Case Report
Chylothorax Secondary to Congestive Heart Failure: A Rare Case Report
- Sandeep Nain *
SGT medical college and hospital, Gurugram, India.
*Corresponding Author: Sandeep Nain, SGT medical college and hospital, Gurugram, India.
Citation: Nain S. (2026). Chylothorax Secondary to Congestive Heart Failure: A Rare Case Report, International Clinical and Medical Case Reports, BioRes Scientia Publishers. 5(1):1-4. DOI: 10.59657/2837-5998.brs.26.057
Copyright: © 2026 Sandeep Nain, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: September 18, 2025 | Accepted: January 02, 2026 | Published: January 09, 2026
Abstract
Chylothorax involves the buildup of chyle-a cloudy lymphatic fluid-in the space around the lungs. Typical triggers include injuries, issues from operations, cancers like lymphoma, infections such as tuberculosis, or birth defects.
Keywords: heart failure; fever; surgery
Introduction: Background
Here, we describe an uncommon instance of chylothorax linked to heart failure. Excessive chyle in this area may cause breathing difficulties, malnutrition from fat depletion, and weakened immunity due to antibody loss in the fluid. Identification typically relies on scans and testing of the pleural fluid to verify the presence of chyle.
Case Report
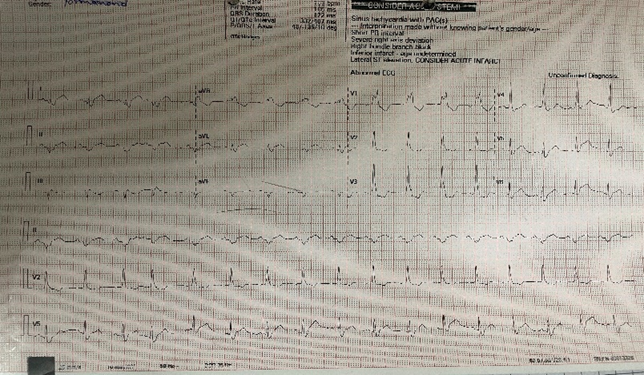
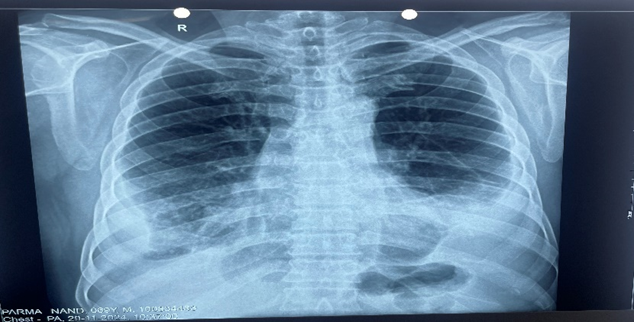
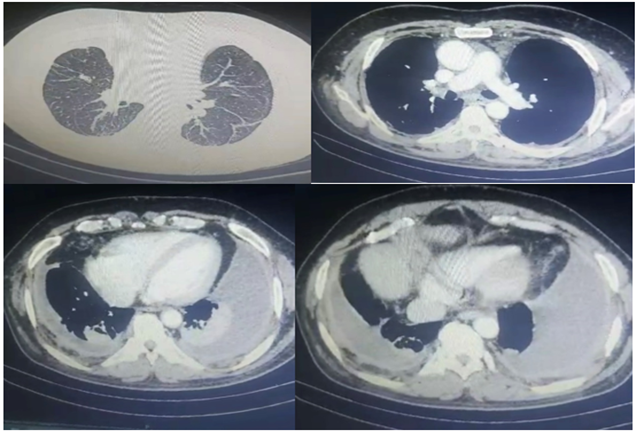
A 69-year-old male came to our emergency with complaints of breathlessness for 3 months which was insidious onset and progressive. He had chest tightness, and cough with whitish mucoid sputum for 1 month. He initially had chest pain 3 months back but it decreased gradually. There was no history of fever, He had a history of pulmonary tuberculosis 40 years back for which he took 9 months of antitubercular therapy. He was a smoker but did not consume alcohol. He was hypertensive for 10 years and was nondiabetic. There was no history of trauma, surgery, or any other interventions in the past. On admission, his blood pressure was 134/90 mm Hg, pulse 102/min, oxygen saturation 88% on room air, and respiratory rate 32/min. There was no pallor, icterus, cyanosis, clubbing, pedal edema, or lymphadenopathy. On inspection, there were decreased chest movements in infra-axillary and mammary areas. On percussion stony dull sound was noticed in the infrascapular, and infra-axillay regions bilaterally. On auscultation, there were decreased air entry in the infrascapular regions bilaterally and S3 gallop prominent Electrocardiogram (ECG) was suggestive of right bundle branch block pattern. Trop T was negative. Chest radio-graph showed blunting of bilateral costophrenic angles. Blood investigations were as follows: hemoglobin 13.2 gm/dl, leucocytes 8110/mm3, platelets 4.24 lakh/mm3, blood urea 31.4 mg/dl, creatinine 0.91 mg/dl, bilirubin 0.5 mg/dl, AST 43 IU/L, ALT 56 IU/L, total protein 5.6 gm/dl4, LDL 98 mg/dl, HDL 29 mg/dl, Triglyceride 125 mg/dl, VLDL 25 mg/dl, blood glucose 138 mg/dl. The urine examination was within normal limits. The patient's abdominal ultrasound was normal. Patients' viral markers HIV, HBsAg, and HCV was negative. Sputum Ziehl neelsen stain and gene xpert were negative for tuberculosis. Thoracic ultrasound revealed bilateral pleural effusion. Under ultrasound guidance, thoracocentesis was done. Pleural fluid was milky in color and its biochemical pathological and microbiological reports were as follows pleural fluid protein 4.06 gm/dl, glucose 137mg/dl, ADA 12.6 IU/L, LDH 806 mg/dl, triglyceride 381 mg/dl, cholesterol 51 mg/dl, gene xpert of pleural fluid was negative for mycobacterium tuberculosis. The culture was sterile. The total leucocyte count was 1000 of which 95%were lymphocytes and 5% neutrophils cytology was negative for malignant cells and no fungal element was seen. Thus, a diagnosis of chylothorax was made. Contrast-enhanced computer tomography was suggestive of a patchy area of consolidation/collapse with few specs of calcifications within and adjacent thin atelectatic bands seen in basal segments of bilateral lower lobes. There was no mediastinal lymphadenopathy. Bilateral pleural effusion seen with underlying atelectatic changes. 2D Electrocardiography revealed dilated cardiac chambers, severe left ventricles dysfunction with ejection fraction of 25-30%. Thus, after ruling out other possible causes of chylothorax, we made a diagnosis of chylothorax due to congestive heart failure which is a rare entity. After starting the treatment for congestive heart failure, patient showed significant improvement symptomatically and Chest radiograph also showed decreased amount of pleural effusion. He was discharged on stable vitals with oxygen saturation 95% on room air.
Figure 1: ECG.
Figure 2: Chest x-ray.
Figure 3: Contrast Enhanced Computer Tomography of chest.
Discussion
Chylothorax refers to the buildup of chyle within the pleural space [1]. Chyle is a cloudy liquid produced in the small intestine's lacteal network, known for its natural antibacterial properties and contents like antibodies and vitamins that dissolve in fat [2-4].
This fluid consists of lymph mixed with broken-down fats and free fatty acids. Key elements include proteins, fats (mostly triglycerides): immune cells such as lymphocytes, elevated levels of fat-soluble vitamins, and protective immunoglobulins [5,6]. Short- and medium-length triglycerides from food are digested by gut enzymes into free fatty acids, which enter the bloodstream via the portal vein. In contrast, longer-chain triglycerides resist breakdown by these enzymes and instead bond with phospholipids and cholesterol to create cholesterol esters, aiding in the assembly of chylomicrons in the jejunum [7,8].
The thoracic duct in adults’ forms from the fusion of the lower two-thirds of the embryonic right duct, the upper third of the left duct, and linking pathways [9]. As the body's primary lymphatic channel, it handles about 75% of lymph drainage from the left side and the right side below the diaphragm. These larger particles are taken up by the intestinal lymphatics to produce chyle. Intestinal lymph then combines with drainage from the legs to create the thoracic duct network, which empties into the general bloodstream. However, if the lymphatic vessels are damaged, the fatty chyle can escape into nearby areas [10-12].
Chylothorax arises when chyle enters the pleural space due to thoracic duct injury. Typically, around 2.4 liters of chyle are produced daily, which can lead to significant buildup in the chest. Though rare, it represents roughly 3% of pleural effusions overall. About 83% of instances are one-sided (50% on the right, 33% on the left): with 17 percentage affecting both sides [15].
Conclusion
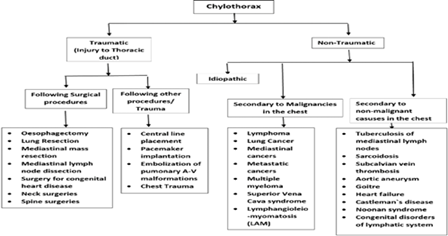
Pleural effusions can stem from numerous causes, so a thorough list of possible alternatives is essential for accurate identification. Distinguishing these is key to confirming chylothorax and initiating suitable management. Common differentials include pseudochylothorax, infection-related effusions like parapneumonic or empyema, other exudative collections, blood in the pleural space (hemothorax): heart failure buildup, effusions linked to HIV/AIDS, and those from cancer.
References
- Sassoon, C. S., & Light, R. W. (1985). Chylothorax and pseudochylothorax. Clinics in Chest Medicine, 6(1):163–171.
Publisher | Google Scholor - Smoke, A., & DeLegge, M. H. (2008). Chyle leaks: Consensus on management? Nutrition in Clinical Practice, 23(5):529–532.
Publisher | Google Scholor - Suslu, N., et al. (2014). The role of somatostatin treatment in the management of chylous fistula after neck dissection. Turkish Archives of Otolaryngology.
Publisher | Google Scholor - Jain, A., et al. (2015). A prospective study on the role of octreotide in management of chyle fistula. American Journal of Laryngology.
Publisher | Google Scholor - Zilversmit, D. B. (1965). The composition and structure of lymph chylomicrons in dog, rat, and man. Journal of Clinical Investigation, 44(10):1610–1622.
Publisher | Google Scholor - Staats, B. A., Ellefson, R. D., Budahn, L. L., Dines, D. E., Prakash, U. B., & Offord, K. (1980). The lipoprotein profile of chylous and nonchylous pleural effusions. Mayo Clinic Proceedings, 55(11):700–704.
Publisher | Google Scholor - Ur Rehman, K., & Sivakumar, P. (2022). Non-traumatic chylothorax: Diagnostic and therapeutic strategies. Breathe (Sheffield):18(1):210163.
Publisher | Google Scholor - Zilversmit, D. B. (1965). The composition and structure of lymph chylomicrons in dog, rat, and man. Journal of Clinical Investigation, 44(10):1610–1622.
Publisher | Google Scholor - Sabin, F. (1916). The origin and development of the lymphatic system. The Johns Hopkins Hospital Reports, 17:347–440.
Publisher | Google Scholor - Yu, D.-X., Ma, X.-X., Zhang, X.-M., Wang, Q., & Li, C.-F. (2010). Morphological features and clinical feasibility of thoracic duct: Detection with nonenhanced magnetic resonance imaging at 3.0 T. Journal of Magnetic Resonance Imaging, 32(1):94–100.
Publisher | Google Scholor - Phang, K. L., Bowman, M., Phillips, A., & Windsor, J. (2014). Review of thoracic duct anatomical variations and clinical implications. Clinical Anatomy, 27(4):637–644.
Publisher | Google Scholor - Nair, S. K., Petko, M., & Hayward, M. P. (2007). Aetiology and management of chylothorax in adults. European Journal of Cardio-Thoracic Surgery, 32(3):362–369. Pan African Medical Journal, 40:209.
Publisher | Google Scholor - Cholet, C., Delalandre, C., Monnier-Cholley, L., et al. (2020). Nontraumatic chylothorax: Nonenhanced MR lymphography. Radiographics, 40(6):1554–1573.
Publisher | Google Scholor - Bessone, L. N., Ferguson, T. B., & Burford, T. H. (1971). Chylothorax. Annals of Thoracic Surgery, 12(6):527–550.
Publisher | Google Scholor - McGrath, E. E., Blades, Z., & Anderson, P. B. (2010). Chylothorax: Aetiology, diagnosis and therapeutic options. Respiratory Medicine, 104(1):1–8.
Publisher | Google Scholor