified by a clinical diagnosis. Local radiation therapy, chemotherapy, and surgical removal, either alone or in conjunction with other techniques, are the primary therapeutic modalities. Despite a decline in the number of deaths, patients with oral and oropharyngeal cancer have physical and psychological complications from treatment (Pingili et al., 2021). Tumor removal, along with or without dissection of the neck lymph nodes, is the surgical treatment for oral cancer. This method can stop distant metastases from the buccal or lingual lymph nodes of the neck by simultaneously removing the main tumor and the neck lymph nodes. However, this procedure might damage a patient's face, neck, or mandible, which would be detrimental to their postoperative self-image and their capacity to open their mouth, swallow, chew, and speak. With surgical treatment, patients' quality of life (QOL) typically declines noticeably. Advanced tumors necessitate a large amount of surgical resection, which is linked to poor quality of life results (Wang et al., 2020).
According to the World Health Organization (WHO), there are numerous ways in which health can impact one's quality of life, including one's physical and mental well-being, degree of freedom, social connections, and associations with notable environmental elements. With the aid of the health-related QOL evaluation, clinicians can better predict patient outcomes and gain an appreciation for the effects that therapies have on the patient's body, mind, and social life. The QOL Questionnaire employs a methodical approach to compile and prioritize items related to patients' concerns. Validity, reliability, consistency, repeatability, and sensitivity to change are the primary requirements that they must meet (Mahalingam et al., 2022). To meet these demands, the European Organization for Research and Treatment of Cancer (EORTC) Quality of Life Group produced a set of questionnaires that were utilized in the current study. The EORTC QLQ-C30 (version 3.0), the core quality of life questionnaire, is a cancer-specific tool that can be used by a wide variety of cancer patients (Husson et al., 2020). The earlier studies supported the validity, reliability, and acceptability of the questionnaire (EORTC QLQ-C30, version 3.0) as a tool for assessing the QOL of cancer patients and suggested its application in clinical settings (Davda et al., 2021; Cocks et al., 2023).
Although many studies have reported the QOL of patients with oral cancer, there is a dearth of studies evaluating the QOL of postoperative oral cancer patients in India, which prompted us to carry out this study. The study aimed to evaluate the impacts of comprehensive nursing intervention on postoperative quality of life among patients with oral cancer.
Materials and Methods
Participants
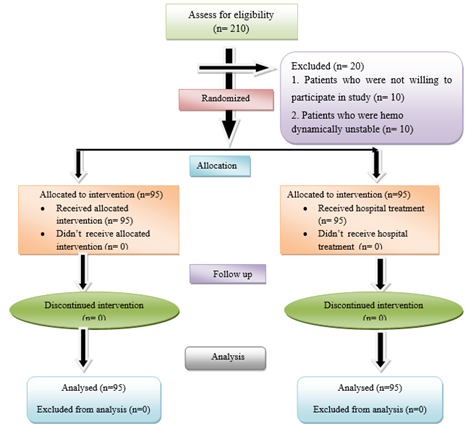
The study was conducted at the Atal Bihari Vajpayee Regional Cancer Centre, Agartala, Tripura, India. The interventional study included 190 postoperative patients with oral cancer. The inclusion criteria of this study were as follows: 1) Patients performed surgery at the Atal Bihari Vajpayee Regional Cancer Centre from 2022 to 2023, 2) patients were diagnosed as stage I - IV before surgery; 3) patients were over 20 years of age 4) the QOL of the patients were evaluated by the EORTC QLQ C30 scale. The study’s exclusion criteria were as follows: 1) under 20 years old, with cognitive and mental disorders, and with a history of other tumors. The study was conducted in accordance with the Declaration of Helsinki and was approved by the Institutional Review Board of Desh Bhagat University, Punjab, India (DBU/RC/2023/2338). Informed written consent was obtained from each patient. The flowchart for the patient enrolment selection procedure is displayed in Figure 1.
Figure 1: The flowchart for the patient enrollment selection procedure.
Demographics and Clinical Characteristics
Participants were categorized based on their age, gender, religion, educational status, occupation, monthly income, marital status, types of surgery, cancer stage, tumor metastasis, and primary site.
Comprehensive Nursing Intervention
Comprehensive nursing intervention includes helping with relaxation techniques, mouth opening exercises, active and passive range of motion, stretching exercises, maintaining good posture, chin tucks, and shoulder blade squeezes by research personnel, in addition to offering educational support on the use of thyme honey, dental care, and counseling among postoperative patients with oral cancer. A PowerPoint presentation and a video featuring the experimental group are used to accomplish this. The educational intervention lasted thirty minutes, while the nursing intervention lasted ten to fifteen minutes. The control group was instructed to adhere to the hospital's standard of care. For five days in a row, it was followed nine to ten times a day, and the patient exercised as needed.
Health-Related QOL Assessment
The following questionnaires were used to measure QOL. The QOL was assessed using the validated Bengali (local language) and English versions of the EORTC QLQ-C30.
The EORTC QLQ-C30 (Version 3.0)
The European Organization for Research and Treatment of Cancer (EORTC) QLQ-C30 is a standard questionnaire used to assess the QOL of cancer patients. It has thirty items that draw from six functioning and several symptom subscales. Each subscale has a value between 0 and 100; higher scores for functional subscales imply better circumstances and vice versa for symptoms.
Statistical Analysis
The statistical package for social sciences (SPSS) with version 25 was utilized to analyze the data. Calculations were made for the variables' percentages and frequency distributions. Analyzing descriptive data involved using the Chi-square or Fisher's exact tests. T-tests on their own and Wilcoxon's test were used to assess the data. An analysis was considered statistically significant if the p-value was less than 0.05.
Results
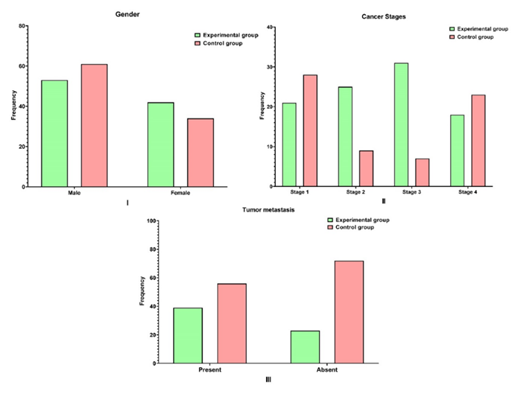
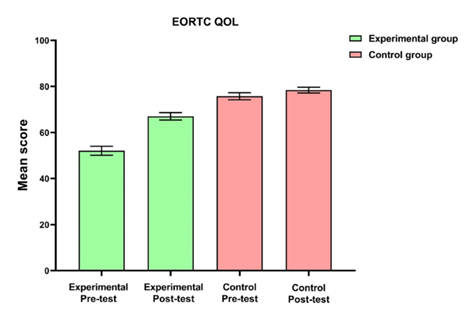
The demographic and clinical characteristics of the experimental and control groups are shown in Table 1. A total of 190 patients were included in this study. Of these participants, 114 were males (95% CI: 52.6-66.9) and 76 were females (95% CI: 33.0-47.3) for each group, respectively, resulting in a male-to-female ratio of 1.5:1. There was a statistically significant difference in terms of cancer stage (p lessthan 0.01) and tumor metastasis (p lessthan 0.01). Descriptive characteristics of pre-and post-test levels of QOL in enrolled patients are summarized in Table 2. Table 3 summarizes the mean and standard deviation of the pre-and post-test levels of quality of life among postoperative patients with oral cancer. Figure 2 shows the gender, cancer stages, and tumor metastasis distribution of patients in the experimental and control groups. After the nursing intervention, the experimental group experienced a significantly higher QOL (p lessthan 0.01) than that of the control group (Figure 3).
Table 1: Socio-demographic and clinical characteristics of the participants (n=190).
| Variables | Characteristics | Experimental Group (n=95) n (%) | Control Group (n=95) n (%) | p-Value |
| Age (Years) | 21-30 | 0 | 0 | 0.06
|
| 31-40 | 8 (8.4) | 13 (13.7) | ||
| 41-50 | 38 (40.0) | 21 (22.1) | ||
| 51-60 | 27 (28.4) | 31 (32.6) | ||
| 61-70 | 22 (23.25.3) | 30 (31.6) | ||
| Gender | Male | 53 (55.8) | 61 (64.2) | 0.23 |
| Female | 42 (44.2) | 34 (35.8) | ||
| Religion | Hindu | 78 (82.1) | 82 (86.3) | 0.21 |
| Muslim | 8 (8.4) | 7 (7.4) | ||
| Christian | 5 (5.3) | 6 (6.3) | ||
| Others | 4 (4.2) | 0 | ||
| Educational Status | No formal education | 20 (21.1) | 24 (25.3) | 0.30
|
| Primary | 45 (47.4) | 35 (36.8) | ||
| Secondary | 30 (31.6) | 29 (30.5) | ||
| Higher secondary | 0 | 7 (7.4) | ||
| Graduate and above | 0 | 0 | ||
| Occupation | Govt | 2 (2.1) | 0 | 0.14
|
| Private | 10 (10.5) | 8 (8.4) | ||
| Self employed | 22 (23.2) | 31 (32.6) | ||
| Daily wager | 18 (18.9) | 25 (26.3) | ||
| Unemployed | 43 (45.3) | 31 (32.6) | ||
| Monthly Income (Rs) | ≤Rs.10, 000 | 49 (51.6) | 22 (23.2) | 0.21
|
| 10, 001-15,000 | 41 (43.2) | 51 (53.7) | ||
| 15, 001-20,000 | 3 (3.2) | 20 (21.1) | ||
| >20,000 | 2 (2.1) | 2 (2.1) | ||
| Marital Status | Single | 5 (5.3) | 3 (3.2) | 0.28 |
| Married | 82 (86.3) | 89 (93.7) | ||
| Widow | 6 (6.3) | 3 (3.2) | ||
| Divorced | 2 (2.1) | 0 | ||
| Types of Surgery | Tumor Resection | 6 (6.3) | 3 (3.2) | 0.30 |
| Micrographic surgery | 2 (2.1) | 0 | ||
| Glossectomy surgery | 17 (17.9) | 20 (21.1) | ||
| Mandibulectomy surgery | 47 (49.5) | 57 (60.0) | ||
| Maxillectomy surgery | 22 (23.2) | 14 (14.7) | ||
| Neck Dissection | 1 (1.1) | 1 (1.1) | ||
| Cancer Stage | I | 21 (22.1) | 51 (23.7) | lessthan 0.01* |
| II | 25 (26.3) | 28 (29.5) | ||
| III | 31 (32.6) | 9 (9.5) | ||
| IV | 18 (18.9) | 7 (7.4) | ||
| Tumor Metastasis | Yes | 39 (41.1) | 23 (24.2) | 0.01* |
| No | 56 (58.9) | 72 (75.8) | ||
| Primary Site | Lip | 7 (7.4) | 4 (4.2) | 0.37 |
| Buccal Mucosa | 62 (65.3) | 48 (50.5) | ||
| Hard Palate | 3 (3.2) | 5 (5.3) | ||
| Posterior molar Region | 2 (2.1) | 4 (4.2) | ||
| Tongue | 12 (12.6) | 17 (17.9) | ||
| Floor of mouth | 1 (1.1) | 4 (4.2) | ||
| Angle of mouth | 3 (3.2) | 1 (1.1) | ||
| Submandibular gland | 1 (1.1) | 2 (2.1) | ||
| Base of tongue | 3 (3.2) | 4 (4.2) | ||
| Maxilla | 0 | 1 (1.1) | ||
| Cheek | 0 | 2 (2.1) | ||
| Alveolus | 1 (1.1) | 3 (3.2) |
Table 2: Descriptive characteristics of pre and post-test level of quality of life in enrolled patients (n=190).
| Variables | Characteristics | Experimental Group | Control Group | ||||
| Pre-Test Mean ± SD | Post Test Mean ± SD | p-value | Pre-Test Mean ± SD | Post Test Mean ± SD | p-Value | ||
| EORTC QLQ-C30 (Item No.1-28) | |||||||
| Very High Quality of Life | 84.5 ± 4.6 | 82.9 ± 4.8 | lessthan 0.01* | 85.5 ± 3.9 | 84.7 ± 4.4 | 0.12 | |
| High Quality of Life | 61.4 ± 7.2 | 62.1 ± 7.3 | 64.7 ± 6.8 | 64.0 ± 7.7 | |||
| Average Quality of Life | 44.0 ± 6.3 | 38.6 ± 6.9 | 45.3 ± 6.6 | 40.2 ± 6.8 | |||
| Low Quality of Life | 23.8 ± 1.3 | 0 | 0 | 0 | |||
| EORTC QLQ-C30 (Item No.29,30) | |||||||
| Very Poor | 0 | 0 | lessthan 0.01* | 0 | 0 | 0.07 | |
| Bad | 0 | 3.8 ± 0.4 | 3.8 ± 0.3 | 3.7 ± 0.5 | |||
| Fairly Bad | 5.5 ± 0.5 | 6.0 ± 0 | 5.6 ± 0.4 | 5.7 ± 0.4 | |||
| Same as before | 7.6 ± 0.5 | 7.8 ± 0.4 | 7.4 ± 0.4 | 7.5 ± 0.5 | |||
| Fairly Good | 9.5 ± 0.5 | 9.7 ± 0.4 | 9.1 ± 0.3 | 9.2 ± 0.5 | |||
| Good | 11.5 ± 0.5 | 11.3 ± 0.5 | 11.0 ± 0 | 11.0 ± 0 | |||
| Excellent | 14.0 ± 0 | 14.0 ± 0 | 0 | 0 | |||
Table 3: Mean and SD of pre and post-test level of quality of life among postoperative patients with oral cancer (n=190).
| Experimental (n=95) | Dimension Of Quality of life | Max Score | Pre test | Post test | Wilcoxon’s Test | |
| Mean ± SD | Mean ± SD | Z-value | P-value | |||
| Physical Function | 20 | 12.7 ± 4.3 | 9.1 ± 4.1 | -6.5 | lessthan 0.01* | |
| Role Function | 8 | 5.6 ± 1.6 | 5.4 ± 1.5 | -1.4 | 0.15 | |
| Cognitive Function | 8 | 5.6 ± 1.8 | 5.7 ± 1.7 | -0.7 | 0.46 | |
| Emotional Status | 16 | 10.8 ± 3.4 | 8.3 ± 4.0 | -5.2 | lessthan 0.01* | |
| Social Functioning | 12 | 7.6 ± 3.1 | 6.1 ± 3.2 | -3.8 | lessthan 0.01* | |
| General Symptoms (fatigue, nausea / vomiting and pain) | 48 | 24.3 ± 7.6 | 17.4 ± 9.3 | -7.5 | lessthan 0.01* | |
| Over All | 112 | 67.1 ± 15.9 | 52.1 ± 17.0 | -8.3 | lessthan 0.01* | |
| Over All Global Health Status | 14 | 9.7 ± 1.7 | 8.0 ± 2.1 | -6.9 | lessthan 0.01* | |
| Control (n=95) | Physical Function | 20 | 16.4 ± 2.9 | 15.9 ± 3.3 | -1.8 | 0.10 |
| Role Function | 8 | 6.6 ± 1.2 | 6.3 ± 1.5 | -1.1 | 0.25 | |
| Cognitive Function | 8 | 6.7 ± 1.4 | 6.6 ± 1.7 | -1.7 | 0.08 | |
| Emotional Status | 16 | 13.0 ± 2.7 | 12.7 ± 2.7 | -1.2 | 0.22 | |
| Social Functioning | 12 | 10.3 ± 1.8 | 9.5 ± 2.4 | -2.6 | lessthan 0.01* | |
| General Symptoms (fatigue, nausea / vomiting and pain) | 48 | 25.8 ± 6.9 | 26.3 ± 3.7 | -0.1 | 0.85 | |
| Over All | 112 | 78.4 ± 12.2 | 77.9 ± 13.8 | -1.4 | 0.12 | |
| Over All Global Health Status | 14 | 7.2 ± 1.5 | 6.9 ± 1.7 | -1.6 | 0.09 | |
Figure 2: Gender, cancer stages and tumor metastasis distribution of patients in the experimental and control group.
Figure 3: Mean and standard error of mean of EORTC QOL of experimental and control group
Discussion
The QOL is a broad phrase that encompasses various "domains" of life, including social, physical, emotional, and functional, which are very intimately related while frequently being overlapped. One important predictor of survival for cancer patients is their QOL. It helps medical personnel better attend to the requirements of their patients. The QOL is a fundamental factor that influences the decisions that physicians make since it aids in the assessment of treatment outcomes in terms of mortality, survival, and success rates (Sharma et al., 2022). The QOL assessment is a helpful supplement to the more conventional methods of evaluating the efficacy of treatments for patients with oral cancer. Oral cancer is still linked to deformities and functional impairments that impact important areas of life, even with recent advancements in diagnosis and treatment. These factors explain why oral health-related QOL evaluations are crucial for patients receiving treatment for oral cancer (Ogino et al., 2021). Providing supportive care is essential for enhancing quality of life. The QOL evaluation ought to serve as an indicator of the effectiveness of interdisciplinary therapy and highlight areas where the impacted patient needs assistance (Scotté et al., 2023).
Head and neck cancer patients cannot hide post-treatment functional changes and must therefore deal with the ensuing negative impact on self-esteem and confidence in all domains. While body scars and alterations are typically hidden during social activities, dramatic situations like a permanent colostomy or vascular shunt for dialysis can be easily managed in public. There is consistently a large disparity in opinions about how post-operative deficits affect QOL on a daily basis (Henry et al., 2022). This could also be connected to the functional and aesthetic impairments that follow or to the requirement for additional surgery, which could worsen the symptoms and psychological state (De Cicco et al., 2021).
Research indicates that the most frequent adverse effects that patients with oral cancers have during treatment, particularly following surgery, include pain, fatigue, and sleep disturbance. The most common symptom for 70% of patients with oral cancer is pain. Patients show more fatigue following increasingly complicated surgical operations, which affects their QOL and increases their risk of developing mental disorders, including depression and anxiety. The reasons behind these postoperative physical and psychological problems are surgical wounds, the stress of the disease, and the interventions (Loh et al., 2022). A recent change in the focus of health measurement has moved beyond conventional health indicators such as mortality and morbidity. Holistic treatment planning requires the inclusion of markers to determine how disease and impairment affect daily activities and behavior, subjective health, and disability or functional status (Ramalingam et al., 2023). Advanced cancer patients suffer from poor QOL (Liu et al., 2022). Many of the structures required for proper articulation are affected by oral surgery. Surgery primarily affects issues with eating, swallowing, and speaking (Goetz et al., 2020). Optimizing the QOL of cancer patients through improved clinical symptoms is best achieved through comprehensive nursing (Wang et al., 2022).
Our research confirms the previous study's findings that adults over 40 are the most likely age group to be impacted by OSCC (Khani et al., 2022). The incidence increased after the age of 40, with the 41-50 and 51-60 age groups in both the experimental and control groups exhibiting the highest incidence, followed by the 61-70 age group. In terms of gender, our research revealed that men seem to be impacted by oral cancer nearly twice as much as women, with higher rates of mortality from alcohol and tobacco use which was also noted by the previous study (Di Spirito et al., 2022). In terms of religion, Hindus had a greater incidence of oral cancer, which the earlier Indian study also supported (Sozio et al., 2021). Since the majority of oral cancer patients were observed in primary schools, our study found a correlation between a low educational level and an increased risk of oral cancer. This correlation may be partially explained by the limited availability of information about oral cancer in general, including diagnosis and treatment. We are supported by a previous study that found those with only primary education were more likely to have a poor result connected to oral cancer than people with higher or university education (Ndayisabye et al., 2022).
In terms of occupation, our research revealed that unemployed people were more likely to acquire oral cancer. This could be because jobless people might find it more difficult to meet their basic necessities financially and might have less access to healthcare. In addition to causing stress and psychological malaise, unemployment can also cause people to engage in unhealthy behaviors that are known to be risk factors for diseases, accidents, and fatalities, as the previous study also noted (Freire et al., 2021). Because the majority of our patients came from lower-income households, our study also demonstrated a relationship between oral cancer and low individual or household income, which was corroborated by another study (Singh et al., 2019). As for marital status, we found that married people had a higher risk of oral cancer, which was also shown in the previous study. This finding may have been due to the fact that single people are often younger, have a delayed diagnosis, or are not at the age at which oral cancer is usually diagnosed (Curtis et al., 2020).
The most common surgical technique among the enrolled patients with oral cancer was a mandibulectomy. The prior study likewise revealed comparable results in addition to the open reduction and internal fixing (Moshy et al., 2019). Since stage I oral cancer is quite treatable, patients should seek treatment as soon as possible. In terms of cancer stages, stage I was more common in our study than in any other stage (stages I-IV). This was connected with tumor metastasis because the majority of patients in our study could be classified as stage I because they did not have tumor metastases. The most common location for OSCC in Southeast Asia is the buccal mucosa. This may be due to the growing acceptance of smokeless tobacco products such as areca nuts and betel quid in this region (Al-Rawi et al., 2023). Since most of the patients who were enrolled had cancer of the buccal mucosa, our analysis supports their findings.
In our study, the experimental group's quality of life significantly improved after the nursing intervention in comparison to the control group. Comprehensive nursing intervention is an effective treatment option that results in obvious improvements in the patients' quality of life, mood, satisfaction, and treatment outcomes for patients with postoperative oral cancer. Comprehensive nursing differs from conventional nursing in that it systematizes the nursing approach, places the patient at the center, outlines the nursing philosophy and duties, offers patients scientifically-based, high-quality, and effective nursing care, and adapts the "customized" nursing plan to the patient's changing needs in order to provide a comfortable nursing service that aids in the patient's recovery.
For the five functional scales (physical, role, emotional, cognitive, and social) and the three symptom scales (fatigue, nausea/vomiting, and pain), the lower the mean scores, the greater the quality of life. In contrast, a higher mean score corresponds to a better global health status. In our study to investigate possible variations in the EORTC QLQ-C30 scale mean scores between the two groups, independent t-tests were calculated. The findings showed that, in comparison to the pre-test and control group, the overall mean score of nearly all QOL subscales, including physical function, role function, cognitive function, emotional status, social functioning, and general symptoms (pain, fatigue, nausea/vomiting, and so on), was significantly lower after the post-test, indicating a better quality of life. In contrast, the mean score for overall global health status was significantly higher. When comparing postoperative oral cancer patients' QOL to both the pre-test and control groups, the comprehensive nursing intervention was found to have a significant positive impact. Similar findings were reported in earlier studies as well (Lu et al., 2021; Khantwal et al., 2021).).
Further research is warranted to explore the correlation between comprehensive nursing interventions and QOL for different ethnic groups and diseases. Furthermore, such interventional studies may be carried out in light of the current study's findings to assess whether data on patients' care requirements and QOL can be used to better support patients' interactions with family and the community during oral cancer treatment and recovery. However, the study has a few limitations. The study was confined to a single centre. There could be an effect on the results because the follow-up period was five days. There's a chance that treatment groups don't accurately represent all cancer patients, especially the more advanced cases. Furthermore, because patient QOL may vary based on lifestyle and support alternatives, it is urged that the current study be performed in different parts of the nation, as the researchers were unable to locate any comparable studies carried out in India for comparison.
Conclusion
In conclusion, when making decisions for patients with postoperative oral cancer, quality of life needs to be taken into account as a significant outcome indicator. The success of therapeutic procedures and treatment has been directly correlated with the patients' quality of life in recent years. The experimental group's quality of life significantly improved as a result of the nursing intervention. Patients' functional activities were found to have dramatically improved using relaxation techniques, which in turn were indicated to have greatly boosted the quality of life for the experimental group. Thus, it is imperative to apply these nursing interventions in clinical practice to mitigate a variety of symptoms and unfavorable outcomes related to cancer and its treatment, as well as to improve patients' quality of life and understanding.
Relevance for Clinical Practice
A comprehensive nursing intervention can be utilized in routine practice to reduce functional impairments following surgery, which in turn can enhance the quality of life in postoperative oral cancer patients. The fields of nursing practice, nursing administration, nursing education, and nursing research are critically affected by the implications that the researcher has uncovered. In order to determine their requirements, nurses and other healthcare professionals can take part in staff development programs that are practice- and evidence-based. Workshops, symposiums, and skill labs can be arranged by educational institutions to address and manage one's quality of life. Nurse administrators should provide funding to uphold acceptable work environments and a happy work environment, in addition to strengthening hospital policies. Generalizing the study's conclusions is made possible by its replication. Publication of research findings in scholarly journals and online will help ensure that they are applied as effectively as possible.
Declarations
Data Availability
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Funding
Nil.
Conflict of Interest
The authors declare no conflict of interest.
Acknowledgment
We acknowledge all the patients who contributed to the study.
Ethics Approval Statement
The study was conducted in accordance with the Declaration of Helsinki, and this research was approved by Desh Bhagat University’s Institutional Review Board (DBU/RC/2023/2338). The research was conducted at Atal Bihari Vajpayee Regional Cancer Centre, Agartala, Tripura, India.
Patient Consent Statement
Informed consent was obtained from all individual participants included in the study.
References
- Al-Rawi, N.H., Hachim, I.Y., Hachim, M.Y., Salmeh, A., Uthman, A.T., et al. (2023). Anatomical Landscape of Oral Squamous Cell Carcinoma: A Single Cancer Center Study in UAE. Heliyon, 9(5):e15884.
Publisher | Google Scholor - Borse, V., Konwar, A.N., Buragohain, P. (2020). Oral Cancer Diagnosis and Perspectives in India. Sens Int, 1:100046.
Publisher | Google Scholor - Cocks, K., Wells, J.R., Johnson, C., Schmidt, H., Koller, M., et al. (2023). Content Validity of The EORTC Quality of Life Questionnaire QLQ-C30 for Use in Cancer. Eur J. Cancer, 178:128-138.
Publisher | Google Scholor - Curtis, D.C., Eckhart, S.C., Morrow, A.C., Sikes, L.C., Mridha, T. (2020). Demographic and Behavioral Risk Factors for Oral Cancer among Florida Residents. J Int Soc Prev Community Dent, 10(3):255-261.
Publisher | Google Scholor - Davda, J., Kibet, H., Achieng, E., Atundo, L., Komen, T. (2021). Assessing The Acceptability, Reliability, And Validity of The EORTC Quality of Life Questionnaire (QLQ-C30) in Kenyan Cancer Patients: A Cross-Sectional Study. J Patient Rep Outcomes, 5(1):4.
Publisher | Google Scholor - De Cicco, D., Tartaro, G., Ciardiello, F., Fasano, M., Rauso, R., et al. (2021). Health-Related Quality of Life in Oral Cancer Patients: Scoping Review and Critical Appraisal of Investigated Determinants. Cancers (Basel), 13(17):4398.
Publisher | Google Scholor - Di Spirito, F., Amato, A., Romano, A., Dipalma, G., Xhajanka, E., et al. (2021). Analysis of Risk Factors of Oral Cancer and Periodontitis from a Sex- and Gender-Related Perspective: Gender Dentistry. Applied Sciences, 12(18):9135.
Publisher | Google Scholor - Freire, A.R., Freire, D.E.W.G., de Araújo, E.C.F., de Almeida, Carrer, F.C., et al. (2021). Socioeconomic Indicators and Economic Investments Influence Oral Cancer Mortality in Latin America. BMC Public Health, 21(1):377.
Publisher | Google Scholor - Goetz, C., Raschka, J., Wolff, K.D., Kolk, A., Bissinger, O. (2020). Hospital Based Quality of Life in Oral Cancer Surgery. Cancers (Basel), 12(8):2152.
Publisher | Google Scholor - Henry, M., Albert, J.G., Frenkiel, S., Hier, M., Zeitouni, A., et al. (2022). Body Image Concerns in Patients with Head and Neck Cancer: A Longitudinal Study. Front Psychol. 13:816587.
Publisher | Google Scholor - Husson, O., de Rooij, B.H., Kieffer, J., Oerlemans, S., Mols, F., et al. (2020). The EORTC QLQ-C30 Summary Score as Prognostic Factor for Survival of Patients with Cancer in the “Real-World”: Results from the Population-Based PROFILES Registry. Oncologist, 25(4):e722-e732.
Publisher | Google Scholor - Khani Jeihooni, A., Jafari, F. (2022). Oral Cancer: Epidemiology, Prevention, Early Detection, and Treatment. IntechOpen.
Publisher | Google Scholor - Khantwal, G., Sharma, S.K., Rani, R., Agarwal, S.P. (2021). Effect of Postsurgical Nurse-led Follow-Ups on Quality of Life in Head-and-Neck Cancer Patients: A Pilot Randomized Controlled Trial. Asia Pac J Oncol Nurs, 8(5):573-580.
Publisher | Google Scholor - Liu, Y., Cao, X., Zhao, X., Shi, X., Lei, M., et al. (2022). Quality of Life and Mental Health Status Among Cancer Patients with Metastatic Spinal Disease. Front Public Health, 10:916004.
Publisher | Google Scholor - Loh, E.W., Shih, H.F., Lin, C.K., Huang, T.W. (2022). Effect of Progressive Muscle Relaxation on Postoperative Pain, Fatigue, And Vital Signs in Patients with Head and Neck Cancers: A Randomized Controlled Trial. Patient Educ Couns, 105(7):2151-2157.
Publisher | Google Scholor - Lu, J., Xiao, D., Sun, J., Huang, J. (2021). Effect of Comprehensive Nursing on The Appearance and Recovery Effect of Oral Squamous Cell Carcinoma Patients. Am J Transl Res, 13(5):5519-5525.
Publisher | Google Scholor - Mahalingam, M., Thiruneelakandan, S., Annamalai, T., Pavithran, V. (2022). Quality of health assessment in oral cancer patients postoperatively - A retrospective study. Advances in Oral and Maxillofacial Surgery, 5:100202.
Publisher | Google Scholor - Moshy, J.R., Sohal, K.S., Shaban, S.D., Owibingire, S.S., Shubi, F.M., et al. (2019). The Spectrum of Oral and Maxillofacial Surgical Procedures at The National Referral Hospital in Tanzania from 2013 to 2017. Ann Res Hosp, 3:3.
Publisher | Google Scholor - Ndayisabye, H., Ndagijimana, A., Biracyaza, E., Umubyeyi, A. (2022). Factors Associated with Oral Cancer Adverse Outcome at the Rwanda Military Hospital, a Retrospective Cross-Sectional Study. Front Oral Health, 3:844254.
Publisher | Google Scholor - Nethan, S.T., Ravi, P., Gupta, P.C. (2022). Epidemiology of Oral Squamous Cell Carcinoma in Indian Scenario. In: Routray, S. (eds) Microbes and Oral Squamous Cell Carcinoma. Springer, Singapore.
Publisher | Google Scholor - Ogino, N., Funayama, A., Niimi, K., Mikami, T., Inoue, M., et al. (2021). Evaluation of Factors Affecting Health-Related Quality of Life in Patients Treated for Oral Cancer. Advances in Oral and Maxillofacial Surgery, 2:100057.
Publisher | Google Scholor - Pingili, S., Ahmed, J., Sujir, N., Shenoy, N., Ongole, R. (2021). Evaluation of Malnutrition and Quality of Life in Patients Treated for Oral and Oropharyngeal Cancer. Scientific World Journal, 9936715.
Publisher | Google Scholor - Ramalingam, K., Krishnan, M., Ramani, P., Muthukrishnan, A. (2023). Quality of Life Assessment with European Organisation for Research and Treatment of Cancer Questionnaire (Head and Neck Module 43) and Its Clinicopathological Correlation Among Patients Treated for Oral Squamous Cell Carcinoma: An Exploratory Study. Cureus, 15(2):e34650.
Publisher | Google Scholor - Scotté, F., Taylor, A., Davies, A. (2023). Supportive Care: The
Publisher | Google Scholor - Sharma, K., Neemawat, K., Sharma, A. (2022). Assessment of Quality of Life for Oral Cancer Patients Receiving Chemotherapy Treatment at a Government Tertiary Care Center. Dentistry and Medical Research, 10(2):55-59.
Publisher | Google Scholor - Singh, A., Peres, M.A., Watt, R.G. (2019). The Relationship between Income and Oral Health: A Critical Review. J Dent Res, 98(8):853-860.
Publisher | Google Scholor - Sozio, S.J., Jhawar, S., Wang, Y., Sayan, M., Parikh, R., et al. (2021). Comparing the Incidence of Buccal Mucosa Cancer in South Asian, White, and Black Populations Residing in the United States: A Cross-Sectional Analysis. Asian Pac J Cancer Prev, 22(1):195-199.
Publisher | Google Scholor - Wang, M., Sun, Y., Zhang, M., Yu, R., Fu, J. (2022). Effects of High-Quality Nursing Care on Quality of Life, Survival, and Recurrence in Patients with Advanced Non-Small Cell Lung Cancer. Medicine (Baltimore), 101(37):e30569.
Publisher | Google Scholor - Wang, T.F., Li, Y.J., Chen, L.C., Chou, C., Yang, S.C. (2020). Correlation Between Postoperative Health-Related Quality of Life and Care Needs of Oral Cancer Patients. Cancer Nurs, 43(1):12-21.
Publisher | Google Scholor