Research Article
Optimizing Gram Stain Interpretation: A Comparison of Four Smear Preparation Methods for Gram Staining of Positively Automated Blood Culture Bottles
1Senior Resident, Department of Microbiology, University College of Medical Sciences, Dilshad Garden, Delhi, India.
2Associate Professor, Department of Microbiology, University College of Medical Sciences, Dilshad Garden, Delhi, India.
3Post Graduate [PG-3], Department of Microbiology, University College of Medical Sciences, Dilshad Garden, Delhi, India.
4Post Graduate [PG-2], Department of Microbiology, University College of Medical Sciences, Dilshad Garden, Delhi, India.
5HOD & Director Professor, Microbiology Department, University College of Medical Sciences, Dilshad Garden, Delhi, India.
*Corresponding Author: Kirti Nirmal, Associate Professor, Department of Microbiology, University College of Medical Sciences, Dilshad Garden, Delhi, India.
Citation: Nirmal S, Nirmal K, Jain N, Banyopadhyay A, Das S. (2025). Optimizing Gram Stain Interpretation: A Comparison of Four Smear Preparation Methods for Gram Staining of Positively Automated Blood Culture Bottles, International Journal of Biomedical and Clinical Research, BioRes Scientia Publishers. 4(4):1-7. DOI: 10.59657/2997-6103.brs.25.097
Copyright: © 2025 Kirti Nirmal, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: September 24, 2025 | Accepted: October 10, 2025 | Published: October 15, 2025
Abstract
Background: Rapid and accurate Gram stain interpretation of positively flagged blood culture broths is essential for early identification of bloodstream infections and timely initiation of empirical therapy. However, artifacts caused by resin and charcoal particles in culture media can compromise diagnostic clarity. This study evaluates and compares four smear preparation methods-conventional, water wash, blood film, and drop and rest-for optimizing Gram stain interpretation.
Methods: A prospective study was conducted on 100 positively flagged blood culture bottles. Each sample was used to prepare smears using all four techniques- conventional, water wash, blood film and drop & rest method. These were evaluated for agreement with the final culture-based Gram stain results. Interfering resin/charcoal particles were graded on a standardized scale. Kappa (κ) statistics were applied to assess concordance.
Results: Among 100 samples, the blood film method showed the highest agreement with culture smears (63 samples, κ = 0.26), followed by conventional (62, κ = 0.24), drop and rest (61, κ = 0.22), and water wash (59, κ = 0.18). Gram-positive cocci in pairs and Gram-negative bacilli were most accurately detected using the blood film method (10 and 17 samples, respectively). Heavy resin/charcoal deposits were most common with water wash (41 samples) and least with blood film (6 samples). The blood film method also produced the highest number of deposit-free samples (29), indicating superior clarity.
Conclusion: Among the four smear preparation methods studied across 100 positive blood culture samples, the blood film method demonstrated the highest diagnostic agreement and the least interference from resin and charcoal particles. It is recommended as the most effective and reliable technique for direct Gram staining in clinical microbiology laboratories.
Keywords: gram stain interpretation; blood film method; kappa (κ) statistics
Introduction
Bloodstream infections, particularly in high-risk individuals, are linked to considerable morbidity and mortality, underscoring the critical importance of early pathogen detection and prompt initiation of therapy to enhance patient outcomes. Among the initial diagnostic tools available in the microbiology laboratory, Gram staining of positively flagged blood culture bottles plays a pivotal role in the rapid preliminary identification of pathogens [1].
Gram staining provides valuable information regarding the morphology and Gram reaction of bacteria, allowing clinicians to initiate or modify empirical antibiotic regimens even before culture results are finalized [2]. Additionally, reliable Gram stain interpretation enables laboratories to perform direct antimicrobial susceptibility testing on blood culture broth, potentially reducing the diagnostic turnaround time by 24 hours [3]. Despite its clinical value, direct Gram stain interpretation from blood culture broth is challenging, and errors in interpretation have been linked to inappropriate therapy and poorer patient outcomes [4].
One of the main limitations is the presence of resin and charcoal particles in the blood culture media [5,6]. These additives are designed to neutralize residual antibiotics in the patient's blood, thereby enhancing microbial recovery. However, they also retain crystal violet stain, appearing Gram-positive and often obscuring or mimicking bacterial forms. This can lead to misinterpretation, especially in cases with low bacterial load or in laboratories with less experienced personnel. Additionally, technical factors such as smear thickness, uneven distribution of the sample, and improper fixation can further compromise the clarity and accuracy of the results [6].
To address these limitations, several smear preparation techniques have been described, including the conventional method, the water-wash method, the blood film method, and the drop-and-rest method [7]. While the conventional method is widely used due to its simplicity, it has been associated with high rates of artifact-related interference. In contrast, the blood film and drop-and-rest methods-recommended by automated blood culture system manufacturers-aim to reduce particulate artifacts by facilitating better separation of microbial elements from background debris [8].
Although multiple smear preparation techniques have been proposed to enhance the clarity and accuracy of Gram stain interpretation from blood culture broths, comparative data evaluating their relative diagnostic performance remain limited. Recognizing this gap, the current study was designed to systematically assess and compare four distinct smear preparation methods-namely, the conventional method, the water-wash method, the blood film method, and the drop-and-rest method—according to the standard references described below [9-12]. The evaluation focused on two key parameters: the level of agreement between each method's Gram stain findings and the final culture-based Gram stain results, and the extent of interference caused by resin and charcoal particles present in the culture medium.
Material and Methods
This prospective cohort study was conducted at the University College of Medical Sciences and Guru Teg Bahadur Hospital, Delhi, India, between January 2025 to May 2025. All positively flagged blood culture bottles from the BacT/ALERT automated system were included.
Upon flagging positive, the bottles were promptly removed from the instrument and transferred to a biological safety cabinet (BSC) for further processing. A sterile, unused syringe was inserted through the alcohol swab to aseptically aspirate 1-2 mL of broth. Subsequently, four different smear preparation techniques were employed: the conventional method, the blood film method, the drop-and-rest method, and the water-wash method. These were performed as per standard reference protocols, detailed in the subsequent section.
Conventional Method
A clean slide was prepared by placing a drop of the sample onto its surface and spreading it evenly to form a smear about 1 cm in diameter. The smear was then left to air dry, followed by heat fixation [9].
Water-Wash Method
For this technique, a drop of blood from the culture bottle was placed on a clean, labelled slide and allowed to dry. The slide was then gently washed with water, directing the flow first over the thumb and then allowing it to cascade freely across the smear. After the water wash, the smear was heat-fixed and stained.
Blood Film Method
This method closely resembles the preparation of a thin peripheral blood smear, following the manufacturer's instructions [9]. A small drop of the sample was placed at one end of a clean, labelled slide. A second clean slide, held at a 25° angle, was used as a spreader to touch the drop. The spreader slide was then pressed firmly against the first slide and quickly pushed forward to distribute the sample evenly. After the smear was prepared, it was air-dried, heat-fixed, and stained. The stained smear was examined in the region where the sample was most thinly and evenly spread.
Drop and Rest Method
In this technique, a drop of the sample from the blood culture bottle was placed on a clean, labelled slide and left undisturbed for about 10 minutes to allow the charcoal particles to settle [9]. After this settling period, the slide was gently tilted so that the clearer portion of the sample could flow away from the settled charcoal. The sample was then spread using an applicator stick, air-dried, and fixed either by heating at 60°C or with 95% methanol for 2 minutes. After staining, the smear was examined in the area where the sample had been spread, rather than at the site where the original drop was placed.
After preparing the smears using the aforementioned methods, all were subjected to Gram staining following the standard protocol [11]. As part of routine laboratory procedures, broth from blood culture bottles that had flagged positively was streaked onto common bacteriological media such as sheep blood agar and MacConkey agar. Gram staining was subsequently performed on smears prepared from colonies that had grown after overnight incubation at 37°C under aerobic conditions.
To ensure consistency in interpretation, the results of the Gram stain were categorized into five distinct groups: Gram-positive cocci, Gram-positive bacilli, Gram-negative cocci, Gram-negative bacilli, and mixed flora. Additionally, the presence of resin or charcoal particles within the smear was evaluated and classified according to the following scale: "Nil" indicated that no resin or charcoal was observed across 20 fields, "Minimal (1+)" was assigned when resin or charcoal appeared in 1 to 5 of the 20 fields, "Moderate (2+)" was used when they were observed in 6 to 10 fields, and "Heavy (3+)" was given when resin or charcoal was present in more than 10 of the 20 fields. This classification ensured a standardized and systematic approach to the interpretation of the Gram stain results [13].
The smear preparations and Gram staining were meticulously performed by the same skilled laboratory technician throughout the study, ensuring consistency. Three independent observers, each possessing a minimum of three years' expertise in Gram stain interpretation, meticulously examined the slides. To eliminate bias, the observers remained blinded to the culture smear results until the study's conclusion. The final interpretation was considered valid only when at least two of the three observers reached a unanimous agreement. Smears where no consensus was reached among all three observers were excluded from the analysis.
The study assessed the agreement between the Gram stain results from the blood culture broth and the Gram stain findings of smears prepared using four different methods. Additionally, the presence and quantity of charcoal and resin particles in the Gram-stained smears from each preparation method were compared. To evaluate the level of agreement between the Gram stain results of the blood culture broth and the culture smear, kappa statistics were applied for statistical analysis.
Results
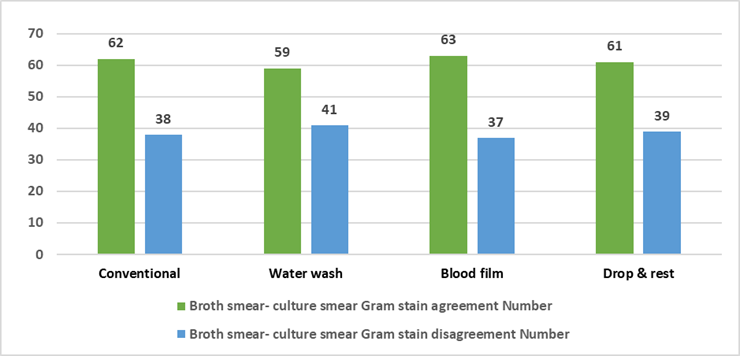
A total of 100 positively flagged automated blood culture bottles were processed to prepare smears using four distinct preparation techniques. The performance of each direct smear method was compared against the Gram stain results of the corresponding culture smear. Statistical analysis of the concordance was conducted using the kappa (κ) coefficient to evaluate the level of agreement. Among the four methods, the highest agreement was observed with the blood film method (κ = 0.26), followed by the conventional method (κ = 0.24), drop and rest (κ = 0.22), and water wash (κ = 0.18). Although all methods demonstrated only slight to fair agreement, the blood film technique showed a marginally better concordance with the reference standard [Table1] [Figure1].
Table 1: Evaluation of concordance between various direct smear preparation methods from positive blood culture broths and Gram stain results of the corresponding culture smears.
| Smear Preparation Techniques for Positive Blood Culture Broths | Broth Smear-Culture Smear | Kappa Value | |
| Gram Stain Agreement, n (%) | Gram Stain Disagreement, n (%) | ||
| Conventional | 62 | 38 | 0.24 |
| Water wash | 59 | 41 | 0.18 |
| Blood film | 63 | 37 | 0.26 |
| Drop and rest | 61 | 39 | 0.22 |
Figure 1: Comparison of agreement between different direct smear preparation methods from positive blood culture broth and the corresponding Gram stain results of culture smears.
As shown in Table 2 and Figure 2, Gram-positive cocci in clusters were most frequently identified using the conventional method (54/100, 54%), closely followed by the drop and rest method (55/100, 55%), blood film method (51/100, 51%), and water wash method (65/100, 65%). Gram-positive cocci in pairs were better detected by the blood film method (10/100, 10%) and water wash method (7/100, 7%), with lower detection rates observed in the drop and rest (4/100, 4%) and conventional methods (5/100, 5%). Gram-negative bacilli were most accurately identified by the blood film method (17/100, 17%), followed by the conventional (11/100, 11%), drop and rest (12/100, 12%), and water wash methods (9/100, 9%). The detection of anaerobic spore bearers (ASB) and budding yeast cells (BYC) remained consistent across all smear preparation techniques, each demonstrating uniform identification rates of 2% and 5%, respectively. Sterility was most frequently noted in smears prepared by the water wash method (14/100, 14%), followed by the conventional method (10/100, 10%), drop and rest (5/100, 5%), and blood film method (6/100, 6%).
Table 2: Comparative analysis of various methods based on final pathogen identification.
| Category | Culture Smear | Broth Gram Stain | |||
| Conventional, n (%) | Water wash, n (%) | Blood film, n (%) | Drop and Rest, n (%) | ||
| GPC in Cluster | 75 | 54 | 51 | 55 | 65 |
| GPC in Pair | 01 | 05 | 07 | 10 | 04 |
| GNB | 13 | 11 | 09 | 17 | 12 |
| ASB | 02 | 02 | 02 | 02 | 02 |
| BYC | 05 | 05 | 05 | 05 | 05 |
| Sterile | 04 | 10 | 14 | 06 | 05 |
| Total | 100 | 87 | 88 | 95 | 93 |
GPC: Gram positive cocci, GPB: Gram positive bacilli, GNB: Gram negative bacilli, BYC: Budding yeast cells
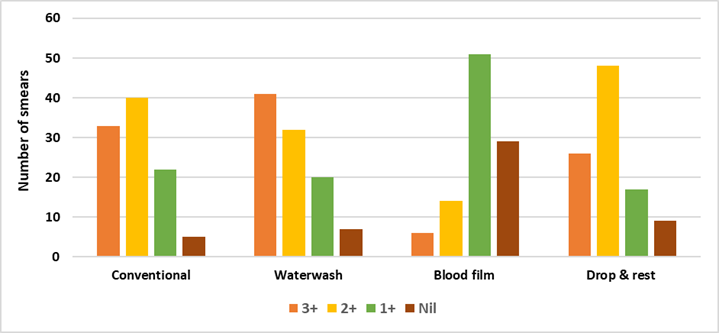
Figure 2: Grading of resin/charcoal among various four methods in the study group (n=100).
Table 3 presents the distribution of resin/charcoal grading (n = 100) in Gram-stained smears prepared by four different smear preparation techniques obtained from positive blood culture bottles. The presence of heavy resin/charcoal deposits (3+) was most frequently observed in smears prepared by the water wash (41%) and conventional (33%) methods, followed by the drop and rest (26%) method. The blood film method demonstrated the lowest frequency of 3+ deposits, with only 6% of smears showing heavy deposits. Moderate (2+) resin/charcoal deposition was most commonly observed with the drop and rest (48%) and conventional (40%) methods. In contrast, water wash and blood film methods showed a lower proportion of 2+ grading, accounting for 32% and 14% of smears, respectively. Mild resin/charcoal deposits (1+) were predominantly seen in the blood film method (51%), followed by drop and rest (17%), water wash (20%), and conventional methods (22%).
Table 3: Grading of resin/charcoal among various four methods in the study group (n=100).
| Methods of Smear Preparation from Positively Flagged Blood Culture Broth | Grading of Resins/Charcoals (n=100) | |||
| 3+ | 2+ | 1+ | Nil | |
| Conventional | 33 | 40 | 22 | 5 |
| Water Wash | 41 | 32 | 20 | 7 |
| Blood Film | 6 | 14 | 51 | 29 |
| Drop & Rest | 26 | 48 | 17 | 9 |
While the blood film and drop-and-rest techniques offered marginally improved detection of Gram-positive cocci in pairs and Gram-negative bacilli, the water-wash method was associated with the greatest frequency of sterile smear results. These results highlight differences in diagnostic efficacy across smear preparation techniques, emphasizing the importance of method selection for optimal outcomes. Additionally, resin/charcoal-free smears were most common with the blood film method (29%), followed by drop and rest (9%), with conventional (5%) and water wash (7%) methods showing minimal absence of deposits [Figure 3].
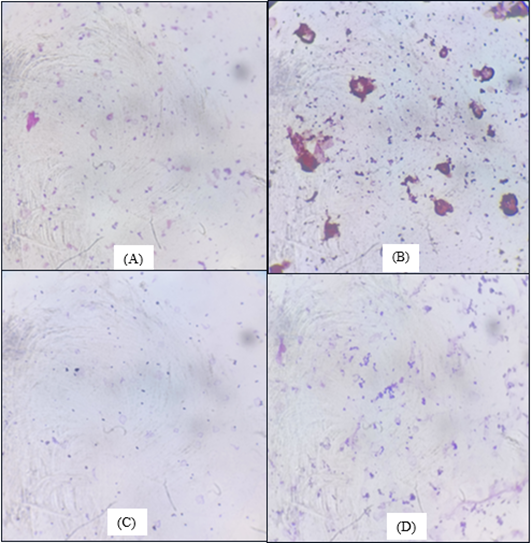
Figure 3: Gram Stain Morphology Demonstrated by Four Different Smear Preparation Methods (A) Drop & rest method (B) Conventional method (C) Blood film method (D) Water wash method.
Discussion
Bloodstream infections (BSIs) carry a high burden of illness and death, with overall mortality rates surpassing 20%. Timely administration of suitable empirical antimicrobial therapy is essential for enhancing patient outcomes. Nevertheless, as many as 40% of BSI cases may initially receive inadequate treatment prior to the first blood culture result being reported. Although automated blood culture systems have improved detection, delays in final culture and susceptibility results often hinder timely therapy. Therefore, the microbiology laboratory must prioritize rapid, stepwise reporting-including direct Gram stain, preliminary identification, and direct susceptibility testing-to guide timely clinical decisions. Among these, Gram staining serves as a rapid and reliable initial diagnostic tool, facilitating early presumptive identification and optimization of empirical therapy [14,15].
The Gram stain is widely regarded as the first and most critical diagnostic step following a positive blood culture, enabling early presumptive identification of pathogens and guiding timely empirical antimicrobial therapy. However, its clinical significance is largely determined by the quality of smear preparation, which plays a crucial role in ensuring accurate interpretation. The American Society for Microbiology underscores the importance of Gram staining in initial reporting and recommends the use of clear, descriptive terminology. As such, Gram stain findings typically form the basis of the first clinical notification, influencing early treatment decisions and potentially improving patient outcomes [14].
A major challenge in Gram staining from blood culture broths is the interference caused by resin or charcoal particles, which obscure microscopy and hinder interpretation. Suboptimal smear preparation, particularly with the conventional method, further contributes to misidentification or missed detection of organisms. Research has indicated that the conventional smear method is linked to a higher rate of inaccurate results[3].
This study was undertaken to evaluate and compare four smear preparation methods-conventional, water wash, drop and rest, and blood film-in terms of resin/charcoal interference. The results demonstrate that the blood film method had the lowest incidence of heavy background deposits, thereby offering superior visual clarity. On the other hand, the water wash and conventional methods showed a higher percentage of smears with heavy (3+) resin/charcoal deposits, potentially hindering accurate interpretation. The drop and rest method showed intermediate results, balancing ease of use and moderate reduction in artifacts.
This study found the blood film method had the highest agreement with culture smears (κ = 0.26) and the best detection of Gram-positive cocci in pairs (10%) and Gram-negative bacilli (17%). The drop and rest method also performed well with fewer false positives. The conventional method showed moderate agreement (κ = 0.24) but more false positives, likely due to resin and charcoal interference. The water wash method had the lowest agreement (κ = 0.18), was time-consuming, and had the most resin/charcoal deposits. The blood film method had the least interference (29% smears deposit-free), supporting its use as the preferred smear preparation technique for accurate and reliable Gram staining from positive blood cultures.
A study conducted by Søgaard et al. [13] reported high sensitivity of direct Gram staining from blood culture broth across various organisms, likely due to technical training, though smear preparation methods were unspecified. Similarly, Nain et al. found the blood film method showed the highest agreement with culture Gram stain (κ = 0.637) among four techniques, with the least resin/charcoal interference (5.2%), while the conventional method had the most interference (33.8%), leading to more inaccurate results [15].
These findings highlight the need for clinical microbiology laboratories to reassess their smear preparation techniques. The blood film method, as demonstrated by Nain et al. [7] offers superior clarity and minimal background interference, thereby enhancing the reliability of Gram stain results, facilitating early pathogen identification, and supporting timely, appropriate antibiotic therapy. It is thus recommended for optimal diagnostic performance.
Conclusion
This study evaluated four smear preparation methods-conventional, water wash, blood film, and drop and rest-for Gram staining of positively flagged blood culture broths. The blood film method demonstrated the highest agreement with culture-based Gram stain results (κ = 0.26), followed by the conventional (κ = 0.24), drop and rest (κ = 0.22), and water wash (κ = 0.18) methods. Notably, the blood film method exhibited the least interference from resin and charcoal particles, with 29% of smears showing no such deposits, compared to 5% for the conventional, 7% for the water wash, and 9% for the drop and rest methods. These findings underscore the importance of selecting an appropriate smear preparation technique to enhance the accuracy of direct Gram stain interpretation from blood culture broths. The blood film method, with its superior clarity and minimal background interference, is recommended for optimal diagnostic performance in clinical microbiology laboratories.
References
- Samuel LP, Balada-Llasat JM, Harrington A, Cavagnolo R. (2016). Multicenter Assessment of Gram Stain Error Rates. J Clin Microbiol. 54(6):1442-1447.
Publisher | Google Scholor - Rand KH, Tillan M. (2006). Errors in Interpretation of Gram Stains from Positive Blood Cultures. Am J Clin Pathol. 126(5):686-690.
Publisher | Google Scholor - Behera B, Mathur P, Gupta B, et al. (2010). Blood Culture Gram Stain, Acridine Orange Stain and Direct Sensitivity-Based Antimicrobial Therapy of Bloodstream Infection in Patients with Trauma. Indian J Med Microbiol.28(2):138-142.
Publisher | Google Scholor - Adler H, Baumlin N, Frei R. (2003). Evaluation of Acridine Orange Staining as a Replacement of Subcultures for BacT/ALERT-Positive, Gram Stain-Negative Blood Cultures. J Clin Microbiol. 41(11):5238-5239.
Publisher | Google Scholor - Fiori B, D’Inzeo T, Di Florio V, et al. (2014). Performance of Two Resin Containing Blood Culture Media in Detection of Bloodstream Infections and In Direct Matrix Assisted Laser Desorption Ionization Time of Flight Mass Spectrometry (MALDI TOF MS) Broth Assays for Isolate Identification: Clinical Comparison of The BacT/Alert Plus and Bactec Plus Systems. J Clin Microbiol. 52:3558-3567.
Publisher | Google Scholor - Kristóf K, Pongrácz J. (2016). Interpretation of Blood Microbiology Results - Function of the Clinical Microbiologist. EJIFCC. 27(2):147-155.
Publisher | Google Scholor - Nain J, Deepashree R, Tamang P, et al. (2018). Comparison of Four Different Methods of Smear Preparation for Gram Staining of Positively Flagged Automated Blood Culture Bottles. J Curr Res Sci Med. 4:98-103.
Publisher | Google Scholor - BACT/ALERT®FAN®PLUS Gram Stain; 2018.
Publisher | Google Scholor - Tille P. (2013). Bailey & Scott’s Diagnostic Microbiology. 13rd ed. St. Louis, Missouri: Elsevier Health US.
Publisher | Google Scholor - Mackie TJ, McCartney JE. (1996). Mackie & McCartney Practical Medical Microbiology. 14th ed. New York: Churchill Livingstone.
Publisher | Google Scholor - BMS Diagnostics. (2013). BD BACTEC™ FX: State-of-the-art blood culture system. Selangor, Malaysia: BMS Diagnostics.
Publisher | Google Scholor - Winn WC, Koneman EW. (2006). Koneman’s Color Atlas and Textbook of Diagnostic Microbiology. 6th ed. Philadelphia: Lippincott Williams & Wilkins.
Publisher | Google Scholor - Chandrasekaran, S., Abbott, A., Campeau, S., Zimmer, B. L., Weinstein, M., et al. (2018). Direct-From-Blood-Culture Disk Diffusion to Determine Antimicrobial Susceptibility of Gram-Negative Bacteria: Preliminary Report from The Clinical and Laboratory Standards Institute Methods Development and Standardization Working Group. Journal of Clinical Microbiology, 56(3):10-1128.
Publisher | Google Scholor - Uehara Y, Yagoshi M, Tanimichi Y, et al. (2009). Impact of Reporting Gram Stain Results from Blood Culture Bottles on The Selection of Antimicrobial Agents. Am J Clin Pathol. 132:18-25.
Publisher | Google Scholor - Søgaard M, Nørgaard M, Schønheyder HC. (2007). First Notification of Positive Blood Cultures and The High Accuracy of The Gram Stain Report. J Clin Microbiol. 45(4):1113-1117.
Publisher | Google Scholor