Re-Print Article
Immature Platelet Fraction as A Useful Predictor of The Aetiology of Thrombocytopenia: Experience from Oman
- Shaikha M. Al Ghafri *
Department of Haematology, Armed Forces Hospital, Muscat, Oman.
*Corresponding Author: Shaikha M. Al Ghafri, Department of Haematology, Armed Forces Hospital, Muscat, Oman.
Citation: Shaikha M. Ghafri. (2025). Immature Platelet Fraction as A Useful Predictor of The Aetiology of Thrombocytopenia: Experience from Oman, Clinical Case Reports and Studies, BioRes Scientia Publishers. 11(1):1-6. DOI: 10.59657/2837-2565.brs.25.283
Copyright: © 2025 Shaikha M. Al Ghafri, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: October 10, 2025 | Accepted: October 24, 2025 | Published: November 12, 2025
Abstract
Clinical evaluation of the possible aetiology of thrombocytopenia is important in the management of thrombocytopenia, which is concomitant with different disease processes. Thrombocytopenia can lead to a range of symptoms varying from petechie and purpura to excessive bleeding from the mucocutaneous membrane, especially when severe. The aetiology of thrombocytopenia is usually not obvious, and different investigations are needed, including invasive methods such as bone marrow examination. The immature platelet fraction (IPF%, the ratio of immature platelets to the total number of platelets in a patient's blood) has emerged as a useful, easy, fast parameter for assessing thrombopoiesis in patients with thrombocytopenia. This assessment helps clinicians identify the underlying cause of thrombocytopenia, namely, increased peripheral platelet destruction or underproduction, and thus can obviate the need for bone marrow examination. The primary objective of this study was to evaluate the usefulness of IPF in directing the end user towards the possible aetiology of thrombocytopenia in the Omani population. A secondary objective of the study was to determine the possible correlation between platelet count and IPF in patients with COVID-19 as an example of consumptive thrombocytopenia.
IPF% was evaluated in 136 patients managed at the Armed Forces Hospital between April and September 2021 who were diagnosed with thrombocytopenia as a result of different diseases.
Data from healthy people and patients were collected from the Sysmex XN blood cell counter database (IPF special channel) and analysed using Minitab 17 Software. The patients with thrombocytopenia included patients with COVID-19 (n=61), patients with infection/sepsis (n=12), gestational thrombocytopenia (n=16), systemic lupus erythematosus (n=8), microangiopathic haemolytic anaemia (n=2), sickle cell disease (n=11), or liver cirrhosis (n=24) and post chemotherapy patients (n=2). Compared to the 55 healthy control group (mean = 4.28), the most significant rise in IPF% was observed in cases of increased peripheral platelet destruction/consumption, such as thrombocytopenia in COVID-19 (mean= 16.23), infection/sepsis (mean=12.98), gestational thrombocytopenia (mean= 17.62), systemic lupus erythematosus (mean= 18.26) and microangiopathic haemolytic anaemia (mean= 10.60). Additionally, the mean IPF% was higher in patients with sickle cell disease and liver cirrhosis (7.44 and 5.45, respectively) than in patients in the control group but remained within the newly determined reference range of IPF% for the Omani population (0.2-8.36). The mean IPF% post chemotherapy (mean= 4.70) was the closest to the control group mean.
This is the first study to examine the utility of IPF% in identifying the aetiology of thrombocytopenia in an Omani population, and based on these data, we can conclude that IPF can be used as a first-line screening parameter that may add corroborative evidence for the differential diagnosis of the aetiology of thrombocytopenia.
This study also reported a reference interval for IPF% in the Omani population (0.2-8.36) for the first time, independent of gender. Moreover, by studying the relationship between the IPF% and PLT count in COVID-19 patients, it was found that the IPF% increased with the decrease in the number of platelets. This finding suggests that IPF may have a role as a prognostic marker in these patients and may lead clinicians to suspect the development of complications.
Keywords: immature platelet fraction; thrombocytopenia; peripheral destruction; platelet underproduction
Introduction
Thrombocytopenia is generally defined as a decrease in the platelet count to less than 150 x 109/l with (100 to 150) x 109/l considered mild thrombocytopenia, (50 to 100) x 109/l labelled as moderate thrombocytopenia, and less than 50 x 109/l defined as severe thrombocytopenia [1, 2]. Thrombocytopenia is not a disease by itself but a common haematologic finding that is caused by different disease processes. Thrombocytopenia can be mild and lead to symptoms such as purpura, petechiae and nosebleeds. However, in some patients with serious bleeding, it is considered one of the life-threatening illnesses that require urgent haematologic consultation [2]. Hence, early diagnosis and treatment are crucial to avoid serious complications. Bone marrow aspiration remains the gold standard for assessing the cause of thrombocytopenia, but it is an invasive and expensive test [3, 4]. The possible causes of thrombocytopenia can be divided into two main categories: decreased platelet production (e.g., aplastic anaemia, myelodysplastic syndrome, acute leukaemia, and chemotherapy-induced thrombocytopenia) and increased peripheral platelet consumption (e.g., immune thrombocytopenia, disseminated intravascular coagulopathy, thrombotic thrombocytopenic purpura or other microangiopathic haemolytic diseases (MAHA) and certain infections) [5].
Thus, determination of the aetiology of thrombocytopenia can be crucial for the early recognition of the category of thrombocytopenia and hence implementation of appropriate management, which may include platelet transfusion and bone marrow examination [6].
The immature platelet fraction (IPF) has recently been shown to be useful in quantifying reticulated immature platelets (newly released platelets from the bone marrow by megakaryocytes). The detection of reticulated immature platelets in the circulation (life span less than 24 hours) may help in discriminating the cause of thrombocytopenia (hyperdestructive/consumption from hypo-productive), as the number of immature platelets is proportional to the rate of thrombopoiesis [7-11]. In general, the IPF % value increases faster than the increase in PLT counts by several days [7].
Newly released platelets are usually larger in size and contain more cytoplasmic RNA content that allows them to be differentiated from mature platelets using a fluorescence staining method on Sysmex XN analysers [12]. One study showed that IPF can be used as a potential useful rapid parameter for assessing thrombopoietic activity [13], and other studies have suggested that IPF may help to identify the possible cause of thrombocytopenia [6, 8, 14].
This study was conducted during the outbreak of COVID-19 infections. Thrombocytopenia was found to be a common finding in this infection. Thrombocytopenia (platelet count of less than 100 x 109/l) appears to be associated with an increased risk of thrombo-inflammation, as it indicates the presence or development of coagulopathy [15-18]. The immature forms of platelets are hyperactive and demonstrate an enhanced thrombotic tendency. [19, 20]. Thus, IPF, as a platelet activation parameter, can play a role in predicting thrombotic events and disease severity in COVID-19 patients [17, 20-22].
The primary objective of this study was to evaluate the usefulness of IPF in directing the end user towards the possible aetiology of thrombocytopenia in the Omani population. In this study, the IPF values of 137 Omani patients diagnosed with thrombocytopenia with a known cause and who attended the Armed Forces Hospital between April and September 2021 were compared to those of a healthy control group. The secondary objective was to predict the correlation between the platelet count and IPF value in COVID-19 patients.
Materials and Methods
Study design
This was a retrospective study of the usefulness of IPF% in patients with thrombocytopenia. It was based on analysing the IFP% of patients with different aetiologies of thrombocytopenia to determine whether the diseases can be identified based on IFP%. The study was conducted during the COVID-19 pandemic, so most of the studied thrombocytopenic cases were of patients diagnosed as COVID positive. In this study, the relationship between the platelet count and IPF% in patients with COVID-19 was tested.
The study was conducted at The Armed Forces Hospital, Oman over a period of six months, from April to September 2021. It was approved by the Armed Forces Medical Services Research Committee.
Patients and control group
A total of 55 healthy volunteers (30 males, 25 females) who attended the blood bank at AFH or blood donation campaigns to donate blood were enrolled as a control group after providing consent for participation. All these volunteers were adults with normal platelet counts and were used as a control group.
A total of 136 patients who presented with thrombocytopenia (platelet count of less than 130 x 109/l) from different clinical causes were included in the study. These patients were adults, over 18 years old and had no recent blood transfusions. These patients were divided into seven groups based on the possible cause of thrombocytopenia. These included 61 patients with COVID-19, 12 patients with infection/sepsis, gestational thrombocytopenia (n=16), systemic lupus erythematosus (n=8), microangiopathic haemolytic anaemia (n=2), sickle cell disease (n=11), or liver cirrhosis (n=24) and post chemotherapy patients (n=2).
IPF measurement
EDTA anticoagulated whole-blood samples were collected from the control group and the patients with thrombocytopenia, and each sample was sent to our laboratory for a full blood count and IPF measurement. The measurements were performed on a Sysmex XN3100 blood count analyser according to the manufacturer’s recommendation. The IPF % was measured automatically using a specific fluorescent channel (PLT-F). Here is a brief description of the principle of fluorescence flow cytometry measurement in PLT-F: after perforating the membranes of the platelets by the lysing reagent (Cellpack DFL), oxazine fluorescent dye (in Fluorocell PLT reagent) is used to label the RNA inside the platelets, avoiding interference with other cells or fragments of similar size. Using forward scattered light and the fluorescence signal, the platelets are separated from red blood cells and white blood cells.
Immature platelets are separated from mature platelets since they are more reactive and contain more RNA than mature platelets, which is reflected by an increased fluorescence signal. The fraction of immature platelets relative to mature platelets (IPF %) was measured.
Statistical analysis
Statistical analyses were performed using Minitab 17 Software. Statistical tests were selected according to the results of the normality test. Normally distributed data are expressed as the mean ± SD, while nonnormally distributed data are presented as the median and interquartile ranges. Nonparametric tests were used to compare the nonnormally distributed data from small sample sizes (<30> between each thrombocytopaenia group and the control group. Values of P less than 0.05 were considered to indicate statistical significance. The Spearman correlation test was used to test the correlation between the nonnormally distributed IPF% of COVID-19 patients and the platelet count (normally distributed) of these patients.
Results
Patients and participants
Approximately 60 volunteers were screened, of whom 55 eligible healthy participants were selected. Blood samples were obtained, and the platelet counts were within the reference range. After the outliers were excluded, the median IPF was 3.80%, and the mean IPF was 4.28% (95% limits, 0.2-8.36). There was no significant difference in IPF percentage between the males and females in this group (P= 0.404).
In the patient group, a total of 136 patient samples were included and divided into groups according to the disease diagnosis, and the mean IPF% was obtained for each group (Table 1), while the median IPF% (16.23, interquartile 11.75-20.30) was detected for the COVID-19 group. The groups represented patients with thrombocytopenia presumed due to increased peripheral platelet destruction and patients with thrombocytopenia due to underproduction. In this study, 12 patients were diagnosed with sepsis/infection, 16 patients with gestational thrombocytopenia, 8 with systemic lupus erythematosus (SLE), 61 with COVID-19, 2 with MAHA, 11 with sickle cell disease and 24 with liver cirrhosis. There were 2 patients undergoing chemotherapy.
Table 1: Mean IPF% of the study cohort (n= 136)
| Infection/Sepsis | Gestational | SLE | MAHA | SCD | Liver Cirrhosis | Post Chemotherapy | |
| Mean of IPF% ± SD | 12.98±3.36 | 17.62±4.68 | 18.26±9.56 | 10.60±4.36 | 7.44±3.172 | 5.446±2.12 | 4.70±0.85 |
Conclusion of analysis
The IPF was significantly higher in patients with thrombocytopenia due to PLT consumptive causes, such as COVID-19, infection/sepsis, gestational thrombocytopenia, SLE and MAHA, than in patients in the control group (4.28 ± 2.08). Patients with sickle cell disease and patients with liver cirrhosis also had higher IPF means (7.44 and 5.45, respectively) than the healthy control group, but the values were within the reference range (0.2-8.36).
Patients with SLE had the highest IPF% mean (18.26± 9.56) of all the patient groups studied. A lower IPF % mean (4.70 ± 0.849) was recorded in the patients in the postchemotherapy group. The highest IPF, 27.5%, was from a pregnant patient with gestational thrombocytopenia.
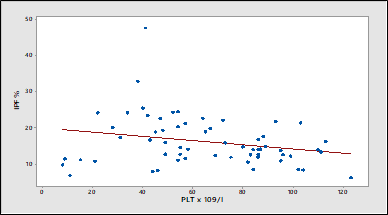
There was an inverse correlation between PLT count and IPF% in COVID-19 patients (Figure. 1).
Figure 1: Inverse correlation between PLT count and IPF% in COVID-19 patients as analysed by the Spearman correlation test
Discussion
In this study, IPF% was found to be higher in hyperdestructive/consumptive thrombocytopenia patients, including COVID-19 patients, patients with infection/sepsis, patients with gestational thrombocytopenia, SLE patients, and patients with MAHA, than in healthy people. These results are consistent with those obtained in several other studies [12-14, 23] reporting that IPF can be used to discriminate the cause of thrombocytopenia and avoid the need for invasive bone marrow examination in diagnosing peripheral hyperdestructive/consumptive thrombocytopenia.
Patients with liver disease and patients with sickle cell disease had IPF results that varied between high (compared to the healthy control group) and normal. The high results could be due to the pooling and sequestration of platelets in the enlarged spleen in some of these patients. On the other hand, thrombocytopenia with normal IPF in patients with liver disease could be explained by impaired thrombopoietin (TPO) production (predominantly produced by the liver) and hence reduced thrombopoiesis in the bone marrow [24].
In our study of COVID-19 patients, the results are in line with a previous report [25] where COVID-19-positive patients showed increased IPF compared to COVID-19-negative and reference-negative populations. This supports the recommendation of using IPF % to assist in the assessment of COVID-19 patients [16]. Although we could not assess the association between IPF and COVID-19 disease severity, by examining the relationship between the IPF% and PLT count in COVID-19 patients, we found that the IPF% increased with a decreasing number of platelets. This finding suggests that IPF may have a role as a prognostic marker in these patients and may lead clinicians to suspect the development of complications.
This study was, however, limited by the small number of patients with hypo-productive thrombocytopenia. A larger cohort of patients is needed to confirm the utility of IPF as a discriminatory parameter between hyperdestructive/consumptive thrombocytopenia and hypo-productive thrombocytopenia. A more comprehensive, multicentre study at the national level is recommended to obtain a reference range for different ages and to validate IPF as a crucial early diagnostic marker for thrombocytopenia.
The present study highlights the need to establish a reliable reference interval for IPF% in the Omani population to promote the clinical utility of this useful parameter in the diagnosis of thrombocytopenia. This reference interval of IPF% for the Omani population does not need to be sex specific, as there was no significant difference in IPF
Conclusion
In conclusion, and to our knowledge, this study is the first to examine the usefulness of IPF parameters in Oman and in GCC countries. It also reports a sex-independent reference interval for IPF in the Omani population (0.2-8.36) for the first time. The study also confirms IPF as an interesting tool for identifying the cause of thrombocytopenia.
Abbreviations
IPF: immature platelet fraction;
SLE: systemic lupus erythematosus;
MAHA: microangiopathic haemolytic anaemia;
SCD: sickle cell disease;
References
- Moore AH (2019). Thrombocytopenia in cirrhosis: a review of pathophysiology and management options. Clin Liver Dis (Hoboken), 14:183-186.
Publisher | Google Scholor - Elsayed MZ, Hamed L, Salama EH, Mohammed HS (2021). Can platelet count alone predicts bleeding in patients with thrombocytopenia? Sohag Med J, 25:73-75.
Publisher | Google Scholor - Asghar MB, Akhtar F, Mahmood A, Rafique N, Rana NA, Khalid UB (2023). Diagnostic accuracy of immature platelet fraction (IPF) to differentiate between thrombocytopenia due to peripheral destruction versus bone marrow failure. J Coll Physicians Surg Pak, 33:760-764.
Publisher | Google Scholor - Saran K, Vidya K, Seema K, Prasad A, Prakash J (2022). Study of platelet indices and their role in evaluation of thrombocytopenia. J Family Med Prim Care, 11:6236-6242.
Publisher | Google Scholor - Norrasethada L, Khumpoo W, Rattarittamrong E, Rattanathammethee T, Chai-Adisaksopha C, Tantiworawit A (2019). The use of mean platelet volume for distinguishing the causes of thrombocytopenia in adult patients. Hematol Rep, 11:7732.
Publisher | Google Scholor - Jeon K, Kim M, Lee J, Lee JS, Kim HS, Kang HJ, Lee YK (2020). Immature platelet fraction: a useful marker for identifying the cause of thrombocytopenia and predicting platelet recovery. Medicine (Baltimore). 99:e19096.
Publisher | Google Scholor - Imperiali CE, Arbiol-Roca A, Sanchez-Navarro L, Dastis-Arias M, Lopez-Delgado JC, et al. (2018). Reference interval for immature platelet fraction on Sysmex XN haematology analyser in adult population. Biochem Med (Zagreb), 28:010708.
Publisher | Google Scholor - Cohen A, Harari E, Cipok M, Laish-Farkash A, Bryk G, et al. (2021). Immature platelets in patients hospitalized with COVID-19. J Thromb Thrombolysis, 51:608-616.
Publisher | Google Scholor - Larsen JB, Pasalic L, Hvas AM (2020). Platelets in coronavirus disease 2019. Semin Thromb Hemost, 46:823-825.
Publisher | Google Scholor - Ashraf B, Jeon-slaughter H, Webb C, Lee NCJ, Chen W, Choi SH, Bat T, Ibrahim IF (2021). Biomarkers for thrombosis in COVID-19: a role for high sensitivity troponin-i and immature platelet fraction? Blood, 138:4260.
Publisher | Google Scholor - Welder D, Jeon-Slaughter H, Ashraf B, Choi SH, Chen W, Ibrahim I, Bat T (2021). Immature platelets as a biomarker for disease severity and mortality in COVID-19 patients. Br J Haematol, 194:530-536.
Publisher | Google Scholor - Van De Wyngaert Z, Fournier E, Bera E, Carrette M, Soenen V, Gauthier J, Preudhomme C, Boyer T (2020). Immature platelet fraction (IPF): a reliable tool to predict peripheral thrombocytopenia. Curr Res Transl Med, 68:37-42.
Publisher | Google Scholor - Ali I, Graham C, Dempsey-Hibbert NC (2019). Immature platelet fraction as a useful marker in the etiological determination of thrombocytopenia. Exp Hematol, 78:56-61.
Publisher | Google Scholor - Briggs C, Kunka S, Hart D, Oguni S, Machin SJ (2004). Assessment of an immature platelet fraction (IPF) in peripheral thrombocytopenia. Br J Haematol, 126:93-99.
Publisher | Google Scholor - Lee YK, Jeon K, Kim M et al (2018). Immature platelet fraction: a useful marker for identifying the cause of thrombocytopenia and predicting platelet recovery. Blood, 132:128.
Publisher | Google Scholor - Benlachgar N, Doghmi K, Masrar A, Mahtat EM, Harmouche H, Tazi Mezalek Z (2020). Immature platelets: a review of the available evidence. Thromb Res, 195:43-50.
Publisher | Google Scholor - Ali U, Knight G, Gibbs R, Tsitsikas DA (2017). Reference intervals for absolute and percentage immature platelet fraction using the Sysmex XN-10 automated haematology analyser in a UK population. Scand J Clin Lab Invest, 77:658-664.
Publisher | Google Scholor - Kariyawasan CC, Botenne CS, Ruhunuhewa US, Dissanayake DMC, Ranatunga SACD (2019). Immature Platelet Fraction (IPF) as a screening test to identify the cause for thrombocytopenia. Int J Sci Res Publ, 9:668-676.
Publisher | Google Scholor - Jose J, Magoon R, Makhija N (2021). Immature platelet fraction and COVID 19: maturing prognostic links. J Card Crit Care, 5:176-178.
Publisher | Google Scholor - Cohen A, Harari E, Yahud E, Cipok M, Bryk G, Lador NK, Mann T, Mayo A, Lev EI (2021). Immature platelets in patients with COVID-19: association with disease severity. J Thromb Thrombolysis, 52:708-714.
Publisher | Google Scholor - Maitta RW (2021). Immature platelets in COVID-19 infection. Br J Haematol, 194:494-495.
Publisher | Google Scholor - Incir S, Komesli Z, Baygul A et al (2021) Immature platelet fraction: is a novel early predictive marker for disease severity in patients with COVID-19 pneumonia? Turk J Biochem, 46:359-366.
Publisher | Google Scholor - Jeon MJ, Yu ES, Kang KW, Lee BH, Park Y, Lee SR, Sung HJ, Yoon SY, Choi CW, Kim BS, Kim DS (2020). Immature platelet fraction based diagnostic predictive scoring model for immune thrombocytopenia. Korean J Intern Med, 35:970-978.
Publisher | Google Scholor - Rauber P, Lammert F, Grotemeyer K, Appenrodt B (2018). Immature platelet fraction and thrombopoietin in patients with liver cirrhosis: a cohort study. PLoS One, 13:e0192271.
Publisher | Google Scholor - Introcaso G, Bonomi A, Salvini L, D'Errico T, Cattaneo A, Assanelli E, Barbieri SS, Biondi ML (2021). High immature platelet fraction with reduced platelet count on hospital admission. Can it be useful for COVID-19 diagnosis? Int J Lab Hematol. 43:1319-1324.
Publisher | Google Scholor