Research Article
Evaluation of Antiepileptic Properties of Ethanol Extract of Blighia Sapida Leaves on Pentylenetetrazole-Induced Epileptogenesis in Adult Male Albino Mice
- John K. Okoro 1*
- Efejeror D. Okereka 1
- Mayowa O. Adu 2
- Emmanuel O. Ogbuagu 3
- Prince C. Unekwe 3
- Augustine T. Ezike 2
- John A. Ekenjoku 3
- Ejieh Nnamdi 2
- Adetula A. Aderopo 4
- Adetula A. Aderopo 5
- Joy S. Ajenikoko 5
- Ifeychukwudi O. Dibie 6
- Bright C. Chukwu 7
- Michael O. Familua 8
1 Department of Pharmacology and Therapeutics, Faculty of Basic Clinical Sciences, Afe Babalola University, Ado-Ekiti, Ekiti State, Nigeria.
2 Department of Human Physiology, Faculty of Basic Medical Sciences, Afe Babalola University, Ado-Ekiti, Ekiti State, Nigeria.
3 Department of Pharmacology and Therapeutics, Faculty of Basic Clinical Sciences, Abia State University, Uturu, Nigeria.
4 Department of Optometry, Faculty of Allied Health, Afe Babalola University, Ado-Ekiti, Ekiti State, Nigeria.
5 Department of Public Health, Faculty of Basic Medical Sciences, Bamidele Olumilua University of Education Science and Technology, Ikere, Ekiti State, Nigeria.
6 Department of Pharmacology and Therapeutics, Faculty of Basic Clinical Sciences, Delta State University, Delta State, Nigeria.
7 Department of Human Anatomy, Faculty of Basic Medical Sciences, Afe Babalola University, Ado-Ekiti, Ekiti State, Nigeria.
8 Department of Pharmacology and Toxicology, Faculty of Pharmacy, Afe Babalola University, Ado-Ekiti, Ekiti State, Nigeria.
*Corresponding Author: John K. Okoro, Department of Pharmacology and Therapeutics, Faculty of Basic Clinical Sciences, Afe Babalola University, Ado-Ekiti, Ekiti State, Nigeria.
Citation: John K. Okoro, Okereka D Efejeror, Adu O Mayowa, Emmanuel O Ogbuagu, Prince C Unekwe, et al., (2026). Evaluation of Antiepileptic Properties of Ethanol Extract of Blighia Sapida Leaves on Pentylene-tetrazole-Induced Epileptogenesis in Adult Male Albino Mice. Journal of Neuroscience and Neurological Research. BioRes Scientia Publishers. 5(1):1-9. DOI: 10.59657/2837-4843.brs.26.037
Copyright: © 2026 Okoro John Kalu, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: December 16, 2025 | Accepted: December 30, 2025 | Published: January 12, 2026
Abstract
Epilepsy affects over 50 million people globally, with limited treatment access in low-income regions. Despite advancements in antiepileptic drugs, adverse effects and treatment resistance remained significant challenges. Blighia sapida (Cupania sapida voigt) known for its rich content of flavonoids, saponins, and alkaloids, demonstrated anticonvulsant potential by mitigating oxidative stress, reducing neuroinflammation, and enhancing GABAergic activity in PTZ-induced seizure models. This study aimed to evaluate the antiepileptic properties of ethanol extract of Blighia sapida leaves in PTZ-induced epilepsy in male albino mice by inducing seizures with PTZ, administering the extract and sodium valproate, and comparing their effects while exploring the extract’s possible mechanisms of action. Ethanol extracts from authenticated, shade-dried leaves were prepared and administered orally to 15 male mice for 22 days following ethical approval; seizure activity was assessed using behavioural scoring and biochemical assays (MDA, NO, SOD), while acute toxicity was determined via Lorke’s method and phytochemicals identified using the method outlined by Junaid R. Shaikh and MK Patil. PTZ-induced seizure severity was highest in the PTZ-only group, lowest in the PTZ+SV group, and moderate in extract-treated groups, with 400 mg/kg showing the most significant reduction in PTZ effects and delayed seizure onset (p < 0.0001), confirming dose-dependent anticonvulsant efficacy. Blighia sapida leaf extract exhibited potent anticonvulsant activity, especially at 400 mg/kg, with effects comparable to sodium valproate a standard, likely due to active ingredients responsible for anti-seizure effect contained in the flavonoids, alkaloids, tannins, or phenolics groups which should be identified, separated and characterized, hence enhanced antioxidant defense and reduced neuronal excitability without observed toxicity up to 1000 mg/kg. These findings indicated that its mechanism of action may involve antioxidant-mediated stabilization of neuronal membranes and modulation of GABAergic or glutamatergic neurotransmission, making Blighia sapida a promising, accessible alternative or adjunct for epilepsy treatment, particularly in underserved communities.
Keywords: epilepsy; pentylenetetrazole; ethanol; blighia sapida; extract; nitric oxide; malondiadehyde; superoxide dismutase
Introduction
Epilepsy is a chronic neurological disorder marked by recurrent, unprovoked seizures resulting from abnormal and excessive neuronal discharges in the brain. It remains one of the most prevalent neurological conditions globally, affecting an estimated 50 million individuals, with nearly 80% of cases occurring in low and middle-income countries where access to effective treatment is often inadequate [1]. Although substantial progress has been made in the development of antiepileptic drugs, a significant proportion of patients continue to experience drug resistance, suboptimal seizure control, or treatment-limiting adverse effects. These challenges have intensified the search for safer and more accessible therapeutic alternatives, including plant-derived remedies [2].
Medicinal plants have attracted considerable scientific interest due to their diverse bioactive constituents and long-standing use in traditional healthcare systems. Increasing evidence suggests that many plant-derived compounds possess neuroprotective and anticonvulsant properties while exhibiting a more favourable safety profile than synthetic agents [3]. Consequently, contemporary epilepsy research has focused on identifying phytochemicals capable of modulating neuronal excitability, reducing oxidative stress, and attenuating neuroinflammatory processes that contribute to epileptogenesis.
Blighia sapida, commonly known as akee, is a tropical plant indigenous to Africa and widely utilized for both nutritional and medicinal purposes. Its leaves are rich in secondary metabolites, including flavonoids, alkaloids, saponins, tannins, and phenolic compounds, which have been reported to exert antioxidant, anti-inflammatory, and neuromodulatory effects [4]. Given the central role of oxidative stress and neuroinflammation in seizure initiation and propagation, the phytochemical profile of B. sapida suggests a plausible therapeutic role in epilepsy management. Previous experimental studies have demonstrated that plant-based extracts can influence excitatory and inhibitory neurotransmitter systems, enhance gamma aminobutyric acid-mediated inhibition, and suppress neuroinflammatory signalling pathways involved in seizure progression [3].
The pentylenetetrazole-induced seizure model is a well-established experimental paradigm for evaluating antiepileptic activity. PTZ acts primarily as a gamma-aminobutyric acid A receptor antagonist, leading to neuronal hyperexcitability and generalized seizures that closely resemble human epileptic manifestations [5]. This model is particularly valuable for assessing both acute anticonvulsant effects and long-term neuroprotective outcomes. Several studies using PTZ kindling have shown that flavonoids and other phytochemicals enhance inhibitory neurotransmission and reduce seizure severity [6]. In addition, neuroinflammatory mediators such as cytokines and nitric oxide have been implicated in PTZ-induced epileptogenesis, further highlighting the relevance of plant-derived anti-inflammatory agents [7].
Oxidative stress represents another critical mechanism underlying seizure related neuronal injury. Excessive generation of reactive oxygen species during seizures promotes lipid peroxidation, protein oxidation, and neuronal degeneration, thereby perpetuating epileptic activity [6]. Flavonoids present in B. sapida possess strong free radical scavenging properties and are capable of restoring redox balance within neural tissues. The combined presence of flavonoids, alkaloids, and saponins in B. sapida suggests a synergistic pharmacological effect, whereby multiple molecular targets involved in seizure generation and propagation are simultaneously modulated. This multitarget approach is increasingly recognized as a desirable strategy in antiepileptic drug development [3].
Materials And Methods
Plant Collection, Identification, and Authentication
Fresh leaves of Blighia sapida were obtained and authenticated by a certified plant taxonomist in the Department of Botany, Faculty of Science, Ekiti State University. Proper identification and authentication were conducted in accordance with standard botanical procedures, and a voucher specimen was deposited with authentication number 2025042.
Experimental Animals
Thirty adult male albino mice weighing between 22 and 25 g were used. The animals were sourced from the Central Animal House of Afe Babalola University, Ado-Ekiti. They were housed in wire gauze cages under standard laboratory conditions at an ambient temperature of approximately 22°C with adequate illumination. The mice were acclimatized for two weeks prior to the commencement of the study and had unrestricted access to commercial feed and clean drinking water. Ethical approval for all experimental procedures was obtained from the University Animal Care and Use Research Ethics Committee.
Drugs, Reagents, and Equipment
Pentylenetetrazole was obtained from Sigma Chemical Company, USA. Sodium valproate and epinephrine were purchased from the Afe Babalola University Pharmacy. Analytical grade ethanol, buffers, and reagents were sourced from reputable suppliers. All laboratory equipment used, including a spectrophotometer, centrifuge, rotary evaporator, Soxhlet extractor, and water bath, was available in the Pharmacology Laboratory of Afe Babalola University, Ado-Ekiti.
Extraction Procedure
The collected leaves were shade-dried at room temperature and pulverized into fine powder. The powdered material was extracted using 50% ethanol in a Soxhlet apparatus for 48 hours. The resulting filtrate was concentrated under reduced pressure using a rotary evaporator at low temperature. The dried extract was stored at refrigerated conditions, and fresh solutions were prepared with distilled water prior to administration. The extract was administered orally once daily for 22 consecutive days.
Phytochemical Screening
Qualitative phytochemical analysis of the ethanol leaf extract was performed using standard methods to identify the presence of alkaloids, flavonoids, saponins, tannins, phenolics, carbohydrates, and steroids, following established protocols [8].
Acute Toxicity and LD50 Determination
Acute Toxicity Study
The acute toxicity study of the extract of Blighia sapida leaves was performed using the method proposed by Lorke in [9]. This method is divided into phase 1 and phase 2
LD50 Experiment
Phase 1: In phase 1, the number of mice used was nine (9). The nine animals were divided into three (3) groups with three animals each. The first group of animals were administered 10mg/kg dose of the extract, the second group was administered a 100 mg/kg dose of the extract and the third group was administered a 1000 mg/kg dose of the extract. Administration of the extract was done orally.
The animals were kept under observation for 24 hours to monitor their behaviour as well as to determine if mortality would occur. However, there was no change in their behaviours, nor did any mortality occur.
Phase 2: In phase 2, three mice were used, which were distributed into three groups of one mouse each. The first group was administered a 1600 mg/kg dose of the extract. The second group was administered a 2900 mg/kg dose of the extract, and the third group was given a 5000 mg/kg dose of the extract. The animals were observed for 24 hours for behavioural change as well as mortality. At the end of the 24 hours of observation, it was noticed that the animals in the first and second groups had behavioral changes. The animal in the first group sneezed continuously, as well as being very restless, with watery poop all over its body. The animal in the second group had a woolly jaw and was covered in yellowish urine and unable to explore within the cage. The animal in the third group, which received a 5000 mg/kg dose of the extract, had died within 24 hours of extract administration.
Calculation of LD50
LD50 is the square root of the product of the minimal lethal dose and the maximal lethal dose.
From the result of the LD50, 1600 mg/kg extract is the minimal lethal dose, while 5000 mg/kg is the maximal lethal dose
LD50 = square root of 1600 multiplied by 5000 = square root of 8,000,000 = 2828.43.
To determine the margin of safety of the extract dose, we would have to find 20% of 2828.43
i.e 20/100 multiply by 2828.43
Therefore, the margin of safety = 566
Induction of Epilepsy and Experimental Design
Epilepsy was induced using the PTZ kindling model as previously described [10]. The mice were randomly assigned to six groups of five animals each. Group one served as the control and received distilled water. Group two received PTZ only and served as the disease control. Groups three to five received PTZ in combination with B. sapida extract at doses of 100, 200, and 400 mg/kg, respectively. Group six received PTZ and sodium valproate at 100 mg/kg. PTZ was administered intraperitoneally at 40 mg/kg on alternate days for 22 days. Seizure activity was observed and scored using a standard seizure severity scale [11].
Biochemical Assays
At the end of the experimental period, animals were sacrificed, and brain tissues were harvested, homogenized, and processed for biochemical analysis. Oxidative stress markers, including malondialdehyde, nitric oxide, and superoxide dismutase, were assessed using standard spectrophotometric methods [12–14].
Statistical Analysis
Data were expressed as mean ± standard error of the mean. Statistical analysis was performed using two-way analysis of variance followed by Bonferroni post hoc test. Independent t-tests were applied for oxidative stress markers. Statistical significance was set at p less than 0.05.
Results
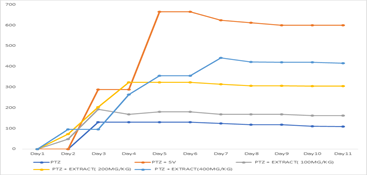
The mean PTZ (pentylenetetrazol) levels across different treatment groups over 11 days are presented in Table 2. The groups include PTZ-only, PTZ+SV (Standard Vehicle), and PTZ with different extract doses (100, 200, and 400 mg/kg). The results show that on Day 1, all groups had a PTZ level of 0, indicating no immediate differences. From Day 2 onwards, PTZ-only levels increased steadily, reaching 2.9 on Day 11. PTZ+SV treatment showed a slower increase in PTZ effects but peaked at 1.9 on Day 11. Extract treatments exhibited a dose-dependent effect, with higher doses leading to lower PTZ levels: 100 mg/kg: PTZ levels peaked at 2.7 on Day 11. 200 mg/kg: PTZ levels remained relatively lower (2.3 on Day 11). 400 mg/kg: PTZ levels were the lowest across all extract groups (2.0 on Day 11). Generally, the results show that higher extract doses appear to reduce PTZ-induced effects, with 400 mg/kg being the most effective.
Statistics for each treatment group over the experimental period with the following observations (as presented in Table 3): PTZ-only group had the highest mean value (1.93) with a high standard deviation (SD = 1.08), indicating large fluctuations. PTZ+SV group had a slightly lower mean (1.53) but a higher SD (1.42). Extract treatments showed reduced means and variability: 100 mg/kg: Mean = 1.99, SD = 1.14, 200mg/kg: Mean = 1.65, SD = 0.93 and 400mg/kg: Mean = 1.56, SD = 1.12. The results show that extract treatments, particularly at higher doses, stabilize PTZ effects, reducing both mean values and variability.
ANOVA Analysis for PTZ+Extract (Table 4). This table presents the results of an ANOVA test, which determines whether differences between groups are statistically significant. The results show the Day effect (p less than 0.0001): Significant variation in PTZ effects over time, and the Treatment effect (p = 0.0011) is a significant effect of extract treatments. The interaction effect (Day * Treatment) (p = 0.0615): was not statistically significant.
The Least Significant Difference (LSD) test results for comparisons among treatment groups is presented in Table 5. The PTZ-only vs. PTZ+SV comparison shows a mean difference of 0.40545 (p = 0.045), suggesting a significance in their differences. The PTZ+SV vs. PTZ+Extract (100 mg/kg) comparison is statistically significant (p = 0.038), indicating that the extract at this dose has a different effect from PTZ+SV. Other comparisons among extract groups are not statistically significant (p > 0.05). Therefore, PTZ Extract (100 mg/kg) significantly differs from PTZ+SV,
The changes in PTZ levels across different treatment groups over 11 days is presented in Table 6. Here are some key observations. PTZ-only group (blue line) shows a steady but lower increase in PTZ effects. PTZ + SV group (orange line) exhibits a rapid increase, peaking around Day 5 and maintaining a high level. PTZ + Extract groups show dose-dependent variations. 100 mg/kg (gray line) results in a mild increase, stabilizing at a lower level. 200mg/kg (yellow line) has a moderate effect, maintaining PTZ levels lower than PTZ + SV. 400mg/kg (dark blue line) shows the most suppression of PTZ effects, increasing but plateauing at a significantly lower level.
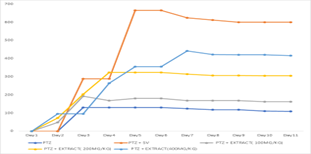
The results presented in Table 7 show that PTZ-only group consistently had the shortest seizure times (~99 seconds on average). Extract-treated groups had significantly longer seizure latencies: 100mg/kg: ~144 seconds, 200mg/kg: ~252 seconds and 400mg/kg: ~307 seconds. The ANOVA analysis shows (p less than 0.0001), confirming that treatment significantly impacts seizure latency. Higher doses of extract significantly delay seizure onset, confirming potential anticonvulsant properties.
No statistically significant differences between GP1 and other groups (p > 0.05), suggesting similar responses (Table 8). GP2 vs. GP3, GP4, GP5, and GP6 also showed no significant differences (p > 0.05), indicating a consistent treatment effect across groups. All group comparisons had wide confidence intervals, which may indicate variability in the data.
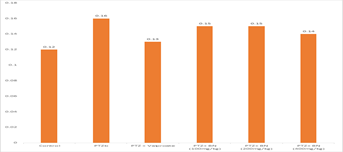
Table 9 presents the mean PTZ (pentylenetetrazol) levels across Enzyme micromole/g protein NO, nmole/g protein MDA and U/mg protein SOD. The results show that at Enzyme micromole/g protein NO, there were significant differences between PTZ only and other levels of PTZ at 0.05 level, except PTZ+ BS (100mg/kg). Likewise, at nmole/g protein MDA, the control and the PTZ only were significantly different from other levels of PTZ. Result in U/mg protein SOD showed the three levels of PTZ +BS (100 mg/kg), PTZ +BS (200 mg/kg) and PTZ +BS (400 mg/kg) significantly different from PTZ only.
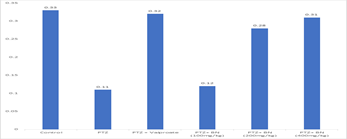
The control group exhibited a baseline enzyme level of 0.33 µmol/g protein (Figure 3). PTZ significantly reduced enzyme activity to 0.11 µmol/g, indicating oxidative damage or neurotoxicity. Valproate co-administration restored activity to near-normal (0.32 µmol/g), suggesting protective effects. Blighia sapida at 100 mg/kg failed to improve enzyme levels (0.12 µmol/g), showing limited efficacy at low doses. However, the 200 mg/kg and 400 mg/kg doses improved enzyme activity to 0.28 µmol/g and 0.31 µmol/g, respectively, demonstrating dose-dependent neuroprotection. The results suggest that higher doses of Blighia sapida leaf extract may counteract PTZ-induced enzyme suppression effectively.
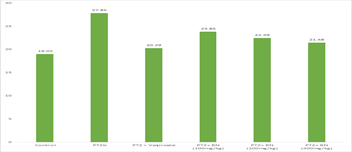
Malondialdehyde (MDA) levels were lowest in the control group (19.02 nmol/g protein), indicating minimal lipid peroxidation (Figure 4). PTZ administration caused a substantial rise (27.85 nmol/g), signifying increased oxidative stress. Valproate co-treatment reduced MDA levels to 20.29 nmol/g, indicating its antioxidant property. Blighia sapida at 100 mg/kg showed a moderate reduction to 23.85 nmol/g, while 200 mg/kg and 400 mg/kg further reduced MDA to 22.49 and 21.48 nmol/g, respectively.
Table 1: Qualitative Phytochemical Analysis of Extract
| Phytochemical | Observation |
| Alkaloid | + + |
| Flavonoid | + + + |
| Amino Acid | + + |
| Phenolic compounds | + + + |
| Tannins | + + + |
| Phytosterols | + + |
| Reducing Sugar | + + |
| Saponin | Negative |
| Phlobatannins | + + |
+ (plus) is an observation showing the presence of a phytochemical. The number of pluses reveals the degree of presence of a phytochemical.
Table 2: The Effect of Sodium Valproate and Three Doses of Blihia Sapida on the PTZ-Induced Seizure
| Day | PTZ | PTZ+SV | PTZ+EXT (100 mg/kg) | PTZ+EXT (200 mg/kg) | PTZ+EXT (400 mg/kg) |
| Day 1 | 0 | 0 | 0 | 0 | 0 |
| Day 2 | 0 | 0 | 0.4 | 0.4 | 0.4 |
| Day 3 | 1.9 | 0.4 | 1.6 | 1 | 0.7 |
| Day 4 | 2.1 | 0.5 | 2.0 | 1.8 | 1.0 |
| Day 5 | 2.3 | 0.7 | 2.1 | 2 | 1.2 |
| Day 6 | 2.3 | 1 | 2.2 | 2 | 1.4 |
| Day 7 | 2.5 | 1.3 | 2.3 | 2.1 | 1.6 |
| Day 8 | 2.6 | 1.5 | 2.4 | 2.1 | 1.8 |
| Day 9 | 2.7 | 1.7 | 2.4 | 2.1 | 1.8 |
| Day10 | 2.7 | 1.7 | 2.6 | 2.3 | 2 |
| Day11 | 2.9 | 1.9 | 2.7 | 2.3 | 2 |
Figure 1: Mean Trend of Sodium Valproate and Three Doses of Blihia Sapida on the PTZ-Induced Seizure.
Table 3: Descriptive Statistics of Each Treatment Group Over 11 Day Experimental Period
| Parameter | Sample | Sum | Average | SD | SEM |
| PTZ | 55 | 106.3 | 1.932727 | 1.085435 | 0.14636 |
| PTZ+SV | 55 | 84 | 1.527273 | 1.422108 | 0.191757 |
| PTZ+EXT(100mg/kg) | 55 | 109.3 | 1.987273 | 1.148599 | 0.154877 |
| PTZ+EXT(200mg/kg) | 55 | 91 | 1.654545 | 0.932305 | 0.125712 |
| PTZ+EXT(400mg/kg) | 55 | 86 | 1.563636 | 1.126509 | 0.151899 |
Table 4: Analysis of Variance Test (ANOVA) Showing Statistically Significant Differences
| Source of Variation | SS | Df | MS | F | P-value | F crit |
| Day | 213.608 | 10 | 21.3609 | 40.4981 | 1.59E-44 | 1.8739 |
| Treatment+Control | 9.99396 | 4 | 2.4985 | 4.7369 | 0.001099 | 2.4127 |
| Interaction(day*Treatment) | 29.8860 | 40 | 0.7472 | 1.4165 | 0.061499 | 1.4496 |
| Error | 116.04 | 220 | 0.5275 |
Table 5: Comparison of Results among Treatment Groups
| Dependent Variable: TREATMENT LSD | ||||||
| (I) FACTOR | Mean Difference (I-J) | Std. Error | Sig. | 95% Confidence Interval | ||
| Lower Bound | Upper Bound | |||||
| PTZ | PTZ+SV | .40545 | .22005 | .045 | -.0278 | .8387 |
| PTZ+EXTRACT (100 mg/kg) | -.05455 | .22005 | .804 | -.4878 | .3787 | |
| PTZ+EXTRACT (200MG/KG) | .27818 | .22005 | .207 | -.1551 | .7114 | |
| PTZ+EXTRACT (400 mg/kg) | .36909 | .22005 | .095 | -.0641 | .8023 | |
| PTZ+SV | PTZ+EXTRACT (100 mg/kg) | -.46000* | .22005 | .038 | -.8932 | -.0268 |
| PTZ+EXTRACT (200 mg/kg) | -.12727 | .22005 | .563 | -.5605 | .3060 | |
| PTZ+EXTRACT (400 mg/kg) | -.03636 | .22005 | .869 | -.4696 | .3969 | |
| PTZ+EXTRACT (100 MG/KG) | PTZ+EXTRACT (200 mg/kg) | .33273 | .22005 | .132 | -.1005 | .7660 |
| PTZ+EXTRACT (400 mg/kg) | .42364 | .22005 | .055 | -.0096 | .8569 | |
| PTZ+EXTRACT (200MG/KG) | PTZ+EXTRACT (400 mg/kg) | .09091 | .22005 | .680 | -.3423 | .5241 |
Table 6: Analysis of Mean Time of Seizure Across 11 Days
| Day | PTZ | PTZ + SV | PTZ + EXTRACT (100 mg/kg) | PTZ + EXTRACT (200 mg/kg) | PTZ + EXTRACT (400 mg/kg) |
| Day1 | 0 | 0 | 0 | 0 | 0 |
| Day2 | 0 | 0 | 48 | 72 | 96 |
| Day3 | 130 | 288 | 192 | 204 | 96 |
| Day4 | 130 | 288 | 168 | 324 | 264.8 |
| Day5 | 130 | 666 | 180 | 324 | 356 |
| Day6 | 130 | 666 | 180 | 324 | 356 |
| Day7 | 124 | 624 | 168 | 314 | 442 |
| Day8 | 118 | 612 | 168 | 307 | 422 |
| Day9 | 118 | 600 | 168 | 306 | 421 |
| Day10 | 111 | 600 | 162 | 305.4 | 420 |
| Day11 | 109 | 600 | 162 | 305.2 | 416 |
Figure 2: PTZ + Extract Mean Over Time Trend
Table 7: ANOVA Test of Seizure Time (Onset of Action) Across All Five Groups Over Eleven Days
| Source of Variation | SS | df | MS | F | P-value | F crit |
| PTZ + Extract | 3818863 | 10 | 381886.3 | 34.63006 | 6.48E-40 | 1.873923 |
| Days | 4202815 | 4 | 1050704 | 95.27951 | 6.77E-47 | 2.412682 |
| Interaction | 1855173 | 40 | 46379.31 | 4.205751 | 3.05E-12 | 1.449587 |
| Within | 2426071 | 220 | 11027.59 | |||
| Total | 12302921 | 274 |
Table 8: Comparing Statistically Significant Differences Between Groups Using an Independent T-Test
| (I) GP | Mean Difference (I-J) | Std. Error | Sig. | 95% Confidence Interval | ||
| Lower Bound | Upper Bound | |||||
| GP1 | GP2 | -40.12500 | 35.90867 | .279 | -115.5663 | 35.3163 |
| GP3 | -32.26000 | 35.90867 | .381 | -107.7013 | 43.1813 | |
| GP4 | -18.23500 | 35.90867 | .618 | -93.6763 | 57.2063 | |
| GP5 | -15.10500 | 35.90867 | .679 | -90.5463 | 60.3363 | |
| GP6 | -16.53500 | 35.90867 | .651 | -91.9763 | 58.9063 | |
| GP2 | GP3 | 7.86500 | 35.90867 | .829 | -67.5763 | 83.3063 |
| GP4 | 21.89000 | 35.90867 | .550 | -53.5513 | 97.3313 | |
| GP5 | 25.02000 | 35.90867 | .495 | -50.4213 | 100.4613 | |
| GP6 | 23.59000 | 35.90867 | .520 | -51.8513 | 99.0313 | |
| GP3 | GP4 | 14.02500 | 35.90867 | .701 | -61.4163 | 89.4663 |
| GP5 | 17.15500 | 35.90867 | .639 | -58.2863 | 92.5963 | |
| GP6 | 15.72500 | 35.90867 | .667 | -59.7163 | 91.1663 | |
| GP4 | GP5 | 3.13000 | 35.90867 | .932 | -72.3113 | 78.5713 |
| GP6 | 1.70000 | 35.90867 | .963 | -73.7413 | 77.1413 | |
| GP5 | GP6 | -1.43000 | 35.90867 | .969 | -76.8713 | 74.0113 |
Table 9: The Effect of Pentylenetetrazole (PTZ), Sodium Valproate (Sv) and Three Doses of Blighia sapida on the Nitric Oxide (NO), Malondialdehyde (MDA) and Superoxide Dismutase (SOD) Levels of Brain Tissue on PTZ-Induced Seizure in Mice
| Parameters | Enzyme micromole/g protein NO | nmole/g protein MDA | U/mg protein SOD |
| Controla | 0.33 0.01 0.01 | 19.02 | 0.12 0.007 0.007 |
| PTZb | 0.11 0.02 0.02 | 27.85 0.37 0.37 | 0.16 0.023 0.023 |
| PTZ + Valproate | 0.32 0.01b 0.01b | 20.29 0.34ab 0.34ab | 0.13 b b |
| PTZ+ BS (100 mg/kg) | 0.12 0.02a 0.02a | 23.85 0.19ab 0.19ab | 0.15 ab ab |
| PTZ+ BS (200 mg/kg) | 0.28 0.03b 0.03b | 22.49 ab ab | 0.15 ab ab |
| PTZ+ BS (400 mg/kg) | 0.31 0.03b 0.03b | 21.48 0.20b 0.20b | 0.14 ab ab |
a Shows a significant difference as compared to the control, b shows a significant difference from PTZ, and ab shows a significant difference from the control and PTZ.
Figure 3: Enzyme (micromole/g protein) NO
Figure 4: Malondialdehyde (MDA) Level
Figure 5: Superoxide dismutase (SOD) activity
Discussion
Phytochemical screening revealed that Blighia sapida leaf extract contains a diverse array of biologically active compounds, notably flavonoids, alkaloids, phenolics, tannins, and phytosterols. These constituents are widely recognized for their antioxidant, anti-inflammatory, and neuroprotective properties, and their presence provides a strong biochemical rationale for the observed anticonvulsant effects. Flavonoids and phenolic compounds, in particular, are known to modulate neurotransmitter systems and suppress neuronal excitotoxicity, both of which play central roles in epileptogenesis [15].
The acute toxicity assessment demonstrated that the extract possesses a wide safety margin, with no mortality observed at doses up to 2900 mg/kg. The calculated LD50 of approximately 2828 mg/kg indicates low acute toxicity, supporting the selection of 100 to 400 mg/kg as safe and pharmacologically relevant doses. This favorable safety profile enhances the therapeutic appeal of B. sapida as a potential antiepileptic agent.
In the PTZ-induced seizure model, the extract produced a clear dose-dependent reduction in seizure severity and frequency. The highest dose exhibited substantial anticonvulsant activity that closely approached the effect of sodium valproate. These findings align with previous reports demonstrating that plant-derived flavonoids and alkaloids enhance inhibitory neurotransmission and counteract PTZ-induced hyperexcitability [5,16]. The prolongation of seizure onset latency further confirms the protective role of the extract against epileptogenic stimuli.
Biochemical analysis revealed that PTZ exposure significantly disrupted oxidative balance, as evidenced by elevated lipid peroxidation and altered antioxidant enzyme activity. Treatment with B. sapida extract markedly improved these parameters, suggesting effective attenuation of oxidative stress. The reduction in malondialdehyde levels and normalization of nitric oxide and superoxide dismutase activities indicate restoration of redox homeostasis, which is critical for neuronal survival and seizure control [12–14].
The convergence of behavioral and biochemical findings suggests that the anticonvulsant activity of Blighia sapida is mediated through a combination of antioxidant defence enhancement, neuroinflammatory suppression, and modulation of inhibitory neurotransmission. The synergistic action of its phytochemical constituents likely underpins these effects.
The anticonvulsant effect of Blighia sapida leaf extract may be attributed to the enhancement of gamma aminobutyric acid mediated inhibitory neurotransmission, combined with its antioxidant activity mediated largely by flavonoids and phenolic compounds.
Conclusion
This study demonstrates that ethanol leaf extract of Blighia sapida exhibits significant anticonvulsant activity in a PTZ-induced epilepsy model, with the 400 mg/kg dose producing effects comparable to sodium valproate. The extract showed a favourable safety profile and effectively reduced seizure severity while improving oxidative stress biomarkers. These findings support the therapeutic potential of Blighia sapida as a natural antiepileptic agent and provide a scientific basis for its traditional use in neurological disorders.
Recommendations For Future Studies
Future research should focus on isolating and characterizing the specific bioactive compounds responsible for the observed anticonvulsant effects. Detailed mechanistic studies exploring interactions with neurotransmitter receptors and ion channels are warranted. Long term toxicity studies and clinical trials are also recommended to establish safety, efficacy, and translational relevance. Formulation development aimed at improving bioavailability and consistency would further support therapeutic application.
References
- .World Health Organization. (2024). Epilepsy.
Publisher | Google Scholor - Adedoyin, A. M., Ezeanya, O. C., & Okorie, et al., (2024). Medicinal plants for epileptic seizures: Phytoconstituents and mechanisms. Journal of Ethnopharmacology, 315:15647.
Publisher | Google Scholor - Chen, H., Li, X., & Zhao, Y. (2024)., Application of LC-MS and NMR spectroscopy in herbal drug research: Identification of bioactive compounds in antiepileptic plants. Phytochemical Analysis, 35(1):72–85.
Publisher | Google Scholor - Ishola, R. O., Akande, I. S., & Omobowale, et al., (2024). Pharmacological potentials of Blighia sapida in traditional medicine. African Journal of Traditional, Complementary and Alternative Medicines, 21(1):56–66.
Publisher | Google Scholor - Murphy, C. A., Salako, L. A., & Enwere, et al., (2024). Seizure induction and management in PTZ-kindled animal models: A pharmacological review. Neuroscience Letters, 831:137248.
Publisher | Google Scholor - Singh, P., & Kumar, R. (2024). Antioxidant and anticonvulsant properties of flavonoids in PTZ-induced epilepsy. Journal of Neuroscience Research, 102(4):564–572.
Publisher | Google Scholor - Okonkwo, E. C., Adebayo, R. A., & Musa, et al., (2025). Anti-inflammatory effects of saponins from medicinal plants and their role in epilepsy. Journal of Inflammation Research, 1: 112–123.
Publisher | Google Scholor - Junaid, R. S., & Patil, M. K. (2020). Qualitative tests for preliminary phytochemical screening. Chemistry Journal.
Publisher | Google Scholor - Lorke, D. (1983). A new approach to practical acute toxicity testing. Archives of Toxicology, 54:275–287.
Publisher | Google Scholor - Kiasalari, M., & Khalili, M. (2012). Antiepileptic and antioxidant effect of Brassica nigra on pentylenetetrazol-induced kindling in mice. Iranian Journal of Pharmaceutical Research, 11:1209–1217.
Publisher | Google Scholor - Racine, R. J. (1972). Modification of seizure activity by electrical stimulation. Electroencephalography and Clinical Neurophysiology, 32:281–294.
Publisher | Google Scholor - Ohkawa, H., Ohishi, N., & Yagi, K. (1979). Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Analytical Biochemistry, 95:351–358.
Publisher | Google Scholor - Misra, H. P., & Fridovich, I. (1972). The role of superoxide anion in the autoxidation of epinephrine and a simple assay for superoxide dismutase. Journal of Biological Chemistry, 247:3170–3175.
Publisher | Google Scholor - Green, L. C., Wagner, D. A., Glogowski, J., Skipper, P. L., Wishnok, J. S., & Tannenbaum, S. R. (1982). Analysis of nitrate, nitrite, and [15N] nitrate in biological fluids. Analytical Biochemistry, 126:131–138.
Publisher | Google Scholor - Omotayo, F. O., & Faniyi, T. O. (2024). Nutritional and phytochemical evaluation of Blighia sapida. Journal of Medicinal Food Research, 18(2):45–56.
Publisher | Google Scholor - Löscher, W., & Schmidt, D. (2025). New horizons in antiepileptic drug development. Epilepsy Research, 190:107021.
Publisher | Google Scholor