Review Article
Exploring New Horizons in Clinical Trials Bridging Science and Medicine
- Khushboo Verma
- Akash Yadav *
- Neelam Balekar
- Dinesh Kumar Jain
IPS Academy College of Pharmacy, Indore, Madhya Pradesh, India.
*Corresponding Author: Akash Yadav, IPS Academy College of Pharmacy, Indore, Madhya Pradesh, India.
Citation: Verma K., Yadav A., Balekar N., Dinesh K. Jain. (2025). Exploring New Horizons in Clinical Trials Bridging Science and Medicine, Journal of Clinical Research and Clinical Trials, BioRes Scientia Publishers. 4(5):1-11. DOI: 10.59657/2837-7184.brs.25.055
Copyright: © 2025 Akash Yadav, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: March 27, 2025 | Accepted: August 20, 2025 | Published: September 08, 2025
Abstract
Clinical trial design and interpretation are fundamental components of evidence-based medicine, serving as the basis for well-informed treatment plans and healthcare decisions. In order to reduce bias and guarantee the validity of study findings, this review article examines the fundamental components of clinical trial design, such as randomization, blinding, control groups, sample size calculation, and ethical issues. The essay also looks at how clinical trial results are interpreted, emphasizing the significance of statistical significance, clinical relevance, and the possibility of confounding variables. It also covers issues including the impact of protocol violations, generalizability, and incorporating trial results into clinical practice. By thoroughly examining both design and interpretation, this review seeks to offer insightful information to researchers, physicians, and policymakers in optimizing the quality and applicability of clinical trial results.
Keywords: clinical trial design; trials of adaptation; significance of clinical trials; data collection
Introduction
Trials are investigations that evaluate the efficacy and safety of drugs. Drugs, cells (and other biological products), surgery, medical technologies, behavioral treatments, radiological procedures, and preventative care actions are examples of such interventions. Trial results have the power to alter patient treatment and clinical procedures. Trials must be carefully planned, data must be gathered, and efficacy and safety must be evaluated to the highest standards in order to minimize patient harm.
The scientific basis for creating novel cures, treatments, and interventions is provided by the planning and analysis of clinical trials, which are the cornerstones of evidence-based medicine. A careful and deliberate approach to clinical trial design is crucial to guaranteeing the robustness and dependability of results as these studies grow more intricate and advanced. To address specific research issues while reducing biases and errors, every stage of a clinical trial must be meticulously planned, from establishing clear objectives and choosing suitable study designs to upholding ethical integrity and transparency [1].
A clinical research study's validity is assessed based on both its design and execution in addition to its findings. Activities related to clinical data management (CDM) set the stage for a trial's statistical analysis. Clinical research data must be properly collected, managed, accessed, and cleaned as part of CDM activity in a trial. Consequently, the quality of the data available for analysis determines the integrity of statistical analysis in a trial [2].
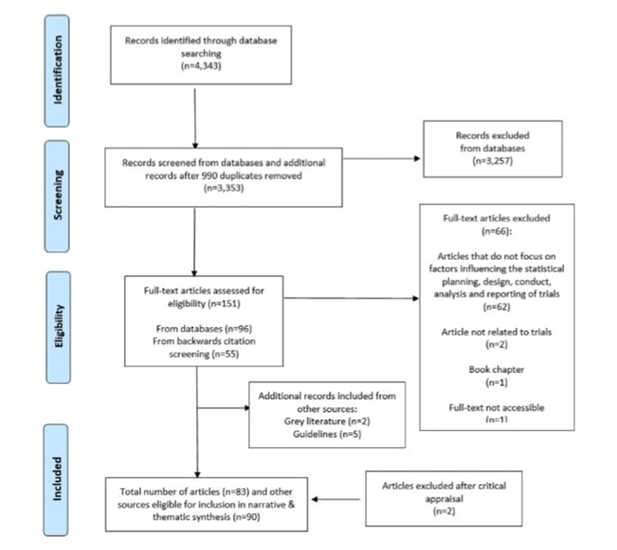
Figure 1: Prisma Flow Diagram.
Techniques
Randomized Controlled Trials: Participants are split into two groups at random: the treatment group and the control group. The goal is to remove bias and make sure that comparable variables don't affect the treatment's results. Blinding: persons don’t know which treatment they are receiving [3].
Types
Single-blind-Participants are unaware of their group assignment. Double-blind Neither the researchers nor the participants are aware of the participants' group assignment. Triple-blind the participants, researchers, and analysts are all unaware of their group affiliation.
Cohort Studies: Described as follows a group of individuals (a cohort) who have been exposed to or treated in a specific way over time and compares them to a cohort that has not. Often employed in observational studies, the goal is to compare the risk or benefit of the groups' outcomes.
Case Control Trials: The experimental treatment and the control treatment are administered to participants at separate times in cross-over trials. Frequently, the order is randomized. Reducing unpredictability and empowering each person to act as their own controller is the goal. The washout period is the time between two treatments to get rid of any side effects from the first one.
Placebo Controlled Trials: An inactive drug, or placebo, is employed as a control to compare the impact of treatment to no intervention. It helps in determining if the improvements seen are attributable to psychological variables such as the placebo effect or the actual treatment. For instance, research evaluating a novel painkiller would administer the drug to one group while giving a placebo to another.
Cross-Over Design: There is a washout interval in between treatments, and participants receive both the experimental therapy and a comparative treatment (such as a placebo) in a sequential fashion. By doing this, each participant can act as their own control, increasing statistical efficiency and lowering the required number of participants. A subject receives the medication initially, then a placebo after a washout time, and the results are recorded at each stage.
Multicenter Trials: These studies are carried out at several different locations. This broadens the pool of participants, enhances the results' generalizability, and speeds up the recruitment process. For instance, a study testing a global health intervention may encompass clinics across several nations or areas.
Longitudinal Design: In order to track the long-term effects of therapies, participants are monitored for a considerable amount of time. The goal of this technique is to investigate long-term effects or chronic illnesses, such as cardiovascular health or cancer remission. An example would be five-year research that tracks cancer patients to determine whether a new treatment regimen increases survival rates.
Adaptive Trials: Adaptive trials permit design changes in response to interim findings. If there is compelling evidence of efficacy or harm, this may include altering the trial's treatment arms, sample size, or even its termination early. By increasing clinical trials' adaptability and responsiveness to new data, it increases their effectiveness. Based on early encouraging results, a clinical trial may include a new treatment group [4].
Analysis of Survival: Time-to-event data, such as the duration till a disease recurs or a patient survives, can be analyzed using this method. It is especially helpful for evaluating results like progression-free survival and comprehending how a treatment affects a patient over time.
Examining the survival statistics of patients receiving a certain cancer treatment is one example.
Factorial Design: Several treatments or factors are examined concurrently by mixing them in various ways in factorial trials. In a single study, this enables researchers to examine the impact of multiple interventions and how they interact with one another. Examining the effects of a medication and a lifestyle change (like exercise) both separately and together [7].
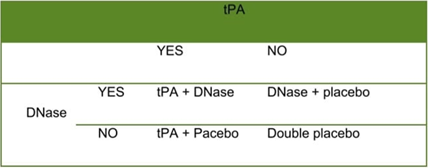
Figure 2: Factorial design comparing treatments for pleural infection.
Cluster Randomized Trials: Whole groups, or clusters, are randomized as opposed to individual participants. In public health trials or community-based initiatives, this is typical. It serves the purpose of being helpful in situations when individual randomization is impractical or unethical. It is also crucial in situations where therapies are aimed at populations rather than individuals. For instance, assigning schools at random to receive various educational interventions instead of specific students.
Equivalence Trials: The purpose of equivalency trials is to ascertain whether a new treatment is as effective as an existing one, within a predetermined margin. For example, a new medication for high blood pressure is compared to an established medication to demonstrate that both have similar effects [8].
End Point Selection: Endpoints are specific outcomes used to assess the effect of an intervention. Primary endpoints are the primary outcome that a trial aims to measure (e.g., overall survival, symptom relief). Secondary endpoints are additional outcomes that are not the primary focus but offer additional context. Appropriate endpoint selection guarantees that the clinical trial provides a satisfactory response to the research issue. For instance, overall survival may be the primary endpoint of a trial on a novel cancer treatment, while quality of life may be the secondary objective.
Methods of Statistics: To evaluate data and derive significant findings from clinical trials, a variety of statistical methods are used, including regression modeling, subgroup analysis, intention to treat analysis, and per protocol analysis. The goal of these techniques is to assist researchers in taking biases, missing data, and outcome variability into consideration. Regression analysis is used, for instance, to predict treatment results based on patient attributes such as age or genetic makeup [8].
Design of Clinical Trials
Different designs are employed based on the trial's setting and goal. Some trials have traits of the multiple designs, therefore the line between different design types is not always simple.
Cross Over Versus Parallel Designs
The majority of trials are carried out using parallel designs, usually comparing one treatment to the standard of care, a placebo, or no treatment at all. When evaluating different dosages of the medicine, for instance, parallel trials may involve more than one active arm. Only one therapy is given to each participant, and results are recorded and compared between groups.
Participants in a crossover design are assigned to one research arm at random for a predetermined amount of time, and then they are switched to the other study arm following a washout period. This makes it possible to measure each participant's results both during and after therapy, and to total the variations across all research participants. The ability to identify a significant treatment impact is increased by crossover designs, which remove baseline factor variance between the study arms [6].
Studies of Equivalency and Superiority vs non-Inferiority
Researchers usually try to show that a novel treatment is better than the standard of care or no treatment (placebo). When there is already a successful treatment for a problem, researchers typically try to demonstrate "non-inferiority," meaning that a novel treatment is comparable to the effective comparator, if not superior. If the new treatment has additional benefits over the comparison, such as fewer side effects or cheaper cost, then demonstrating non-inferiority rather than superiority may be sufficient [1].
Cluster Allocation and Group Randomization Experiments
Because it is the most statistically efficient, it is usually preferable to randomly assign individuals to different treatment groups on an individual basis. Randomization to alternative therapies may take place at the group level when it is not suitable or viable to do so. For instance, researchers could want to assess how a community's positive health behaviors are affected by a health promotion effort.
Groups can be randomly assigned and followed concurrently, or they can be forced to switch between therapies with a washout period in between. Groups may not always be able to crossover in both directions; for instance, a group assigned to a health promotion campaign may not be able to switch to no intervention because the exposure to the campaign cannot be reversed [6].
Factorial Design
Although some studies compare two therapies to a comparator, it might occasionally be interesting to assess the effects of the treatments collectively as well as each treatment separately. The effects of each treatment can be concurrently assessed against a comparator both alone and in combination thanks to a factorial design. DNase and intrapleural tissue plasminogen activator (t-PA) were assessed for the treatment of adult pleural infections in a classic factorial design scenario.
Useful Designs
When assessing a treatment's clinically relevant benefit under programmatic or real-world of the settings, pragmatic trials are employed; they often recruit a sizable number of participants from the entire target patient group based on broad eligibility criteria. To lessen the strain on the participants and increase participation and decrease dropout, follow-up is usually made as easy as a possible and data collecting needs are kept to a minimum, usually limiting each participant to one or two visits. If a drug's safety has already been established, such trials may also make it impossible to monitor adverse outcomes.
Clinical Trial Types
Observational Versus Interventional Research
Two basic forms of research designs utilized in clinical trials and epidemiological studies are interventional and observational investigations. Interpreting the findings of any clinical research requires an awareness of the main distinctions between them, which are how they respond to the exposure or intervention [9].
Figure 3: Types of Clinical Trial [4].
Interventional Research: When you have a clear hypothesis on cause and effect and wish to assess the direct effects of a particular treatment or intervention under controlled conditions, interventional studies are perfect. In clinical trials for novel drugs, therapies, or interventions, they are crucial.
When direct participant intervention is impractical, immoral, or impossible, observational studies are the most effective method (e.g., researching the effects of smoking on lung cancer in non-smokers). They are especially helpful for investigating uncommon illnesses, long-term consequences, or behaviors in authentic environments [10].
Randomized Controlled Experiments
In clinical research, a Randomized Controlled Trial (RCT) is regarded as the gold standard for assessing the efficacy of therapies. RCTs are used to assess the safety and effectiveness of a particular treatment or intervention in a controlled setting and to ascertain whether it produces the intended result [11]. An extensive examination of RCTs, including their design, benefits, and possible drawbacks, is provided below.
- An RCT is an experimental study design in which participants are randomized to either the control group or one or more treatment groups. An RCT's primary characteristic is the random assignment of participants to these groups, which helps to minimize bias by ensuring that the groups are similar at the beginning of the trial [12].
- Assessing the causal impact of an intervention (such as medication, treatment, or lifestyle modification) on particular outcomes is the main objective of a randomized controlled trial (RCT).
Randomized Controlled Trial Examples
The DCCT, which stands for Diabetes Control and Complications Trial Evaluation of the impact of strict glucose management on the onset and advancement of Type1diabetes complications is the major goal.
Research Design: Individuals with Type 1 diabetes were randomly assigned to one of two groups: intense therapy, which involved strict blood glucose control, or conventional therapy, which involved normal blood glucose management. The purpose of the study was to evaluate how strict glucose control affected long-term consequences such as neuropathy, nephropathy, and retinopathy.
Results: The likelihood of complications, such as renal and eye issues, was considerably decreased by strict glucose control. The standard of care for Type 1 diabetes was altered by this trial, which emphasized how crucial strict glycemic control is to avoiding complications.
Impact: The DCCT offered compelling proof that strict blood glucose control in Type 1 diabetes can postpone or avoid long-term consequences. It is a groundbreaking study in the field of diabetes care.[12]
The Action to Control Cardiovascular Risk in Diabetes (ACCORD) Study
To ascertain how severe blood pressure control, cholesterol-lowering medication, and glucose control affect cardiovascular events in people with Type 2 diabetes.
Research Design: The experiment had three arms: strict blood pressure management, aggressive lipid-lowering therapy (using statins), and intense glucose control (aiming for very low blood sugar). Individuals with high cardiovascular risk and Type 2 diabetes were randomized to either usual care or one of these therapies. Results are When compared to normal care, intensive glucose man. The Impact of the ACCORD study redirected clinical attention toward blood pressure and cholesterol management, challenging the commonly accepted notion that extremely strict glucose control would lower cardiovascular risk in Type 2 diabetes [13].
Non-Randomized Studies
Clinical studies that do not randomly allocate individuals to various intervention groups are known as non-randomized trials. Non-randomized trials use other techniques to assign treatments or interventions, even though randomized controlled trials (RCTs) are known for their randomization. When randomization is impracticable, unethical, or not possible, these studies are frequently employed [14]. Non-randomized trials have various drawbacks but can nevertheless offer insightful information about the efficacy of therapies despite not being randomized.
Non-Randomized Trial Types
Cohort Research: A cohort study compares the outcomes of a group of participants (referred to as a cohort) over time, comparing those who got a certain exposure or intervention to those who did not. Cohort study participants are not assigned to groups at random like they are in RCTs. For instance, smokers (the exposed group) and non-smokers (the unexposed group) might be compared over time in a study examining the long-term impact of smoking on lung cancer.
Advantages are Cohort studies are useful for long-term research on uncommon exposures or consequences. The Cons are they are susceptible to confounding and selection bias, where results could be influenced by other factors (such as lifestyle or genetics) [15].
Case-Control Research: Participants in case-control studies are chosen according to whether or not they experience a specific outcome (such as illness). After that, researchers evaluate their exposure to a particular risk factor or intervention by looking back. For instance, in a study examining the connection between childhood immunizations and autism, children with autism (cases) would be chosen, and their vaccination histories could be compared against those of children without autism (controls).
Advantages are Case-control studies are effective and practical for researching uncommon conditions or results. The cons are they are susceptible to selection bias (since the selection of cases and controls may not be representative of the general population) and recall bias (because they depend on participants' memories of prior exposures).
Pre-Post Studies (Before-After Studies): A before-after study contrasts the results obtained prior to and following the implementation of an intervention. When randomization is not feasible and the intervention is administered to every participant, this kind of study is frequently employed [16]. As an illustration, a hospital may start a new medication regimen to treat high blood pressure and compare the blood pressure measurements of the patients before and after the intervention.
Advantages are the method can provide preliminary information about the efficacy of an intervention and is rather straightforward. The Cons are there is no control group, it is challenging to credit any changes in results to the intervention alone because other factors may be at play.
Uncontrolled Trials, Or Single-Arm Trials: A single-arm study is defined as one in which no comparison group is used and all participants receive the same therapy. Early-stage clinical trials, such as Phase I or II trials, frequently employ this design. For instance, a study evaluating the impact of a novel chemotherapeutic medication on cancer patients may only include one patient group in order to ascertain the medication's safety and initial effectiveness.
Advantages: Single-arm trials are helpful for preliminary evaluations of novel therapies. Cons are there is a chance of bias because it is impossible to tell if the effects being seen are because of the treatment or something else without a control group.
Studies That Are Not Experiments
Definition: Studies that use exposures or interventions but do not use random assignment are known as quasi-experimental studies. Rather, individuals are grouped according to other factors (e.g., self-selection, geographical location). Example are one city (the intervention group) may implement a new anti-smoking program as part of a public health campaign to reduce smoking. then contrast it with a nearby city (the comparison group) that does not have the program.
Advantages: When randomization is impractical for practical, logistical, or ethical reasons, quasi-experimental designs are frequently employed. The Cons are the groups being compared may differ in significant ways other than the intervention, these studies may still be subject to bias and confounding [17].
Trials of Adaptation
Instead, then following a set protocol from the start, adaptive trials are a kind of clinical study that permits modifications to the trial design and treatment plans based on interim results. Because of this flexibility, researchers can make decisions in real time to maximize the trial's effectiveness, boost efficiency, and possibly hasten the creation of cures. Clinical research is using adaptive trials more and more, particularly in medication development to adapt to evolving conditions throughout the trial [16].
Adaptive Trial Examples
The Breast Cancer I-SPY 2 Trial: The purpose of this phase 2 adaptive trial is to assess several experimental medications in conjunction with conventional treatment for breast cancer. To determine whether a medicine will be beneficial, the study employs a Bayesian adaptive design, and treatments are modified in response to interim findings.
Adaptation: The trial adjusts as data comes in by eliminating treatments that don't work and concentrating on those that do. It has been effective in quickening the assessment of prospective treatments for breast cancer.
The COVID-19 Treatments NEJM Trial
Goal: Adaptive designs were used in several trials evaluating COVID-19 treatments (e.g., vaccines, anti-viral). In one adaptive trial, patients were randomized to receive one of several treatments; as new data became available, the study was set up to accommodate the addition of new therapies.
Adaptation: In light of new information, the study was changed to incorporate further interventions, such as the addition of monoclonal antibodies or the modification of treatment plans in response to patient outcomes.
The Cardiac Treatment TASTE Trial: The purpose of this experiment is to assess various approaches of revascularization, or the therapy of blood arteries, in cases with heart disease. The trial's design allowed for the possibility of altering treatment plans in response to preliminary results. Adaptation are the study was modified to place a greater emphasis on that treatment approach when preliminary findings indicated that one treatment was superior to another.[14]
The Function of Clinical Studies Pertaining to Nutrition
A strong body of evidence supports dietary recommendations, including excellent clinical trials, some of which have been created to demonstrate a causal link between dietary changes and lowering the risk of ASCVD. However, the difficulty of carrying out these studies has led to criticism of food and nutrition since the quality and strength of the evidence for certain medicinal therapies is not up to par. The purpose of this paper is to raise knowledge of the benefits of ASCVD demonstrated by nutrition-related clinical trials and the ways in which this data has influenced dietary recommendations.
Clinical studies pertaining to nutrition entail a number of special issues when compared to those involving pharmacological treatments. Numerous lines of data, such as those from epidemiological studies, randomized controlled trials, animal and in vitro research, and other studies, are used to support evidence-based dietary recommendations. The basic evidence for the significance of nutrition in ASCVD prevention, management, and treatment has been supplied by this research. A significant consensus has developed regarding the essential components based on the clinical trials that have been carried out. Because dietary exposures are multi-factorial, interdependent, and interconnected, finding a tested, repeatable intervention is a prerequisite for carrying out nutrition-focused clinical studies.
Thus, nutrition-focused Pharmaceutical clinical studies, in which the intervention group and the control group are only exposed to a known amount of a single medicine, are fundamentally different from clinical trials. Questions concerning the necessity and worth of nutrition interventions arise in light of the strong evidence that pharmaceutical agents, such as lipid/lipoproteins with statins4, blood pressure-lowering drugs, and GLP-1 receptor agonists, can significantly reduce ASCVD events and major CVD risk factors.
However, this ignores the advantages of maintaining a balanced diet for primary and fundamental prevention at the individual, social, and health system levels. eliminating other significant chronic illnesses, and raising life expectancy and quality of life Approximately one in five deaths in economically developed nations and a growing percentage in emerging cultures are attributed to atherosclerotic cardiovascular disease (ASCVD), which remains the world's leading cause of death. The mainstay of ASCVD prevention is adhering to a healthy dietary pattern. However, due to the numerous complexities, creating dietary recommendations based on excellent clinical trials that demonstrate a causal link between food and ASCVD risk is difficult. Related to planning and carrying out these investigations.
This paper aims to give a summary of the difficulties in carrying out clinical studies with a nutrition emphasis that support high evidence ratings during the guideline generation process and the consequences for dietary recommendations [4].
Chronic Pain Disorders That Affect the Lower Abdomen and Pelvis
This are common and frequently difficult to control, and may have a detrimental impact on sexual and psychological well-being as well as health-related quality of life. There are now approved therapies for certain illnesses from the European Medicines Agency (EMA) or the Food and Drug Administration (FDA). The methodological difficulties in creating randomized clinical trials (RCTs) to assess the effectiveness of new treatments in certain illnesses contribute to the lack of available medicines. First, lower abdominal discomfort frequently co-occurs with other symptoms (such as diarrhea, constipation, or frequent urination), which might impact the intensity of the pain.
Furthermore, pain may only occur during particular activities (such as sexual activity) and the illness may feature flare-ups of pain, which are periods of heightened symptom severity. It is difficult to create a single primary endpoint that has clinical significance for patients because of these features and receptive to therapy. While various outcomes can be evaluated in RCTs, in order to properly examine. To reduce false positive results in clinical trials, it is vital to determine a single primary end point or statistically correct for numerous primary analyses when evaluating the overall effect of a treatment in conditions with various symptoms.
Investigate is required to determine the elements of research design and techniques that can optimize the clinical significance and assay sensitivity of end points in these trials (e.g., qualitative patient interviews regarding the impact of various symptom patterns; secondary analyses that compare the effect sizes associated with various end points in existing trial data). Additionally, to enhance the dissemination and integration of results into clinical practice, more consistent reporting of design characteristics that are crucial for the interpretation of trial results and for meta-analyses of results across studies is necessary [13].
Clinical Studies of Treatments for Cirrhosis Using the Gut Microbiota
Research on the microbiome, especially hepatology, has exploded, with studies showing that liver disease is associated with changed microbial composition. More recently, attempts have been undertaken to comprehend the relationship between microbiome and characteristics with clinical results and to create treatments that target the microbiome.
Although microbiome therapies have a lot of potential, the planning and execution of clinical trials can be complicated by their special characteristics. The indications for microbiome therapy in cirrhosis, the various microbiome therapies, and the molecular pathways that these therapies target will all be briefly reviewed here. The best techniques and crucial factors for clinical trials of gut microbiota therapies in cirrhosis will then be our main emphasis. Considerations for the design of clinical trials for microbiome therapies in cirrhosis [15].
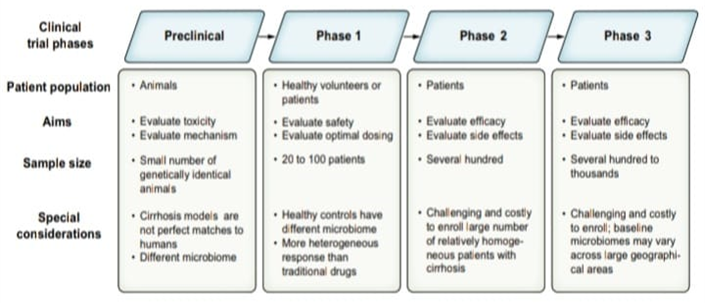
Phases of Clinical Trials
Most therapy testing begins with preclinical research in animal models (Fig. 6). Although there are numerous animal models available for the study of liver diseases, most preclinical research uses three animal models of cirrhosis in particular: carbon tetrachloride, thioacetamide, and common bile duct ligation. There are certain advantages to these models. For instance, carbon tetrachloride frequently causes cirrhosis and a number of liver dysfunction symptoms, such as ascites development, hepatocyte apoptosis, and other types of decompensations. Regretfully, there are a number of issues with these models. The elimination of carbon tetrachloride can ameliorate fibrosis, although the model varies from animal to animal and produces a heterogeneous effect. Unlike thioacetamide, always result in enough fibrosis to cause cirrhosis. By design, common bile duct ligation restricts biliary flow, making it more difficult to thoroughly assess the clinical impact of microbiome therapies [18].
Figure 4: Phases of clinical trials for microbiome therapeutics in cirrhosis.
Usually, several hundred patients with the illness are enrolled in phase II research. Determining side effects and efficacy are the goals. It takes a lot of money, careful planning, and multiple study locations to recruit a sizable and somewhat uniform cohort of cirrhosis patients. When it comes to microbiome therapy trials, the ability to assess colonization and clinical efficacy across patient subgroups with different cirrhosis aetiologies, indigenous bacterial compositions, and concurrent use of medications. For instance, it was discovered that baseline recipient.
Greater assessment of potential variable efficacy in subgroups will be feasible with larger phase II investigations. Hundreds to thousands of individuals with the disease are usually included in phase III research. It is difficult to enroll this many people with cirrhosis, necessitating extensive and often invariably global collaborations. Regional variations in diet and microbiota makeup lead to additional impact heterogeneity and a higher sample size needed to achieve sufficient statistical power. These larger trials enable additional examination of microbiome therapies in various patient subgroups and baseline enterotypes, despite the difficulty of enrolling a larger patient population [16].
End Points of Clinical Trials
The choice of endpoints is essential to any trial's design. The choice of endpoint affects both the trial's design and its findings. The regulatory results for such treatments are then established by these endpoints. Since the Liver Hope Consortium has just examined appropriate clinical trial endpoints in cirrhosis, this review will not be comprehensive. Hospitalizations, new or worsening decompensation, and survival (or transplant-free survival) are the main recognized outcomes. As previously said, the clinical trial phase determines the optimal outcomes.
Cirrhosis, particularly decompensated cirrhosis, has a negative impact on health-related quality of life. Quality of life is crucial for our patients, even while traditional main objectives like hospitalization or survival are obviously important. In cirrhosis, there are a number of well-established patient-reported outcomes. However, neither the FDA nor the EMA have yet to recognize these as established main endpoints. Pilot tests have been carried out recently. with the main outcomes being those reported by the patient, such as muscle soreness and health-related quality of life. Before being used as primary endpoints in studies, some patient-reported outcomes require additional validation and advocacy (to persuade regulatory agencies to accept them as clinical endpoints) [17].
Observation of Safety
There is a wealth of information linking the gut microbiota to almost every organ system in humans, almost twenty years after the National Institutes of Health Human Microbiome Project was started. It has been demonstrated that microbiome therapies affect a variety of illnesses, including neurological, immunological, oncologic, and gastroenterological ones. Consequently, microbiome therapies may have an effect outside of the liver and gastrointestinal system, whether positive or negative.
The majority of gastrointestinal side events in microbiome therapy trials for liver illness have been documented; however, this could be due to bias in the adverse event assessment procedure. Trials are not clearly recording weight, immunity or inflammatory incidents, changes in skin condition, mood swings, and a host of other potential negative outcomes. The site investigator is responsible for ascertain whether a symptom is an adverse occurrence and how linked it is.
Beyond gastrointestinal symptoms, adverse events should be rigorously monitored in future microbiome therapy trials in cirrhosis. Finally, to assess potential rare and related adverse events, data from several trials should be combined. Lastly, short follow-up periods one year at most have been a feature of microbiome therapy trials in cirrhosis to date [18].
We must have a deeper comprehension of the potential long-term consequences of microbiome alteration in the future. There is growing evidence that the composition and function of the gut microbiota differ in cirrhosis patients when compared to a number of control groups. Furthermore, it is evident that significant clinical outcomes in cirrhosis are influenced by immune response and gut metabolism. The field of hepatology has now in the age of therapeutically beneficial gut microbiota modification.
There are significant and particular factors to take into account while conducting clinical trials of microbiome therapies in cirrhosis patients, as this review has outlined. Realizing the full potential of microbiome therapies in this population will require navigating these subtleties, which is doable [19].
Conclusion
A key component of evidence-based medicine is the planning and analysis of clinical trials, which guarantees that the findings of studies are trustworthy and relevant to actual clinical situations. Important guidelines including blinding, randomization, and suitable control groups are essential for reducing bias and confounding variables and, eventually, improving the validity of the study's conclusions. How well the study can answer the research questions like therapy efficacy or safety is also determined by its design.
Clinical relevance, statistical significance, and the possibility of biases that could skew results must all be carefully taken into account when interpreting clinical study results. Evaluating the findings' consistency across different research groups and environments is also crucial. To guarantee their appropriate integration into clinical practice, clinical trial data should also be reported in a transparent manner and interpreted in light of current medical knowledge and public of the health requirements [20]. To sum up, improving patient outcomes and expanding medical knowledge depend heavily on the efficient design and interpretation of clinical trials. Clinical trials must be conducted with rigorous procedures, meticulous analysis, and open reporting in order to provide significant evidence for healthcare decision-making.
A multifaceted examination is necessary for the interpretation of clinical trial outcomes, taking into account both clinical relevance and statistical significance. Statistical techniques provide information about whether observed effects are more likely to be the result of the intervention than chance. However, the influence of data on patient care is ultimately determined by clinical importance. Even if a result is statistically significant, its practical use may be contingent on variables including patient demographics, treatment feasibility, and risk-benefit analysis [20].
Declarations
Acknowledgement
I would like to show my sincere gratitude towards IPS Academy College of Pharmacy for providing the necessary requirements and facilities throughout the study. Lastly, I would like to express my appreciation to all my colleagues and peers for their helpful discussions and moral support, which contributed to the success of this work.
Conflict of Interest
The authors declare no conflict of interest.
References
- Vogel, J. M., Pollack, B., Spier, E., McCorkell, L., Jaudon, T. W., et al. (2024). Designing and Optimizing Clinical Trials for Long COVID. Life Sciences, 122970.
Publisher | Google Scholor - Zaki, M., O'Sullivan, L., Devane, D., Segurado, R., McAuliffe, E. (2022). Factors Influencing the Statistical Planning, Design, Conduct, Analysis and Reporting of Trials in Health Care: A Systematic Review. Contemporary Clinical Trials Communications, 26:100897.
Publisher | Google Scholor - Gewandter, J. S., Chaudari, J., Iwan, K. B., Kitt, R., As-Sanie, S., et al. (2018). Research Design Characteristics of Published Pharmacologic Randomized Clinical Trials for Irritable Bowel Syndrome and Chronic Pelvic Pain Conditions: An ACTTION Systematic Review. The Journal of Pain, 19(7):717-726.
Publisher | Google Scholor - Kris-Etherton, P. M., Petersen, K. S., Lamarche, B., Karmally, W., Guyton, J. R., et al. (2024). The Role of Nutrition-Related Clinical Trials in Informing Dietary Recommendations for Health and Treatment of Diseases. Journal of Clinical Lipidology.
Publisher | Google Scholor - Bloom, P. P., Chung, R. T. (2024). The Future of Clinical Trials of Gut Microbiome Therapeutics in Cirrhosis. JHEP Reports, 101234.
Publisher | Google Scholor - Zhang, Y., You, P., Liu, R., Lu, Y., Li, J., et al. (2024). Artificial Intelligence in Clinical Trials of Lung Cancer: Current and Future Prospects. Intelligent Oncology.
Publisher | Google Scholor - Schultz, A., Saville, B. R., Marsh, J. A., Snelling, T. L. (2019). An Introduction to Clinical Trial Design. Paediatric Respiratory Reviews, 32:30-35.
Publisher | Google Scholor - Pocock, S. J. (1985). Current Issues in The Design and Interpretation of Clinical Trials. Br Med J (Clin Res Ed), 290(6461):39-42.
Publisher | Google Scholor - Pocock, S. J. (1997). Clinical Trials with Multiple Outcomes: A Statistical Perspective on Their Design, Analysis, And Interpretation. Controlled Clinical Trials, 18(6):530-545.
Publisher | Google Scholor - Ceelen, W., Soreide, K. (2023). Randomized Controlled Trials and Alternative Study Designs in Surgical Oncology. European Journal of Surgical Oncology, 49(8):1331-1340.
Publisher | Google Scholor - Maltais, F., Vogelmeier, C. F., Kerwin, E. M., Bjermer, L. H., Jones, P. W., et al. (2022). Applying Key Learnings from The EMAX Trial to Clinical Practice and Future Trial Design in COPD. Respiratory Medicine, 200:106918.
Publisher | Google Scholor - McDermott, M., Meah, M. N., Khaing, P., Wang, K. L., Ramsay, J., et al. (2024). Rationale and Design of SCOT-HEART 2 Trial: CT Angiography for The Prevention of Myocardial Infarction. Cardiovascular Imaging, 17(9):1101-1112.
Publisher | Google Scholor - Bogaerts, J., Sydes, M. R., Keat, N., McConnell, A., Benson, A., et al. (2015). Clinical Trial Designs for Rare Diseases: Studies Developed and Discussed by The International Rare Cancers Initiative. European Journal of Cancer, 51(3):271-281.
Publisher | Google Scholor - Best, A. M., Lang, T. A., Greenberg, B. L., Gunsolley, J. C., Ioannidou, E., et al. (2024). The OHStat Guidelines for Reporting Observational Studies and Clinical Trials in Oral Health Research: Explanation and Elaboration. Journal of Oral and Maxillofacial Surgery.
Publisher | Google Scholor - Soler, M., Lu, E., Masch, R., Alfaro, K., Anderson, J. R., et al. (2023). Affordable Cancer Technologies: Lessons Learned from The Design and Implementation of Two Randomized Clinical Trials to Develop Innovative Treatments for Cervical Precancer. Contemporary Clinical Trials Communications, 36:101224.
Publisher | Google Scholor - Sutherland, L., Carter, L. (2024). Sex As a Biological Variable in Early-Phase Oncology Clinical Trials: Enhancing the Path to Personalised Medicine. Heliyon, 10(12).
Publisher | Google Scholor - Mauri, L., D’Agostino Sr, R. B. (2017). Challenges in the Design and Interpretation of Noninferiority Trials. New England Journal of Medicine, 377(14):1357-1367.
Publisher | Google Scholor - Bhide, A., Shah, P. S., Acharya, G. (2018). A Simplified Guide to Randomized Controlled Trials. Acta Obstetricia et Gynecologica Scandinavica, 97(4):380-387.
Publisher | Google Scholor - Finkelstein, A., Zhou, A., Taubman, S., Doyle, J. (2020). Health Care Hotspotting-A Randomized, Controlled Trial. New England Journal of Medicine, 382(2):152-162.
Publisher | Google Scholor - Harrer, S., Shah, P., Antony, B., Hu, J. (2019). Artificial Intelligence for Clinical Trial Design. Trends in Pharmacological Sciences, 40(8):577-591.
Publisher | Google Scholor