Research Article
Effect of Livolin Forte on Cognitive Impairment Caused by Sub-Chronic Lead Acetate Administration in Wistar Rats
1Department of Research, Innovation and Technology Development, Federal College of Fisheries and Marine Technology, Victoria Island, Lagos, Nigeria.
2Department of Veterinary Pharmacology and Toxicology, Ahmadu Bello University, Zaria, Kaduna State, Nigeria.
*Corresponding Author: Ezekiel Moses, Department of Research, Innovation and Technology Development, Federal College of Fisheries and Marine Technology, Victoria Island, Lagos, Nigeria.
Citation: Moses E., Mohammed M., Muftau S. (2025). Effect of Livolin Forte on Cognitive Impairment Caused by Sub-Chronic Lead Acetate Administration in Wistar Rats, Journal of BioMed Research and Reports, BioRes Scientia Publishers. 8(5):1-9. DOI: 10.59657/2837-4681.brs.25.204
Copyright: © 2025 Ezekiel Moses, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: September 06, 2025 | Accepted: September 20, 2025 | Published: September 27, 2025
Abstract
Several nutraceuticals have been used to alleviate the toxic effects of chemicals in living organisms. Lead acetate as one of the environmental pollutants causes threats to life of living creatures in many ways. The treatment of lead poisoning is classically by chelation therapy. However, there are documented reports of side effects accompanying the use of synthetic chelators, thus, necessitating the search for safer alternative treatments. Therefore, the aim of this study was to evaluate both pretreatment and post-treatment effects of Livolin Forte® on cognition following sub-chronic lead acetate administration in male Wistar rats. Thirty-six (36) male adult Wistar rats weighing 150 to 200 g were divided into six groups of six rats each using simple randomization. Group I rats were given 2 mL/kg body weight of distilled water, Group II rats were given 2 mL/kg of propylene glycol, Group III rats were given Livolin Forte® (5.2 mg/kg body weight) only, Group IV rats were given Lead acetate (Based on the reported oral LD50 of lead acetate which is 600 mg/kg body weight for Wistar rats (14), 1/10th of the LD50, i.e. 60 mg of Lead acetate per kg body weight), and in Group V rats, were dosed LF® (5.2 mg/kg) then Lead acetate (60 mg/kg) after 1hr (pretreatment). While Group VI rats were given Lead acetate (60 mg/kg) followed by Livolin forte® (5.2 mg/kg) after 1hr (post-treatment). The regimen was given orally once a day for seven weeks. During this period, the rats were monitored for signs of toxicity. At the end of the treatment period, the survived rats were taken for cognition studies. Significantly, (P ≤ 0.05) higher time it takes to step down in short-term memory evaluation (68.8 ± 15.5 sec) was recorded in lead acetate exposed group when compared to other groups. From the result, it was concluded that LF had no pretreatment and post-treatment effects on sub chronic lead acetate induced cognitive impairment in adult male Wistar rats.
Keywords: lead acetate; Sub-chronic; cognition; Livolin forte; toxicity
Introduction
Technological advancements and the necessity to meet man's basic requirements, a rising number of chemicals are being purposely released into the environment. These substances interact with one another and with the environment, resulting in negative health and environmental effects (Ravi et al., 2021). As a result of its vast industrial and residential applications, lead is the most common heavy metal pollutant (Patrick, 2006a). Lead (Pb) poisoning has a negative impact on both human and animal health. It is utilized in industry and in houses, and found in soil, where it poses numerous threats to humans and animals (US EPA, 2006). Known as the major polluter of the environment, and its remains have been found in children's toys, domestic kitchen products, watering cans, and pipelines (Ravi et al., 2021).
Plants are typically exposed to Pb through water absorption, while animals are exposed to it through the ingestion of plants carrying Pb residues (Hedayati and Darabitabar, 2017). It can also enter the body through drinking water and inhaling airborne polluted dust. Once absorbed, Pb rapidly diffuses through the bloodstream to numerous organs such as the brain, liver, and kidney, as well as highly calcified tissues such as teeth and bone (Alya et al., 2015).
Lead poisoning (also known as plumbism, Colica pictonum, saturnism, Devon colic, or painter's colic) is a medical illness caused by an excessive amount of lead in the body. Developing nations are particularly at high risk of lead poisoning and carry the highest burden of this hazard (Samuel et al., 2017). In Nigeria, a suspected case of Pb poisoning occurred at Unguwan Magiro and Unguwan Kawo communities in Rafi Local Government Area of Niger state in which 48 persons mostly children, (with BLL between 171.5- 224 µg/dL) including 14 deaths were reported in May15, 2015 (WHO, 2015). In March, 2010, Medecins Sans Frontieres, MSF/ doctors without Borders, an international, independent, medical humanitarian organization was alerted to a high number of child fatalities in Zamfara state, northern Nigeria, with an estimate of 400 children died. Laboratory testing confirmed high levels of lead in the blood of surviving children in that incidence. The root cause of the lead poisoning crisis was unsafe mining and ore processing (MSF, 2012).
One of the known major mechanisms of associated illnesses is oxidative damage caused by oxidative stress (Alya et al., 2015). Oxidative damage is caused by an imbalance between the synthesis and manifestation of reactive oxygen species (ROS) and the ability of a biological system to readily detoxify the reactive intermediates or repair the ensuing changes (El-Erain et al., 2015).
Livolin forte® (LF) is a relatively new medicine that is used in hospitals around the world to treat and control liver disease (Olaoluwa et al., 2014). It mostly comprises polyunsaturated phosphatidylcholine (300mg), vitamin B1 (thiamine, 10mg), vitamin B2 (riboflavin, 6mg), vitamin B6 (pyridoxine, 10mg), vitamin B12 (cyanocobalamin, 10g), nicotinamide (30mg), and vitamin E (alpha tocopherol, 10mg) (Olaoluwa et al., 2014).
Lead (Pb) is one of the metallic xenobiotics found as a contaminant in air, water, and food (Onyem et al., 2021). It is a toxin that affects almost all organs and has major detrimental effects on the neurological, renal, hepatic, reproductive, cardiovascular, and hematological systems (Patra et al., 2011). In Nigeria, an epidemic of lead poisoning has been documented, and sublethal exposure with chelating agents such as (dimercaptosuccinic) remains the most common therapy (Onyem et al., 2021).
Although chelation therapy is traditionally used to treat Pb poisoning, there is documented evidence of the side effects of synthetic chelators (Thuppil and Tannir, 2013). Commonly used chelators include dimercaptosuccinic acid (DMSA), calcium disodium versenate (CaNa2EDTA), dimercaprol (BAL), Unithiol (DMPS), and D- penicillamine (DPA). Ethylenediaminetetraacetic acid (EDTA) has been linked to renal toxicity and cardiac problems caused by hypocalcaemia; penicillamine has been linked to abdominal pain, skin lesions, alopecia, stomatitis, glossitis, leucopoenia, thrombocytopenia, and enuresis; succimer has been linked to nausea, vomiting, diarrhoea, and skin rash; and DMSA has been linked to nausea, vomiting (Thuppil and Tannir, 2013).
Despite substantial study on heavy metal toxicity, little or no attention has been made to the moderating effects of naturally occurring antioxidants and their potential nutritional values in humans and animals during heavy metal exposure.
Materials and Methods
Location of Experiment
The experiment was carried out at the Department of Veterinary Pharmacology and Toxicology, Faculty of Veterinary Medicine, Ahmadu Bello University, Zaria, Nigeria.
Experimental Animals
Thirty- six (36) adult male (150-200 g) Wistar strain Albino rats served as subjects. They were obtained from the Laboratory animal house of the Department of Veterinary Pharmacology and Toxicology. The rats were kept in the Department's animal holding facility. The Ahmadu Bello University committee on Animal Use and Care guidelines were strictly adhered to, in order to minimize unwanted stress or discomfort to the animals during experimental procedures by ensuring minimal handling. The rats were given free access to standard commercially prepared rat chow and water.
Ethical Clearance
The Ahmadu Bello University committee on Animal Use and Care provided ethical approval for the use of Wistar Rats in this study, with clearance approval number: ABUCAUC//2023/023
Chemical Acquisition and Preparation
Lead acetate was purchased from Sigma Aldrich (St. Louis, MO, USA). It was reconstituted in distilled water to make a stock solution. Livolin forte® (500 mg LF per capsule) was purchased from a reputable pharmaceutical store in Kaduna state, Nigeria. According to (Olaoluwa et al., 2014), one capsule of 500 mg was dissolved in 20 mL of propylene glycol to make a 25 mg/mL suspension daily before administration.
Animal Grouping
Thirty-six (36) male adult Wistar rats were divided into six groups of six rats each using simple randomization (Table 1). Group I rat were given 2 mL/kg body weight of distilled water, Group II rats were given 2 mL/kg of propylene glycol, Group III rats were given LF (5.2 mg/kg) only, Group IV rats were given Pb (Based on the reported oral LD50 of Pb which is 600 mg/kg body weight for Wistar rats (14), 1/10th of the LD50, i.e., 60 mg of Pb per kg body weight). Group V rats were given LF (5.2 mg/kg) followed by Pb (60 mg/kg) after 1hr. While, Group VI rats were given Pb (60 mg/kg) followed by LF (5.2 mg/kg) after 1hr. The regimens were administered by gavage, once daily for seven weeks. During this time, the rats were monitored for signs of toxicity.
Table 1: Dosing protocol showing various treatments in rats
| Group | Treatment | Dosage regimen |
| I | Distilled water | 2 ml/kg |
| II | Propylene glycol | 2 ml/kg |
| III | Livolin Forte® | 5.2 mg/kg |
| IV | Lead acetate | 60 mg/kg |
| V | Livolin Forte® then Lead acetate after 1hr | 5.2 mg/kg + 60 mg/kg |
| VI | Lead Acetate then Livolin Forte® after 1hr | 60 mg/kg + 5.2 mg/kg |
Evaluation of Neurobehavioral Changes
Evaluation of learning
The effect of lead on learning task in rats and the possible ameliorative effect of antioxidant vitamin (ascorbic acid) was assessed 48 hours to the termination of the study using the step-down inhibitory avoidance learning task as described by (Zhu et al., 2001). The apparatus used for the learning test was a 40 × 25 × 25 cm acrylic chamber, consisting of a floor made of parallel 2-mm calibre stainless steel bars spaced 1cm apart. An electric shock was administered through the floor bars. A 2.5-cm-high, 8 × 25 cm wooden platform was placed on the left extreme of the chamber. Each animal was gently placed on the platform. Upon stepping down, the rat immediately received a single 80-volt foot-shock. If the animal did not return to the platform, the foot-shock was repeated every 5 seconds. A rat was considered to have learned the avoidance task, if it remained on the platform for more than 2 minutes. The number of foot-shocks applied before the animal learned the avoidance task was recorded as an index of learning acquisition.
Evaluation of short-term memory
Short-term memory was assessed in individual rat from each group using the stepdown avoidance inhibitory task as described by (Zhu et al., 2001). The apparatus used for the memory test consisted of 40 × 25 × 25 cm acrylic chamber with floor made of parallel 2-mm-calibre stainless steel bars spaced 1 cm apart. A 2.5-cm-high, 8 × 25 cm wooden platform was placed on the left extreme of the chamber. In this case, the rat was again placed gently on the platform 24 hours after performing the learning task. The time an animal remained on the platform was recorded as an index of memory retention. Staying on the platform for 2 minutes was counted as maximum memory retention (ceiling response).
Data Analysis
Data obtained were expressed as Mean ± standard error of the mean (SEM) and subjected to one-way analysis of variance (ANOVA) followed by Tukey’s post-hoc multiple comparison test. Graph pad prism version 5, San Diego, California, USA (www.graphpad.com) was used to analyse all the data. Values of p ≤ 0.05 were considered significant.
Results
Sub-chronic Toxicological Studies
Clinical Signs
There were no toxicity signs in the control animals that were given distilled water alone and those that were given Propylene glycol alone, and the group that were dosed with Livolin forte® alone. However, toxicity signs such as spasms, weight loss, weakness, decrease in feed intake, and rough hair coat were observed in the Pb -treated animals, the pretreatment group as well as the post-treatment group.
Effect of Livolin forte® and Lead acetate on Cognition
Effect of livolin forte and lead acetate on learning
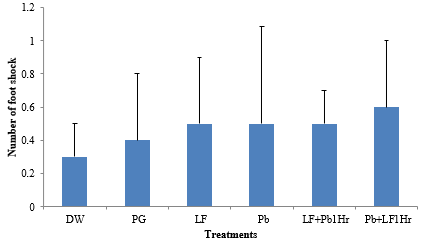
There was no significant difference (P ≥ 0.05) in learning test across the groups (Figure 1).
Effect of livolin forte® and lead acetate on short-term memory
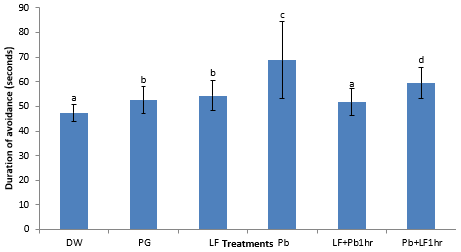
There was a significant increased (P ≤ 0.05) time it takes to step down in the lead acetate exposed rats (68.8 ± 15.5 sec) when compared to the other groups DW (47.4 ± 3.4 sec), PG (52.6 ± 5.6 sec), LF (54.4 ± 6.1 sec), LF+Pb1hr (51.8 ± 5.3 sec), and Pb+LF1hr (59.4 ± 6.3 sec) (Figure 2).
Figure 1: Effect of sub-chronic administration of LF and Pb on learning. Values are presented as Mean±SEM, (P˃0.05), n=6.
Key: DW: Distilled Water (2 ml/kg); Pg: Propylene glycol (2 ml/kg); Pb: Lead acetate (60 mg/kg); LF: Livolin forte® (5.2 mg/kg); LF+Pb1Hr: Livolin forte® (5.2 mg/kg) then Lead acetate (60 mg/kg) after 1hr, Pb+LF1Hr: Lead acetate (60 mg/kg) then Livolin forte® (5.2 mg/kg) after 1hr.
Figure 2: Effect of sub- chronic administration of LF and Pb on memory. Values are presented as Mean ± SEM. Values with different letters (a, b, c, d) are significantly different (P ≤ 0.05), n = 6.
Key: DW: Distilled Water (2 ml/kg); Pg: Propylene glycol (2 ml/kg); Pb: Lead acetate (60 mg/kg); LF: Livolin forte® (5.2 mg/kg); LF+Pb1Hr: Livolin forte® (5.2 mg/kg) then Lead acetate (60 mg/kg) after 1hr, Pb+LF1Hr: Lead acetate (60 mg/kg) then Livolin forte® (5.2 mg/kg) after 1hr.
Discussion
Lead is a common heavy metal that pollutes the environment and accumulates in human and animal body through absorption, bioavailability, bioconcentration, and biomagnification disrupts the neurological, skeletal, reproductive, haematopoietic, renal, and cardiovascular systems (Collin et al., 2022). The results of the present study showed that sub-chronic exposure of Wistar rats to lead acetate had no significant effect on learning in rats. The group exposed to Pb had an increase in learning, when compared to the other groups. It has been shown that Pb in toxic quantity have no significant effect on learning in rats (Susan et al., 2013). This could be the reason why the learning in the Pb treated group was not affected.
The current results on short-term memory demonstrated a decrease in the cognitive function of rats exposed to repeated low-dose lead, indicating a reduction in the index of short-term memory retention of the experimental animals. Several studies have demonstrated the effect of lead on short-term memory in animals (Aldridge et al., 2005; Haider et al., 2005; Azzaoui et al., 2009). The exposed rats spent less time on the platform in the step-down avoidance inhibitory apparatus, compared to rats in other treatment groups. This fact, apparently, showed that lead impairs cognitive function in rats. Several mechanisms of altered cognitive function following lead exposure have been proposed. One is the alterations in glutamate release from the hippocampus (Wisden et al., 2000). The hippocampus is a brain area which is rich in glutamate receptors. Appropriate glutamate activity is critical for the development of the central nervous system, including the ontogeny of learning and memory (Adams et al., 2009). The effects of glutamate are often mediated through the N-methyl-D-aspartate (NMDA), alpha-amino-3-hydroxy-5-methyl-4- isoxazole-4-propionic acid (AMPA) and kainate receptors (Frerking et al., 2001; Bortolotto et al., 2005; Adams et al., 2009). Infact, NMDA receptors are important in their role in long-term potentiation (Rison and Stanton, 1995; MacDonald et al., 2006), which is a form of synaptic plasticity often used as a model system for the study of cognitive functions (Porterfield and Hendrick, 1993; Laroche et al., 2000; O’Mara et al., 2000). The memory process is based on the creation and remodeling of synapses and the toxic effect of lead on 112 this process suggests that lead specifically damages synaptic functions (Bradbury and Deane, 1993; Lasley and Gilbert, 2000; Bouton et al., 2001). Damage to the hippocampus has been reported to cause severe impairments in memory (Eichenbaum et al., 1990; McDonald and White, 1995; Grigorenko et al., 1997), but this damage was found not to affect spatial information (Gilbert et al., 2005). Another mechanism is via decrements in the activities of acetylcholinesterase. Sub chronic exposure to lead has resulted in a decrease in activity of acetylcholinesterase in rats (Nehru and Sidhu, 2002; Reddy et al., 2003; Saxena and Flora, 2004). Similarly, the central cholinergic system has been linked to synaptic plasticity and memory processes in several studies (Sachdev et al., 1998; Reddy et al., 2007). A decrease in brain serotonin (5- hydroxytryptamine) levels has also been implicated as a cause of impaired cognitive function in lead-treated rats (Haider et al., 2005). In addition, rat brain neurotoxicity has been demonstrated to occur via the induction of oxidative stress as a result of the accumulation of delta-aminolevulinic acid during lead exposure (Tandon et al., 2002; Patrick, 2006b; Wang et al., 2006). The result of the present study demonstrated a beneficial effect of pre-treatment and post-treatment with Lf, as short-term memory of lead-exposed rats was slightly improved. In addition, agents that inhibit generation of reactive oxygen species in the brain may block tissue damage resulting from excitotoxicity from glutamate and loss of cellular calcium (Bose et al., 1992; Siesjo, 1993). This mechanism involved in the pathogenesis of neuronal damage resulting in impaired cognition, further confirms the beneficial role of Lf in improving memory in rats.
The World Health Organization (WHO) report of 31st August, 2022, stated that the neurological and behavioural effects of lead are believed to be irreversible (WHO, 2022). This may be due to the inhibition of δ-ALAD in the plasma. Excess of δ-ALA (gamma aminolevulinic acid) causes severe neurological effects (Dehari-Zeka et al., 2020). This could be the reason why Lf could not modulate the toxic effect of lead on memory. Exposure to lead element, and thus its toxicity, has increased around the world as industries have developed. The central nervous system is the most vulnerable to lead intoxication. Lead-induced neurotoxicity (Saeedeh et al., 2016) results in behavioral, morphological, and electrophysiological changes (Sharfi et al., 2002).
The current study found that continuous lead exposure had a negative impact on rat memory. The findings are consistent with experimental and clinical research that has shown that lead exposure disrupts cognitive functioning and damages processes in humans and animals (Finkelstein et al., 1998). A number of mechanisms have been proposed to explain lead neurotoxicity. The increase of reactive oxygen species triggers lipid peroxidation and oxidative stress processes, which is one of the most critical pathways in lead-induced neurotoxicity (Kuhlmann et al., 1997). Furthermore, lead binds to the thiol groups of biological macromolecules such as glutathione, reducing their reductive potency and antioxidant activity (Marchetti, 2003). Other pathways involved in lead neurotoxicity have been documented to include the activation of the apoptotic cascade, an increase in inflammatory mediators, changes in glutamate-induced neuroplasticity, and abnormalities in calcium homeostasis (Xu et al., 2006). Many vitamins have been discovered to help reduce the neurotoxic consequences of lead exposure. One of the most critical supplements required for the regular functioning of neurons in the neurological system is vitamin B12, which is included in Lf. As a cofactor, it regulates CNS enzyme activity and ensures proper metabolic function (Saeedeh et al., 2016).
The higher number of steps down in Pb-exposed rats in this study implies that Pb induces memory loss, which is consistent with the work of (Daniela et al., 2021). This reduction in cognitive ability could be due to oxidative damage to brain tissue, particularly the hippocampus, which is important for learning and memory (Nathaniel and Gabriel, 2020).
During the development of industries in the world, exposure to lead element and consequently its toxicity has been increased worldwide. Central nervous system is the most important target for the lead toxicity. Lead induced neurotoxicity include behavioral, morphological, and electrophysiological disruptions (Sharfi et al., 2002).
Several mechanisms have been proposed to explain the neurotoxicity of pb2+. One of the most important mechanisms in lead-induced neurotoxicity is the triggering of lipid peroxidation and oxidative stress processes by accumulation of reactive oxygen species [58]. In addition, lead binds to the thiol groups of biologic macromolecules like glutathione and decreases their reductive potencies and antioxidant activities (Marchetti, 2003). It has been reported that the initiating of apoptosis cascade, increasing inflammatory mediators, alteration in glutamate-induced neuroplasticity and changes in calcium homeostasis are some of other mechanisms involved in lead neurotoxicity [60].
One of the most important disorders following lead exposure is neurotoxicity [55]. Neurotoxicity is a popular disorder from lead exposure which involves both central and peripheral nervous system. Lead induced neurotoxicity triggers a wide range of structural and behavioral changes in the nervous system [56].
Conclusion
In summary, the present study shows that livolin forte had both pretreatment and post-treatment effects on memory impairment induced by lead element.
This study supports the role of oxidative stress in the pathophysiology of low-level lead poisoning. The beneficial effects of LF were not observed on cognition in rats.
Declarations
Authors’ Contributions
E.M. conceived and planned the experiments. E.M. carried out the experiments. E.M. planned and carried out the simulations. E.M. contributed to sample preparation. M.M. and S.M. contributed to the interpretation of the results. E.M. took the lead in writing the manuscript. All authors provided critical feedback and helped shape the research, analysis and manuscript.
Acknowledgements
We would like to thank Mr. Dennis Otie of the Toxicology lab, Department of Veterinary Pharmacology and Toxicology, Faculty of Veterinary Medicine, Ahmadu Bello University, Zaria, Nigeria.
Competing Interests
The authors declare that there is no conflict of interest.
References
- Adams, A.L., Doucette, T.A., James, R. and Ryan, C.L. (2009). Persistent changes in learning and memory in rats following neonatal treatment with domoic acid. Physiology and Behaviour, 96 (4-5):505-512.
Publisher | Google Scholor - Aldridge, J.E., Levin, E.D., Seidler, F.J. and Slotkin, T.A. (2005). Developmental exposure of rats to chlorpyrifos leads to behavioural alterations in adulthood, involving serotonergic mechanisms and resembling animal models of depression, 113(5):527-531.
Publisher | Google Scholor - Alya, A., Ines, B.D., Montassar, L., Najoua, G.H. and Saloua, E.F. (2015). Oxidative stress, biochemical alterations and hyperlipidemia in female rats induced by lead chronic toxicity during puberty and post puberty periods. IJBMS; 18:1034-1043.
Publisher | Google Scholor - Azzaoui, F.Z., Ahami, A.O.T. and Khadmaoui, A. (2009). Impact of lead sub-chronic toxicity on recognition memory and motor activity of Wistar rats. Pakistan Journal of Biological Sciences, 12 (2):173-177.
Publisher | Google Scholor - Bortolotto, Z.A., Nistico, R., More, J.C., Jane, D.E. and Collingridge, G.L. (2005). Kainate receptors and mossy fibre LTP. Neurotoxicology, 26:769-777.
Publisher | Google Scholor - Bose, R., Schnell, C. L., Pinsky, C. and Zitko, V. (1992). Effects of excitotoxins on free radical indices in mouse brain. Toxicology Letters, 60:211-219.
Publisher | Google Scholor - Bouton, C.M., Freline, C.P., Forde, C.E., Godwin, H.A. and Pevsner, J. (2001). Synaptotagmin 1 is a molecular target for lead. Journal of Neurochemistry, 7:1724-1735.
Publisher | Google Scholor - Bradbury, M.W. and Deane, R. (1993). Permeability of the blood-brain barrier to lead. Neurotoxicology, 14:131-136.
Publisher | Google Scholor - Collin, M.S., Senthil, K.V., Naveensubramaniam, V. Kanimozhi, V., Arbaaz, S.M., et al., (2022). Bioaccumulation of lead (Pb) and its effects on human: A review. Journal of Hazardous Materials Advances, 7:100094.
Publisher | Google Scholor - Daniela, R.O., Dinora, F.G.E., Tonali, B.A., Benjamin, P., Saul, G.M., et al. (2021). Cognitive Impairment induced by lead exposure during lifespan: Mechanism of lead Neurotoxicity. Toxics, 2(9): 23.
Publisher | Google Scholor - Dehari-Zeka, M., Letaj, K.R., Selimi, Q., Elezaj, I. (2020). Blood lead level (BLL), δ- aminolevulinic acid dehydratase activity (ALAD), haemoglobin (Hb) and haematocrit (hct) in primary school- children and adult residents living in smelter rural areas in Kosovo. Journal of Environmental Science and Health Part A/ Hazardous Substances and Environmental Engineering, 55(10):1-9.
Publisher | Google Scholor - Eichenbaum, H., Stewart, C. and Morris, R.G. (1990). Hippocampal representation in place learning. Journal of Neuroscience, 10:3531-3542.
Publisher | Google Scholor - El-Erain, A., Anis, M. and Ibrahim, H. (2015). Oxidative stress markers as early predictors of neuropathy in type 2 diabetic patients. Nature and Sciences, 13(1):120-126.
Publisher | Google Scholor - Finkelstein, Y., Markowitz, M.E. and Rosen, J.F. (1998). Low- level lead- induced neurotoxicity in children: an update on central nervous system effects. Brain Research Reviews, 27(2):168-176.
Publisher | Google Scholor - Frerking, M., Schmitz, D., Zhou, Q., Johansen, J. and Nicoll, R.A. (2001). Kainate receptors depress excitatory synaptic transmission at CA3 – CA1 synapses in the hippocampus via a direct presynaptic action. Journal of Neuroscience, 21:2958-2966.
Publisher | Google Scholor - Gilbert, M.E., Kelly, M.E., Samsam, T.E. and Goodman, J.H. (2005). Chronic developmental lead exposure reduces neurogenesis in adult rat hippocampus but does not impair spatial learning. Toxicological Sciences, 86(2):365-374.
Publisher | Google Scholor - Grigorenko, E., Glazier, S., Bell, W., Tytell, M., Nosel, E., Pons, T. and Deadwyler, S.A. (1997). Changes in glutamate receptor subunit composition in hippocampus and cortex in patients with refractory epilepsy. Journal of the Neurological Sciences, 153(1):35-45.
Publisher | Google Scholor - Haider, S., Saima, S., Shahida, P.A., Tahira, P. and Darakhshan, J.H. (2005). Repeated administration of lead decreases brain 5-HT metabolism and produces memory deficits in rats. Cellular and Molecular Biology Letters, 10:669-676.
Publisher | Google Scholor - Haider, S., Saima, S., Shahida, P.A., Tahira, P. and Darakhshan, J.H. (2005). Repeated administration of lead decreases brain 5-HT metabolism and produces memory deficits in rats. Cellular and Molecular Biology Letters, 10:669-676.
Publisher | Google Scholor - Hedayati, A. and Darabitabar, F. (2017). Lethal and sub-lethal impacts of lead on some haematological, biochemical and immunological indices in Caspian roach. Pollution, 3(1):21-27.
Publisher | Google Scholor - Kuhlmann, A.C., McGlothan, J.L. and Guilarte, T.R. (1997). Developmental lead exposure causes spatial learning deficits in adult rats. Neuroscience letters, 233(2-3):101-104.
Publisher | Google Scholor - Laroche, S., Davis, S. and Jay, T.M. (2000). Plasticity at hippocampal to pre-frontal cortex synapses: dual roles in working memory and consolidation. Hippocampus, 10(4):438-446.
Publisher | Google Scholor - Lasley, S.M. and Gilbert, M.E. (2000). Glutamatergic components underlying lead-induced impairments in hippocampal synaptic plasticity. Neurotoxicology, 21(6):1057-1068.
Publisher | Google Scholor - MacDonald, J.F., Jackson, M.F. and Beazely, M.A. (2006). Hippocampal long-term synaptic plasticity and signal amplication of NMDA receptors. Critical Reviews in Neurobiology, 18(1-2):71-84.
Publisher | Google Scholor - Marchetti, C. (2003). Molecular targets of lead in brain neurotoxicity. Neurotoxicity Research, 5(3):221-236.
Publisher | Google Scholor - McDonald, R.J. and White, N.M. (1995). Hippocampal and non-hippocampal contributions to place learning in rats. Behavioural Neuroscience, 109:579-593.
Publisher | Google Scholor - Medecins Sans Frontieres [MSF] (2012). Lead poisoning crises in Zamfara State, Northern Nigeria, in Proceedings of the International Lead poisoning Conference Abuja.
Publisher | Google Scholor - Nathaniel, O.A. and Gabriel, O.O. (2020). Lead acetate- induced neurodegenerative changes in the dorsolateral prefrontal cortex of mice: the role of vitexin. Environmental Analysis Health and Toxicology, 35(1):e2020001.
Publisher | Google Scholor - Nehru, B. and Sidhu, P. (2002). Neurotoxic effects of differential doses of lead on rat brain followed by recovery. The Journal of Trace Elements in Experimental Medicine, 15(3):131-140.
Publisher | Google Scholor - O’Mara, S.M., Commins, S. and Anderson, M. (2000). Synaptic plasticity in the hippocampal area CA1-subiculum projection: implications for theories of memory. Hippocampus, 10(4):447-456.
Publisher | Google Scholor - Olaoluwa, S.O., Rufus, O.A., Kayode, D.B., Akande, O.A., Raphael, E.O., Ronald, A.B. (2014). Protective and Currative effects of Livolin forte® on carbon tetrachloride-induced liver damage in Wistar Rats. Journal of Exp Integr Med, 4(1):57-65.
Publisher | Google Scholor - Onyem, J.C., Ozoko, L.E.C., Enaohwo, M.T., Otabor, F.O., Okudayo, V.A., Udi, O.A. (2021). Antioxidative properties of Ocimum gratissimum alters Lead acetate induced oxidative damage in lymphoid tissues and hematological parameters of adult Wistar. Toxicol, Rep. 8:215-222.
Publisher | Google Scholor - Patra, R.C., Rautray, A.K. and Swarup, D. (2011). Oxidative stress in lead and cadmium toxicity and its amelioration. Vet. Med. Int., 22(4):1-9.
Publisher | Google Scholor - Patrick, L. (2006). Lead Toxicity Part II: The Role of free Radical Damage and the use of Antioxidants in the Pathology and treatment of Lead Toxicity. Altern Med Rev, 11:114-127.
Publisher | Google Scholor - Patrick, L. (2006). Lead toxicity, a review of the literature. Part 1: Exposure, evaluation, and treatment. Alteration Medicine Review, 11:2-22.
Publisher | Google Scholor - Porterfield, S.P. and Hendrich, C.E. (1993). The role of thyroid hormones in prenatal and neonatal neurological development – current perspectives. Endocrine Reviews, 14(1):94-106.
Publisher | Google Scholor - Ravi, N., Bhabananda, B., Lan, R.W., Julian, C., Brajesh, K.S., Pau, C.N., Frederic, C., Kirk, T.S., Kevin, C.J., Adam, B., Robert, J.A. (2021). Chemical Pollution: A growing peril and potential catastrophic risk to humanity. Journal of Environment international, 156:106616.
Publisher | Google Scholor - Reddy, G. R., Devi, B. C. and Chetty, C. S. (2007). Developmental lead neurotoxicity: alterations in brain cholinergic system. Neurotoxicology, 28(2):402-407
Publisher | Google Scholor - Reddy, G.R., Basha, M.R., Devi, C.B., Suresh, A., Baker, J.L., et al. (2003). Lead-induced effects on acetylcholinesterase activity in cerebellum and hippocampus of developing rat. International Journal of Development Neuroscience, 21(6):347-352.
Publisher | Google Scholor - Rison, R.A., and Stanton, P.K. (1995). Long-term potentiation and N-methyl-D-aspartate receptors: foundations of memory and neurologic disease? Neuroscience and Biobehavioural Reviews, 19:533-552.
Publisher | Google Scholor - Sachdev, R.N., Lu, S.M., Wiley, R.G. and Ebner, F.F. (1998). Role of the basal forebrain cholinergic projection in somatosensory cortical plasticity. Journal of Neurophysiology, 79(6):3216-3228.
Publisher | Google Scholor - Saeedeh, A.M., Mohammed, R., Mohammed, S. and Ali, H. (2016). Protective effect of Vitamin C, Vitamin B12 and Omega- 3 on lead- induced memory impairment in rat. Research in Pharmaceutical Science, 11(5):390-396.
Publisher | Google Scholor - Samuel, J.O., Herbert, O.C.M. and Orish, E.O. (2017). Lead induced hepato-renal damage in male albino rats and effects of activated charcoal. Frontiers in Pharmacology, 8:107.
Publisher | Google Scholor - Saxena, G. and Flora, S.J.S. (2004). Lead-induced oxidative stress and haematological alterations and their response to combined administration of calcium disodium EDTA with a thiol chelator in rats. Journal of Biochemical and Molecular Toxicology, 18:221-223.
Publisher | Google Scholor - Sharfi, A.M., Baniasadi, S., Jorjani, M., Rahimi, F., Bakhshayesh, M. (2002). Investigation of acute lead poisoning on apoptosis in rat hippocampus in vivo. Neuroscience letters, 329(1):45-48.
Publisher | Google Scholor - Siesjo, B.K. (1993). Basic mechanisms of traumatic brain damage. Annals of Emergency Medicine, 22:959-969.
Publisher | Google Scholor - Sujatha, K., Srilatha, C., Anjaneyulu, Y. and Amaravathi, P. (2011). Lead acetate induced neurotoxicity in Wistar albino rats: A pathological, immunological, and ultrastructural studies. The Journal of Pharmaceutical and Biological Science, 2:459-462.
Publisher | Google Scholor - Susan, B., Nicholas, D. and Wolgarg, H.V. (2013). Effect of Lead acetate on learning and Memory in rats. Archives of Environmental Health: An International Journal, 22(3):370-372.
Publisher | Google Scholor - Tandon, S.K., Singh, S., Prasad, S., Srivastava, S. and Siddiqui, M.K.J. (2002). Reversal of lead-induced oxidative stress by chelating agent, antioxidant, or their combination in the rat. Environmental Research, 90:61-66
Publisher | Google Scholor - Thuppil, V. and Tannir, S. (2013). Treating lead toxicity: possibilities beyond synthetic chelation. JKIMSU, 2(1):4-31.
Publisher | Google Scholor - United States Environmental Protection Agency (US EPA). (2006). Evaluation Report. Opportunities to improve data Quality and Children’s Health through food Quality Protection Act. Office of Inspector General Report no. - p- 009.
Publisher | Google Scholor - Wang, J., Wu, J. and Zhang, Z. (2006). Oxidative stress in mouse brain exposed to lead. Annals of Occupational Hygiene, 50(4):405-409.
Publisher | Google Scholor - Wisden, W., Seeburg, P.H. and Monyer, H. (2000). AMPA, Kainate and NMDA ionotropic glutamate receptor expression: an in-situ hybridization atlas. In: Otterson, O. P. and Storm-Mathisen, J. (Ed.). Handbook of Chemical Neuroanatomy, New York: Elsevier; 18:99-143.
Publisher | Google Scholor - World Health Organization (WHO). (2015). Lead poisoning in Nigeria. Geneva: World Health Organization.
Publisher | Google Scholor - World Health Organization (WHO). (2022). Lead Poisoning.
Publisher | Google Scholor - Xu, J., Ji, L.D. and Xu, L.H. (2006). Lead – induced apoptosis in pc12 cells: involvement of ps3, Bcl- 2 family and caspage- 3. Toxicology letters, 166(2):160-167.
Publisher | Google Scholor - Zhu, H., Robin, W., Rockhold, R.W., Baker, R.C., Kramer, R.E. and Ho, I.K. (2001). Effects of single or repeated dermal exposure to methyl parathion on behaviour and blood cholinesterase activity in rats. Journal of Biomedical Science, 8:467-474.
Publisher | Google Scholor