Research Article
Biomedical Engineering (BME) in the Epoch of AI: A Systematic Investigation into Technological Transformation
1Department of Computing, Institute of Electrical and Electronics Engineers, United States of America.
2Department of Computer Science, American International University-Bangladesh, Bangladesh.
*Corresponding Author: Zarif Bin Akhtar, Department of Computing, Institute of Electrical and Electronics Engineers, United States of America.
Citation: Akhtar ZB, Rawol AT. (2025). Biomedical Engineering (BME) in the Epoch of AI: A Systematic Investigation into Technological Transformation, International Journal of Biomedical and Clinical Research, BioRes Scientia Publishers. 5(1):1-18. DOI: 10.59657/2997-6103.brs.25.105
Copyright: © 2025 Zarif Bin Akhtar, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: November 13, 2025 | Accepted: December 15, 2025 | Published: December 23, 2025
Abstract
In today’s rapidly advancing technological environment, continuous developments in computational sciences have driven substantial progress across multiple branches of biomedical engineering (BME), health informatics, and medical science. Emerging technologies such as computer vision, image processing, machine learning, deep learning, and data science have collectively revolutionized healthcare by enabling more precise analysis of medical data and enhancing the understanding of human anatomy. These innovations not only strengthen disease diagnosis, prevention, and treatment strategies but also address complex challenges in medical imaging and anatomical modeling. This study examines the influence of advanced computing methodologies on modern biomedical engineering practices, emphasizing their integration with bioinformatics, biomedical mechanics, and artificial intelligence (AI). Furthermore, it explores the growing role of functional genomics in uncovering novel insights into disease mechanisms and future health trends, underscoring the transformative potential of computational intelligence in shaping next-generation healthcare solutions.
Keywords: artificial intelligence; biomedical engineering; bioinformatics; deep learning; functional genomics; healthcare informatics; machine learning; medical informatics
Introduction
Biomedical Engineering (BME) represents a rapidly evolving discipline situated at the intersection of engineering, medicine, and biological sciences. It applies engineering design principles to address complex medical and healthcare challenges, integrating technological innovation with human health needs [1-3]. Once viewed primarily as an interdisciplinary specialization, BME has now matured into a well-established field that bridges fundamental science with clinical application.
The scope of biomedical engineering spans an extensive range of research and practical domains, including diagnostics, therapeutic systems, medical device design, and hospital equipment management [4-6]. Through collaborative efforts among engineers, clinicians, and researchers, biomedical engineering has facilitated the creation of advanced diagnostic tools, regenerative tissue technologies, and medical imaging systems such as magnetic resonance imaging (MRI), electrocardiography (ECG), and electroencephalography (EEG). These technologies have significantly enhanced disease detection, patient monitoring, and treatment precision.
Closely aligned with BME, bioengineering extends engineering methodologies to biological systems, emphasizing the development of innovative, sustainable, and economically viable biotechnological solutions [7-9]. Drawing upon concepts from biomechanics, biocatalysis, bioinformatics, and biotransport phenomena, bioengineering contributes to areas such as prosthetic design, biosensor fabrication, renewable bioenergy, and ecological system optimization [10-12]. Research in this domain also encompasses synthetic biology applications, including engineered microorganisms for biomanufacturing, portable diagnostic platforms, and tissue-engineered organs.
Biomedical and bioengineering advancements have collectively transformed modern healthcare, improving patient outcomes and quality of life [13-15]. Breakthroughs such as artificial organs, dialysis systems, prosthetic implants, and cardiovascular devices exemplify how engineering-driven innovation can restore physiological function and extend life expectancy. The continued convergence of these disciplines not only fosters medical innovation but also deepens our understanding of biological processes-laying the foundation for next-generation healthcare technologies and precision medicine.
Methods and Experimental Analysis
This study employed a structured and iterative methodological framework to investigate the influence of Artificial Intelligence (AI) and accelerated computing techniques on biomedical systems within the broader domain of Biomedical Engineering (BME). The overall approach combined data-driven analytics, computational modeling, and visual representation to achieve comprehensive and reliable outcomes.
The research process began with an extensive literature analysis to establish a solid theoretical foundation and identify existing research gaps. This preliminary investigation involved a systematic review of current advancements in AI-driven biomedical systems, accelerated computing frameworks, and their integrated applications in healthcare. The insights gained from this review informed the design and direction of the experimental framework. Subsequently, relevant datasets were collected and processed using the KNIME data analytics platform. Within KNIME, data mining and analytical workflows were implemented using various functional modules to ensure effective preprocessing, transformation, and post-processing.
These steps ensured data consistency, accuracy, and contextual relevance across all experimental stages. The processed data were then utilized to develop performance analytics and visualization prototypes. These prototypes were designed to evaluate and illustrate the interaction between AI algorithms, accelerated computing functionalities, and biomedical system performance. Multiple metrics were applied to assess computational efficiency, accuracy, and scalability. Comparative analyses were also conducted against conventional computational approaches to highlight performance improvements and methodological innovations.
Following the experimental phase, all results were analyzed and interpreted with respect to the research objectives. The discussion emphasized the implications of AI integration and accelerated computing for biomedical applications, addressing both the benefits and limitations observed during experimentation. This evaluation provided meaningful insights into potential advancements in diagnostic modeling, bioinformatics, and data-driven healthcare optimization.
Finally, the findings were consolidated into a comprehensive summary, acknowledging the study’s limitations and outlining potential directions for future research. The adopted methodology thus facilitated an in-depth exploration of how AI-enhanced accelerated computing can strengthen the analytical and operational capabilities of Biomedical Engineering, paving the way for innovative and intelligent medical solutions.
Background Research and Investigative Explorations for Available Knowledge
Bioengineering is a multidisciplinary domain that applies engineering concepts and analytical techniques to address complex biological and medical problems [1-11]. Historically, the origins of this field can be traced back to early medical innovations, including ancient prosthetic devices such as wooden toes found in Egyptian tombs-early evidence of humankind’s efforts to restore physical function.
During the 18th century, Luigi Galvani’s pioneering research on bioelectricity revealed the link between electrical signals and muscular activity, laying the foundation for modern electrophysiology and electrocardiology. His student, Alessandro Volta, later invented the first electrical battery, which facilitated the use of controlled electrical currents for therapeutic applications. The discovery of X-rays by Wilhelm Roentgen in the late 19th century further revolutionized diagnostic medicine through non-invasive imaging.
The 20th century marked a turning point in bioengineering, as mechanical, electrical, and chemical engineering principles merged to produce complex medical systems. This era saw the development of transformative technologies such as dialysis machines, pacemakers, artificial hearts, intelligent prosthetic limbs, and DNA-based diagnostic tools. Moving into the 21st century, bioengineering continues to evolve rapidly, integrating emerging fields such as nanotechnology, synthetic biology, and data-driven computational analysis to enhance human health and quality of life.
The formal recognition of bioengineering as a distinct field occurred in the mid-20th century. The term “bioengineering” was first introduced by Heinz Wolff in 1954 to describe the systematic application of engineering to biological systems. Initially, the discipline focused primarily on electrical engineering, owing to its early influence on medical device design. However, as collaboration between engineers, biologists, and clinicians deepened, the field expanded to encompass a broader understanding of life sciences. This multidisciplinary integration encouraged engineers to study biology, physiology, and medicine to design more effective and biologically compatible technologies.
Over time, biological engineering expanded its focus beyond medical systems to include environmental and agricultural applications. It became instrumental in areas such as soil conservation, ecosystem rehabilitation, and sustainable resource management. The first academic program dedicated to biological engineering was established at the University of California, San Diego in 1966, followed by similar programs at MIT and Utah State University. Many agricultural engineering departments worldwide have since redefined themselves as “Agricultural and Biological Engineering” or “Biosystems Engineering,” reflecting the growing convergence between biological and engineering sciences.
Biological engineering operates across multiple scales-from molecular and cellular processes to tissues, organisms, and entire ecosystems. It encompasses numerous subdisciplines, including microbiology, neurobiology, pharmacology, immunology, and systems biology [12-22]. This broad scope enables engineers and scientists to tackle a variety of challenges, ranging from biomedical device innovation to environmental restoration.
Bioinformatics, another crucial interdisciplinary domain, combines computer science, mathematics, and statistics to analyze and interpret biological data. It supports the study of genomics, proteomics, and molecular biology by enabling efficient data storage, pattern recognition, and algorithmic modeling. Through advanced computational tools, bioinformatics assists in identifying genetic variations such as single nucleotide polymorphisms (SNPs), which are instrumental in understanding disease mechanisms, evolutionary relationships, and desirable traits in agriculture. Beyond its practical applications, bioinformatics also aims to uncover the organizational logic of biological sequences, thereby enhancing our comprehension of molecular structure and function.
Biomedical engineering (BME), as an applied branch of bioengineering, integrates diverse subfields that collectively advance healthcare innovation. Biomechanics investigates the physical behavior of biological structures, applying mechanical principles at scales ranging from cellular to organismal. Biomaterials science explores material-tissue interactions for the design of implants, prosthetics, and tissue scaffolds. Biomedical optics studies the interaction of light with biological tissues for imaging, sensing, and therapy [23-33]. Tissue engineering combines biology and engineering to fabricate artificial tissues and organs for transplantation. Genetic engineering enables precise modification of genetic material for applications in medicine, agriculture, and biotechnology. Neural engineering seeks to interface with or restore neural systems through computational and device-based solutions. Pharmaceutical engineering integrates chemical and biological processes to optimize drug formulation, delivery, and therapeutic efficacy.
Functional genomics represents another transformative area of modern biomedical research. Unlike traditional genomics, which focuses on the static sequencing of DNA, functional genomics aims to elucidate the dynamic roles and interactions of genes and proteins. By leveraging high-throughput technologies such as transcriptomics, proteomics, and metabolomics, it provides insights into gene expression regulation, protein function, and molecular networks [34-44]. These genome-wide studies help to establish causal relationships between genetic variations and phenotypic outcomes, forming the foundation of systems biology. Ultimately, functional genomics enhances our understanding of disease mechanisms and contributes to the development of precision medicine strategies.
The collective evolution of bioengineering, biomedical engineering, bioinformatics, and functional genomics underscores the interdisciplinary nature of modern life sciences. Their convergence continues to drive innovation, enabling the development of intelligent medical systems, personalized healthcare solutions, and sustainable biotechnological applications for the future.
Innovations within Health Informatics
Biomedical engineering has experienced remarkable progress in recent years, driven by the integration of advanced technologies, medical expertise, and data-centric innovation. This interdisciplinary synergy has fostered a climate of creativity that enables biomedical engineers to design unprecedented solutions aimed at improving global health and quality of life. Among the most notable outcomes of this evolution are the groundbreaking healthcare innovations that have redefined diagnostic, therapeutic, and monitoring practices worldwide.
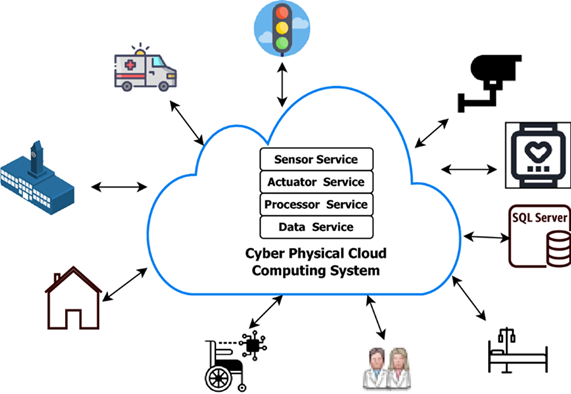
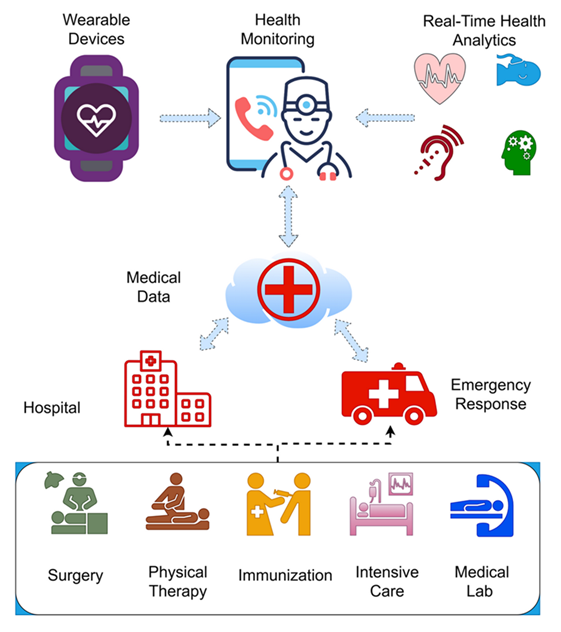
One of the most impactful areas of advancement lies in the development of medical devices and healthcare technologies. Biomedical engineers have introduced cutting-edge inventions such as prosthetic limbs with sensory feedback, artificial hearts, bionic contact lenses, and the ingestible “camera pill” - a miniature capsule equipped with a camera, light source, battery, and transmitter capable of capturing internal physiological processes. These devices have revolutionized medical diagnostics and treatment delivery, offering patients innovative and minimally invasive alternatives to traditional procedures. Beyond the creation of physical devices, biomedical engineering has significantly influenced pharmaceutical development and clinical practice. Research into human physiology and cellular mechanisms has facilitated the design of advanced drug delivery systems and targeted therapies for conditions such as cancer and cardiovascular diseases. Biomedical engineers have also contributed to the refinement of medical procedures, including laser-assisted surgeries, enhancing precision, safety, and patient recovery outcomes. Figures 1, 2 illustrate the interconnection between physical and cyber healthcare infrastructures and the broad retrospective spectrum of healthcare data, respectively.
Figure 1: Cyber Physical Healthcare Systems in Action.
Figure 2: Healthcare Data Spectrum in Action.
Collaboration among biomedical engineers, physicians, surgeons, and clinical technologists has been fundamental to these advancements. Such interdisciplinary cooperation has led to the creation of essential diagnostic and therapeutic tools, including magnetic resonance imaging (MRI) systems, dialysis machines, ultrasound devices, and real-time monitoring equipment. These technologies have transformed medical practice by improving the accuracy of disease diagnosis, enabling earlier interventions, and enhancing overall patient care.
Biomedical engineers have also expanded the frontier of personalized healthcare through innovations such as wearable biosensors and implantable pacemakers. These intelligent systems enable continuous health monitoring and remote diagnostics, empowering patients with greater autonomy and clinicians with real-time data for decision-making. The integration of biological insights with digital technologies exemplifies the growing field of health informatics, where data analytics and machine learning are increasingly applied to predict health outcomes, optimize treatment plans, and streamline healthcare management.
In summary, biomedical engineering continues to transform healthcare through innovation in both physical and digital domains. From advanced medical devices to data-driven health informatics, the field bridges engineering and medicine to deliver personalized, efficient, and sustainable healthcare solutions. These technological strides not only enhance clinical capabilities but also redefine patient experiences-marking a new era of intelligent, connected, and patient-centered medicine.
Progressions within Medical Science
Biomedical Engineering (BME) stands at the forefront of medical innovation, driving transformative advancements that are redefining modern healthcare. Through the integration of emerging technologies, biomedical engineers are revolutionizing diagnosis, treatment, and patient management, ultimately enhancing both clinical outcomes and the quality of life.
By combining engineering precision with medical expertise, BME continues to create tools and systems that expand the boundaries of what is possible in medical science. One of the most notable breakthroughs within this field is the adoption of robotic systems in surgery. Robotic-assisted surgical platforms offer unparalleled precision, stability, and control, minimizing human limitations such as hand tremors. These systems enable minimally invasive procedures, resulting in smaller incisions, shorter hospital stays, faster recovery times, and reduced risks of post-operative complications. Biomedical engineers are also pioneering the concept of telesurgery, where surgeons can remotely operate robotic instruments-expanding access to expert surgical care in geographically remote or underserved areas.
Tissue engineering represents another transformative frontier in biomedical science. The emergence of 3D bioprinting technologies has enabled the fabrication of complex biological structures, including tissues and organ models, that closely replicate natural human anatomy. These bioprinted constructs are being used not only for potential transplantation but also as advanced models for disease study, therapeutic testing, and regenerative medicine research. For instance, the successful printing of vascular networks has deepened our understanding of cardiovascular health and facilitated investigations into pathological conditions such as atherosclerosis. The convergence of Artificial Intelligence (AI) and Virtual Reality (VR) has further accelerated innovation in biomedical engineering. AI-driven algorithms are now integral to medical imaging and diagnostics, assisting clinicians in identifying abnormalities, predicting disease progression, and optimizing treatment strategies with enhanced accuracy. Meanwhile, VR technologies are revolutionizing medical training and surgical planning. Immersive virtual simulations allow physicians to visualize complex anatomical structures and practice high-risk procedures in a controlled environment. Furthermore, VR-based training enhances communication skills, empathy, and patient-care competence among healthcare professionals.
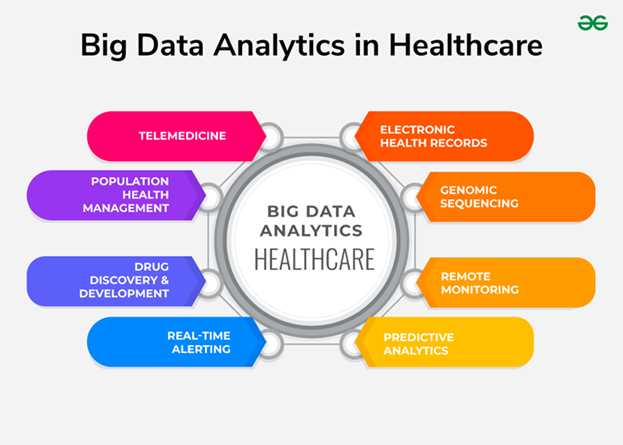
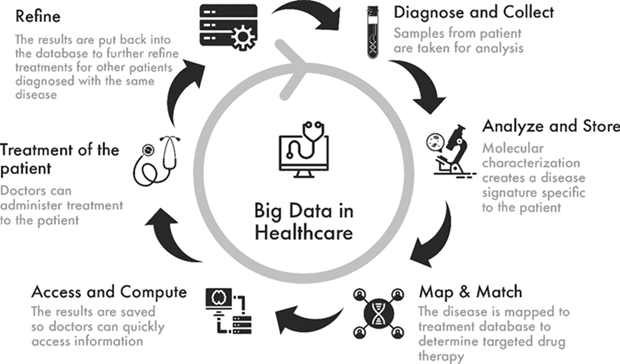
The rapid expansion of Big Data and medical data analytics (Figures 3, 4) complements these technological developments. Data science plays a pivotal role in integrating large-scale biomedical datasets to uncover hidden patterns, predict health outcomes, and support personalized treatment approaches. The synergy between data-driven models and intelligent medical systems represents a fundamental shift toward evidence-based, precision-oriented healthcare.
Figure 3: Types of Big Data within Healthcare Informatics.
Figure 4: Medical Data in Action.
Importantly, these innovations are no longer confined to research laboratories-they are increasingly being deployed in hospitals and clinics worldwide. From robotic-assisted surgeries and real-time imaging systems to AI-guided diagnostics and VR-based training environments, biomedical engineering is reshaping the global healthcare landscape. By harnessing the collective power of engineering, computation, and medical sciences, the field continues to pioneer solutions that enhance longevity, improve quality of care, and redefine the future of medicine.
Current Improvements & Advances within BME
Recent advancements in Biomedical Engineering (BME) are fundamentally transforming the landscape of modern healthcare. Through the convergence of engineering principles, biological sciences, and digital technologies, BME continues to pioneer innovations that enhance diagnostic accuracy, therapeutic precision, and overall patient well-being. The field is rapidly expanding into diverse domains such as prosthetics, surgical systems, diagnostic devices, and advanced imaging modalities-reflecting a shift toward personalized, data-driven, and minimally invasive healthcare solutions.
A major contemporary development in this field is the evolution of wearable and implantable medical technologies. Devices such as smart fitness trackers, biosensors, and implantable cardiac monitors have enabled continuous, real-time physiological monitoring. These systems facilitate early symptom detection, timely medical intervention, and long-term remote patient management. Beyond improving diagnostic accuracy, wearable and implantable technologies also contribute to reducing healthcare costs and improving treatment efficiency through predictive analytics and personalized therapeutic responses. Another promising area of innovation lies in nanorobotics and nanoscale engineering. Nanorobots-microscopic machines designed to operate within the human body-hold immense potential for targeted therapeutic delivery, pathogen detection, and cellular-level diagnostics. Recent advances in nanotechnology have demonstrated the feasibility of deploying these robots to selectively target cancerous cells, shrink tumors, and minimize the side effects commonly associated with conventional treatments like chemotherapy. As the precision and control of nanorobotic systems continue to advance, they are expected to play a pivotal role in next-generation, minimally invasive therapies. Equally transformative are Brain-Computer Interfaces (BCIs), which create direct communication pathways between the human brain and external devices. By translating neural signals into actionable commands, BCIs offer new possibilities for individuals with motor impairments to regain control over prosthetic limbs or assistive technologies. Ongoing research aims to improve the resolution, speed, and accuracy of signal interpretation, thereby broadening potential applications to include cognitive augmentation and neurorehabilitation. While ethical considerations regarding privacy and human enhancement persist, BCIs continue to represent one of the most promising intersections of neuroscience, engineering, and artificial intelligence.
Another frontier of biomedical innovation is 3D bioprinting, a technology that integrates living cells, biomaterials, and growth factors to fabricate tissue-like structures. Significant progress has already been made toward creating complex biological constructs such as vascularized tissues, cartilage, and even prototype human organs. For example, recent achievements in 3D bioprinting have enabled the creation of human-sized cardiac models and artificial skin for burn therapy. In the long term, this technology could revolutionize regenerative medicine by addressing the global shortage of donor organs and enabling highly personalized treatments.
Collectively, these advancements highlight the growing influence of biomedical engineering in shaping the future of healthcare. The integration of wearable biosensors, nanorobotic therapies, brain–machine communication systems, and 3D bioprinting technologies signifies a transition toward smarter, more adaptive, and patient-centered medical solutions. As these technologies mature, they will not only redefine clinical practices but also enhance the overall quality of human health, offering new pathways for prevention, diagnosis, and treatment in the decades ahead.
Major Movements in BME
Biomedical Engineering (BME) continues to be one of the most transformative fields in modern science, driving innovations that have fundamentally reshaped healthcare delivery and patient outcomes. Through the fusion of engineering, biology, and information technology, biomedical engineers have developed a wide spectrum of devices, systems, and processes that enhance diagnosis, treatment, and rehabilitation. Among the most impactful innovations are prosthetic and bionic technologies, which have dramatically improved the lives of individuals with physical impairments. Advanced artificial limbs, bionic exoskeletons, and sensory-enhancing contact lenses exemplify how BME bridges human capability and technology. These systems restore lost functions and, in some cases, extend natural abilities through biomechanical integration and adaptive control mechanisms.
Robotic and laser-assisted surgical systems represent another milestone in biomedical progress. By enhancing precision, minimizing invasiveness, and reducing recovery times, these technologies have revolutionized traditional surgical practices. Modern robotic platforms enable complex procedures to be performed with superior accuracy, while laser-based instruments contribute to improved patient safety and outcomes. The development of implantable devices and drug delivery systems has also reshaped therapeutic practices. Targeted delivery mechanisms-such as implantable pumps and microcapsule systems-ensure localized treatment and reduced side effects. In parallel, advances in medical imaging-including X-ray, CT, MRI, and PET technologies-have dramatically improved clinicians’ ability to visualize, diagnose, and monitor diseases with exceptional accuracy. Radiation-based therapies now serve as critical tools in the management and treatment of cancers, supported by continuous advancements in imaging-guided precision therapy.
Emerging materials and nanotechnologies have further expanded the boundaries of biomedical innovation. Nanomaterials enable controlled drug release, tissue regeneration, and biosensing applications, while 3D bioprinting has introduced the ability to construct biologically functional tissues and organs. These developments hold transformative potential for regenerative medicine and organ transplantation, addressing long-standing challenges such as donor shortages and immune compatibility.
In parallel, genome editing technologies, particularly CRISPR-based systems, have opened new frontiers in genetic medicine by allowing precise modification of DNA sequences. These breakthroughs bring hope for the treatment and prevention of hereditary diseases, ushering in a new era of personalized genetic therapy.
Contemporary trends within BME also emphasize integrated and data-driven healthcare systems. Technologies such as organs-on-chips, microbubbles, and transdermal drug delivery patches are enabling sophisticated diagnostic and therapeutic capabilities on a microscale. Meanwhile, wearable biosensors and health-monitoring devices provide real-time physiological data that support preventive medicine and individualized care management.
The integration of Artificial Intelligence (AI) and Machine Learning (ML) into biomedical applications is further transforming diagnostics and decision-making. AI-driven image analysis enhances early disease detection, while predictive algorithms assist in treatment planning and patient outcome forecasting. Combined with Virtual and Augmented Reality (VR/AR), these technologies are also redefining medical training, rehabilitation, and surgery simulation.
One of the most influential recent developments is the Internet of Medical Things (IoMT)-a network of interconnected medical devices, systems, and applications. IoMT enables continuous patient monitoring, automated diagnostics, and seamless data exchange between healthcare professionals. From wearable trackers to intelligent hospital equipment and remote monitoring platforms, IoMT is enhancing the accessibility, precision, and efficiency of modern healthcare delivery.
In essence, the major movements within Biomedical Engineering represent a paradigm shift toward personalized, connected, and intelligent healthcare systems. The synergy between prosthetics, nanotechnology, robotics, bioprinting, and digital informatics continues to redefine the boundaries of medicine. As these technologies evolve, they promise not only to extend human longevity but also to elevate the quality of life through innovation-driven medical excellence.
BME: AI Perspectives
Artificial Intelligence (AI), particularly Machine Learning (ML), has emerged as a transformative force in biomedical engineering, offering advanced methods for analyzing complex, high-dimensional data and uncovering patterns that were previously beyond human interpretation. The integration of AI into biomedical research enables predictive modeling, automated diagnostics, and intelligent data-driven decision-making, marking a paradigm shift in how medical data is understood and utilized.
One of the most significant contributions of ML lies in its predictive capabilities-the ability to learn from measurable data and forecast clinical outcomes. In psychiatric and behavioral medicine, for instance, ML algorithms have been used to infer emotional states and mood variations through smartphone sensor data, capturing subtle behavioral cues from daily activities. Similarly, in neuroscience, ML models have been applied to decode neural activity, enabling the prediction of motor intentions and facilitating the control of prosthetic and assistive devices through brain–computer interfaces (BCIs). These advancements not only enhance patient rehabilitation but also expand the boundaries of human-machine symbiosis.
Beyond prediction, ML plays a crucial role in model validation and system understanding. By comparing algorithmic inferences with human-devised models, ML helps identify missing variables, incorrect assumptions, and unrecognized relationships within biological systems. It excels in modeling nonlinear and recurrent interactions, which are characteristic of physiological processes such as neural signaling, gene regulation, and metabolic pathways. This ability allows for a more accurate and comprehensive representation of the intricate dynamics underlying biological systems.
As biomedical datasets continue to expand-encompassing genomic sequences, medical imaging, wearable sensor outputs, and electronic health records-ML becomes indispensable for data integration and interpretation. Traditional analytical methods often struggle to capture the latent structures and high-dimensional relationships within such datasets. In contrast, ML algorithms such as deep neural networks, random forests, and gradient-boosted trees can extract meaningful insights, identify hidden correlations, and produce reliable predictions, even when individual variable contributions remain ambiguous.
The application of ML in neural decoding and encoding provides an exemplary case of its effectiveness. Neural networks and ensemble-based models have surpassed conventional statistical approaches in mapping brain activity patterns to external stimuli and behavioral responses. By accurately modeling the nonlinear dependencies between neural firing rates and external variables, ML has redefined our understanding of neural computation and brain dynamics. These achievements serve as benchmarks for advancing both theoretical neuroscience and practical neuroengineering applications.
Importantly, recent developments have made ML increasingly accessible to biomedical researchers. The availability of open-source libraries, automated ML frameworks, and intuitive analytical platforms has lowered the barrier to entry, allowing scientists to apply complex algorithms without requiring deep expertise in computer science. This democratization of AI empowers researchers to focus on scientific inquiry-formulating hypotheses, interpreting model outcomes, and translating computational findings into clinical applications.
AI and ML are now central to the evolution of biomedical engineering, enabling innovations that range from behavioral prediction and neural decoding to advanced diagnostics and personalized therapy. By embracing complexity and leveraging data-driven intelligence, these technologies not only enhance our understanding of human biology but also pave the way for a new era of precision medicine-where healthcare decisions are informed by computational insight, predictive modeling, and continuous learning. The growing synergy between AI and biomedical science promises to revolutionize how we diagnose, treat, and ultimately prevent diseases in the years ahead.
Functional Genomics: AI Perspectives
Artificial Intelligence (AI) and Machine Learning (ML) have become indispensable in the modern study of biology and bioinformatics, particularly in functional genomics. Their ability to process and interpret large-scale, multidimensional biological datasets has redefined how researchers explore genomic, proteomic, and cellular functions. By uncovering patterns and relationships hidden within massive data repositories, AI-driven approaches are accelerating discoveries, improving predictive accuracy, and deepening our understanding of biological complexity.
In genomics, ML algorithms are widely applied in regulatory, structural, and functional studies. They assist in identifying gene expression patterns, predicting regulatory elements, classifying protein structures, and uncovering interactions among genes. ML methods, often enhanced through natural language processing (NLP), have proven valuable in parsing and interpreting vast genomics-related text data—facilitating tasks such as relation extraction, annotation, and entity recognition. A major milestone in this domain has been the integration of ML with next-generation sequencing (NGS) technologies. This combination has drastically reduced the cost and processing time of genome sequencing, making large-scale genomic studies feasible and efficient. Furthermore, ML assists in gene editing applications like CRISPR by improving target site selection and minimizing off-target effects, thereby enhancing precision in genetic modification.
In proteomics, machine learning has revolutionized how proteins and their interactions are analyzed. Advanced algorithms now interpret mass spectrometry data with higher accuracy, enabling efficient protein identification and quantification. These developments have strengthened disease diagnosis, facilitated biomarker discovery, and revealed novel protein expression patterns. Likewise, microarray data analysis-once limited by data complexity—has benefited from ML-driven classification and clustering techniques, allowing researchers to discern relationships between genes, predict expression profiles, and map disease associations with greater reliability.
ML and NLP techniques also play a crucial role in biological text mining, enabling researchers to process large volumes of scientific literature. Through automated extraction of biological knowledge, ML aids in identifying molecular interactions, discovering drug targets, and annotating gene or protein functions. Such computational tools make it possible to integrate data from diverse sources and generate comprehensive biological insights far beyond manual analysis.
In systems biology, machine learning supports the modeling and simulation of intricate biological systems. Techniques such as probabilistic graphical models, Bayesian networks, and genetic algorithms are utilized to capture dynamic relationships between genes, proteins, and metabolites.
These models help decide how genetic and molecular interactions give rise to observable phenotypes, advancing our understanding of complex biological behavior. Additionally, ML facilitates genotype-phenotype correlation studies, revealing how genetic variations influence organismal traits and disease mechanisms.
The synergy between AI and functional genomics has not only enhanced data interpretation but also transformed experimental design and hypothesis generation. Deep learning models can detect nonlinear and hierarchical relationships within genomic sequences, while reinforcement learning frameworks are being explored to optimize gene network simulations. This integration bridges the gap between computational prediction and experimental validation, accelerating translational research and personalized medicine.
Several specialized AI-powered tools have exemplified this transformation. DeepVariant, a deep learning–based tool, accurately identifies genetic variants from sequencing data, offering scalable and cloud-compatible genomic analysis. Atomwise leverages AI for molecular modeling and drug discovery, enabling the exploration of atomic-level interactions in proteins. Meanwhile, CellProfiler, powered by ML algorithms, allows quantitative cellular analysis from microscopy images, facilitating large-scale biological image processing with remarkable precision.
The application of AI and ML in functional genomics has thus reshaped the landscape of biological and medical research. These technologies enable high-throughput data interpretation, personalized genomic insights, and predictive modeling that guide clinical decision-making. By merging computational intelligence with biological inquiry, AI-driven genomics continues to unlock new frontiers in precision medicine, drug discovery, and molecular diagnostics-bringing us closer to a future where genomic information directly informs individualized healthcare strategies.
Healthcare Informatics: AI Deep Dive
Artificial intelligence (AI) has revolutionized the healthcare industry by improving patient care and outcomes. AI in healthcare has the potential to transform the way we diagnose diseases, develop treatments, and prevent illnesses. The use of AI technology, such as machine learning and natural language processing, has enabled medical professionals to make more accurate diagnoses, personalize treatments, and streamline clinical processes. Machine learning, one of the most common AI techniques in healthcare, has facilitated medical diagnosis and treatment by processing large amounts of clinical data, identifying patterns, and making predictions with higher accuracy. It has been used for precision medicine, predicting treatment success based on individual patient characteristics, and detecting correlations and changes in health data that may indicate health risks. Deep learning, a subset of machine learning, has also been applied to tasks such as speech recognition and natural language processing, aiding in medical record analysis and clinical decision-making. To better understand AI enabled mechanics in terms of healthcare further research explorations are of immense importance in the upcoming years.
Artificial Intelligence (AI) has emerged as a transformative force within healthcare informatics, offering unprecedented opportunities to enhance diagnostics, treatment planning, and patient management. Through technologies such as machine learning (ML) and natural language processing (NLP), AI enables healthcare professionals to interpret vast datasets with improved precision, leading to more personalized and efficient medical care. Among AI’s most impactful applications, machine learning plays a central role in medical data analysis, enabling the identification of complex patterns that assist in predicting disease outcomes, detecting anomalies, and optimizing therapeutic interventions. These capabilities have supported the rise of precision medicine, where predictive models consider individual genetic, clinical, and behavioral factors to recommend targeted treatments and preventive measures.
Deep learning, a specialized subset of ML, has further expanded the analytical capacity of AI systems, allowing for sophisticated recognition of speech, text, and images in clinical contexts. This technology aids in interpreting medical imaging scans, streamlining documentation through automated language analysis, and supporting clinicians in data-driven decision-making. Figure 5 provides an illustrative overview of AI-enabled mechanics in healthcare, emphasizing how these tools reshape medical processes from diagnosis to treatment optimization.
Figure 5: AI Enabled Healthcare.
NLP continues to transform healthcare workflows by allowing computers to comprehend, extract, and process medical information expressed in natural language. By analyzing electronic health records (EHRs), physician notes, and clinical reports, NLP tools enhance diagnostic accuracy and enable real-time recommendations for patient-specific care.
These systems not only improve the efficiency of clinical operations but also help in structuring unorganized data into actionable insights for medical research and health analytics. Although traditional rule-based expert systems once served as foundational tools for clinical decision support, they are increasingly being replaced by ML-based models, which offer adaptive learning, scalability, and superior predictive accuracy.
AI applications in healthcare extend well beyond clinical diagnostics to encompass administrative optimization, data management, and decision support. Automating routine administrative and data-entry tasks significantly reduces the likelihood of human error, improves time efficiency, and allows healthcare providers to focus more on direct patient interaction. However, the integration of AI into healthcare infrastructure also presents challenges-particularly regarding patient data privacy, model transparency, regulatory compliance, and physician trust. Addressing these issues is critical for the ethical and reliable implementation of AI-driven healthcare technologies.
From a broader perspective, AI’s influence in specialized medical fields continues to expand. In cardiovascular care, AI algorithms now support the identification and risk assessment of coronary artery disease, while wearable devices and remote monitoring tools enable early detection of cardiac events. In dermatology, deep learning models have surpassed human accuracy in detecting and classifying skin cancers. Similarly, AI-driven systems in gastroenterology enhance the accuracy of endoscopic imaging, enabling early recognition of gastrointestinal abnormalities. Applications in infectious disease management utilize AI for predicting antimicrobial resistance, identifying treatment outcomes, and detecting diseases such as malaria and tuberculosis.
AI’s integration into oncology represents one of its most profound clinical impacts. Machine learning algorithms assist in cancer detection, tumor characterization, and personalized therapy selection, particularly in breast and prostate cancer diagnostics. Ophthalmology has adopted FDA-approved AI tools for diagnosing diabetic retinopathy, while pathology has benefited from digital image analysis powered by AI for identifying malignancies in tissue samples. In radiology, deep neural networks now achieve expert-level accuracy in interpreting CT and MRI scans, enhancing both interpretive and non-interpretive tasks such as image enhancement and quality assurance. AI models based on Bayesian and neural network frameworks are also being leveraged for disease classification and clinical prediction through EHR data analytics. Figures 6, 7 illustrate the growing significance and historical evolution of AI within life sciences and medical practice, respectively.
Figure 6: AI Advances within Life and Medical Sciences.
Figure 7: History of AI in Healthcare Markets.
Beyond clinical diagnosis, AI facilitates telemedicine and remote healthcare delivery-particularly valuable in developing regions where access to medical specialists is limited. Through continuous monitoring and automated alerts based on sensor data, AI enables proactive healthcare interventions.
Machine learning algorithms can analyze patient history and family background to assess disease susceptibility, while NLP-driven systems streamline EHR management, ensuring data consistency and improved report generation. Moreover, AI tools now support the identification of potential drug–drug interactions by analyzing large corpora of biomedical literature and user-generated pharmacovigilance data.
Global technology companies have played a pivotal role in advancing AI for healthcare. IBM’s Watson Oncology collaborates with leading medical institutions to provide evidence-based cancer treatment recommendations.
Microsoft’s Hanover Project leverages AI to accelerate drug discovery by predicting effective cancer therapies. Google’s DeepMind works with the UK’s National Health Service to improve risk detection and tissue analysis, while Tencent and Intel are investing in intelligent diagnostic imaging and AI-based healthcare platforms. Meanwhile, Neuralink explores neural interface technologies that may redefine patient rehabilitation and brain-computer integration.
The widespread adoption of AI in healthcare is accompanied by evolving regulatory frameworks. Policies such as the Health Insurance Portability and Accountability Act (HIPAA) in the United States and the General Data Protection Regulation (GDPR) in Europe ensure the protection of patient data and ethical algorithm deployment.
The U.S. Food and Drug Administration (FDA) has developed an action plan for AI-powered medical devices, and the Department of Health and Human Services (HHS) promotes ethical guidelines emphasizing autonomy, beneficence, and justice. Similar initiatives across Europe and Asia continue to strengthen governance for responsible AI utilization in healthcare.
AI-driven healthcare informatics is redefining medical science by integrating data intelligence with human expertise. Through advancements in predictive modeling, image recognition, clinical decision support, and administrative automation, AI enhances healthcare accessibility, efficiency, and patient outcomes worldwide. Continued collaboration between AI developers, clinicians, and policymakers will be essential for addressing challenges related to bias, interpretability, and trust-ensuring that AI fulfills its transformative potential for global health improvement.
Results and Findings
The outcomes of this research demonstrate the transformative potential of Artificial Intelligence (AI) across multiple dimensions of healthcare innovation. One of the most significant findings is AI’s unparalleled ability to process and interpret vast and complex datasets with high speed and accuracy. Through the use of advanced machine learning algorithms, AI systems can efficiently identify subtle correlations and predictive patterns within medical data, thereby enhancing diagnostic precision and supporting evidence-based treatment strategies. This capability not only accelerates clinical decision-making but also minimizes human error, leading to more reliable and consistent medical outcomes.
Medical imaging emerged as a major area of impact in this study. The integration of AI algorithms into imaging workflows-particularly in modalities such as X-ray, computed tomography (CT), and magnetic resonance imaging (MRI)-has demonstrated strong potential for early disease detection and classification.
Trained deep learning models can accurately recognize structural anomalies, lesions, or tumor formations that may be overlooked by traditional diagnostic methods. Such AI-assisted analysis contributes to faster image interpretation, improved diagnostic accuracy, and reduced workload for radiologists, ultimately enhancing overall patient care efficiency.
Another important finding highlights AI’s contribution to personalized and precision medicine. By synthesizing a patient’s medical history, genomic profile, and lifestyle factors, AI can generate individualized treatment recommendations. These adaptive models offer insights into the most effective therapies for each patient, improving treatment outcomes and minimizing adverse effects. The ability to tailor healthcare interventions at the individual level signifies a paradigm shift from generalized treatment protocols to more targeted, patient-centered approaches.
The research also underscores the growing significance of remote patient monitoring enabled by AI-driven systems. Through continuous data collection from wearable sensors and health monitoring devices, AI can detect early signs of physiological deterioration or potential complications in real time. Predictive analytics then allows clinicians to intervene proactively, preventing hospital readmissions and supporting long-term patient management. This shift toward preventive and continuous care represents a key advancement in modern healthcare informatics. Collectively, the results illustrate how AI empowers healthcare systems to move from reactive to proactive care, integrating predictive intelligence with clinical expertise. Figures 8, 9 present a visual summary of the experimental outcomes and analytical insights, while Figures 10, 11 depict the proposed AI-driven experimental design framework for future healthcare applications. These visualizations collectively highlight how AI’s integration with medical technologies can strengthen diagnostic efficiency, optimize treatment precision, and redefine patient management strategies. The findings confirm that AI is not merely a supportive technology but a transformative enabler in healthcare, providing the foundation for next-generation innovations in diagnosis, treatment, and patient-centered care.
Figure 8: A Visual Representation of the Findings from the Research Results 1.
Figure 9: A Visual Representation of the Findings from the Research Results 2.
Figure 10: A Visualization of Future Healthcare (Experimental) 1.
Figure 11: A Visualization of Future Healthcare (Experimental) 2.
Discussions and Future Directions
Recent research in Artificial Intelligence (AI) and healthcare continues to redefine the boundaries of modern medicine, fostering innovation in diagnostics, therapeutics, and patient management. The convergence of AI technologies-particularly in medical imaging, drug discovery, and personalized healthcare-has demonstrated the potential to improve clinical accuracy and operational efficiency across multiple domains. By leveraging powerful data-driven algorithms, AI systems can process vast repositories of medical data, including patient histories, genomic datasets, and clinical trial outcomes, to identify patterns that might otherwise remain undetected. This ability to synthesize complex information enables earlier and more precise disease detection, informed decision-making, and optimized treatment pathways.
Natural Language Processing (NLP) represents another critical advancement discussed within this research. NLP facilitates the interpretation of unstructured textual data, such as physician notes, radiology reports, and patient narratives, allowing for the extraction of meaningful clinical insights. Through the automation of data interpretation, NLP-based systems reduce the cognitive burden on healthcare professionals, improve the accuracy of medical records, and support intelligent clinical decision support systems. These developments mark an important step toward integrating unstructured and structured data sources into unified, AI-powered healthcare ecosystems.
The emergence of wearable and connected health technologies further underscores the transformative role of AI in healthcare delivery. Equipped with advanced sensors and predictive analytics, wearable devices continuously monitor vital parameters such as heart rate, oxygen saturation, and physical activity levels.
The data collected can be analyzed in real-time using AI algorithms to detect early indicators of potential health issues, enabling timely medical interventions. This approach shifts healthcare from a reactive to a proactive model-emphasizing prevention, early diagnosis, and personalized care outside of traditional clinical environments.
Looking ahead, the future of AI in healthcare promises continued innovation and broader integration across clinical, administrative, and research domains. Future directions are expected to focus on explainable and ethical AI, ensuring transparency, fairness, and accountability in algorithmic decision-making. Research will likely emphasize the development of federated learning frameworks and privacy-preserving models to safeguard patient data while maintaining the benefits of collaborative AI training across institutions. Additionally, advances in multimodal AI-combining visual, genomic, and textual data-will further enhance predictive accuracy and diagnostic precision, leading to truly holistic healthcare intelligence systems.
Moreover, the continued collaboration between clinicians, data scientists, and engineers will be essential to translating AI innovations into real-world medical practice. Standardization of datasets, validation through clinical trials, and adherence to evolving regulatory and ethical guidelines will determine the safe and effective implementation of AI-driven healthcare solutions.
AI stands at the forefront of a new era in healthcare innovation. Its ability to transform large-scale medical data into actionable intelligence is revolutionizing diagnostics, enabling personalized therapies, and improving patient outcomes. Ongoing research and technological advancement will continue to expand the horizons of AI in healthcare, paving the way for a more efficient, predictive, and patient-centered healthcare ecosystem in the future.
Conclusions
Accelerated computing has fundamentally transformed the way humanity perceives, processes, and visualizes information-pushing the boundaries of technological capability to unprecedented heights. Despite remarkable progress in artificial intelligence (AI), high-performance computing, and biomedical innovation, the rapid pace of advancement raises critical concerns regarding human health, safety, and ethical responsibility. As systems grow more powerful and complex, so too do the risks and implications associated with their application in healthcare and life sciences.
Human civilization stands at a pivotal crossroads where engineering excellence and medical innovation are deeply intertwined. The evolution of computational intelligence continues to reshape how we interact with machines, interpret data, and deliver care. However, with this power comes profound responsibility. Ethical governance, moral integrity, and regulatory vigilance must remain central to all technological progress-particularly when it concerns human life. Without stringent oversight, even the most advanced systems may yield unintended and potentially catastrophic outcomes.
The COVID-19 pandemic served as a stark reminder of humanity’s vulnerability and the far-reaching consequences of systemic failures. It highlighted the importance of responsible innovation, transparent data management, and global collaboration in safeguarding human well-being.
Moving forward, the fusion of biomedical engineering and AI will continue to redefine the landscape of healthcare, offering transformative solutions to challenges once deemed insurmountable. Yet, amid this progress, it is essential that human values, empathy, and accountability guide the trajectory of technological development. The future will undoubtedly bring extraordinary achievements and unforeseen complexities as humanity ventures further into the digital and biological frontier.
As we pursue innovation, we must also remain mindful of the ethical boundaries that protect the sanctity of life. Ultimately, every technological leap must be balanced with compassion, caution, and conscience. In closing, one fundamental truth must endure above all others: every human being is fallible, and it is through humility, integrity, and reflection that progress attains its true meaning. The evolution of technology should serve humanity-not surpass it-and the measure of our advancement will rest not solely in our inventions, but in the wisdom with which we wield them.
Supplementary Information
The various original data sources some of which are not all publicly available, because they contain various types of private information. The available platform provided data sources that support the exploration findings and information of the research investigations are referenced where appropriate.
Acknowledgments
The authors would like to acknowledge and thank the GOOGLE Deep Mind Research with its associated pre-prints access platforms. This research exploration was investigated under the platform provided by GOOGLE Deep Mind which is under the support of the GOOGLE Research and the GOOGLE Research Publications within the GOOGLE Gemini platform. Using their provided platform of datasets and database associated files with digital software layouts consisting of free web access to a large collection of recorded models that are found within research access and its related open-source software distributions which is the implementation for the proposed research exploration that was undergone and set in motion. There are many data sources some of which are resourced and retrieved from a wide variety of GOOGLE service domains as well. All the data sources which have been included and retrieved for this research are identified, mentioned and referenced where appropriate.
Declarations
Funding
No Funding was provided for the conduction concerning this research.
Conflict of Interest/Competing Interests
There is no Conflict of Interest or any type of Competing Interests for this research.
Ethics Approval
The authors declare no competing interests for this research.
Consent to Participate
The authors have read, approved the manuscript and have agreed to its publication.
Consent for Publication
The authors have read, approved the manuscript and have agreed to its publication.
Availability of Data and Materials
The various original data sources some of which are not all publicly available, because they contain various types of private information. The available platform provided data sources that support the exploration findings and information of the research investigations are referenced where appropriate.
Code Availability
Mentioned in details within the Acknowledgements section.
Authors’ Contributions
Described in details within the Acknowledgements section.
References
- Akhtar, Z. B. (2025). Artificial intelligence within medical diagnostics: A multi-disease perspective. Artificial Intelligence in Health, 5173.
Publisher | Google Scholor - Akhtar, Z. B. (2024). The design approach of an artificial intelligent (AI) medical system based on electronical health records (EHR) and priority segmentations. The Journal of Engineering, 4:e12381.
Publisher | Google Scholor - Akhtar, Z. B., Rawol, A. T. (2025). Advanced Methods towards Functional Genomics & Medical Informatics: The Artificial Intelligence (AI) and Integrated Computing Perspectives. Trends Telemed E-Health, 5(5).
Publisher | Google Scholor - Kim, S. J., Jeong, B., Park, K. D., Hahn, S. K., Noh, I. (2025). Smart Biomaterials for Delivery of Drugs and Cells. Biomaterials Research, 29:0227.
Publisher | Google Scholor - Mozafari, M. (2025). How artificial intelligence shapes the future of biomaterials? Next Materials, 7:100381.
Publisher | Google Scholor - Rahaman, M. T., Hossain Khan, M. S. (2025). Biomaterials for manufacturing environmentally sustainable textiles and apparel: sources, applications, challenges, enablers and future directions. International Journal of Environmental Science and Technology, 1-56.
Publisher | Google Scholor - Vinchurkar, K., Bukke, S. P. N., Jain, P., Bhadoria, J., Likhariya, M., et al. (2025). Advances in sustainable biomaterials: characterizations, and applications in medicine. Discover Polymers, 2(1):2.
Publisher | Google Scholor - Qiu, S., Chen, G. Y., Qin, Y. D., Li, T. T., Yang, F. Q. (2025). Recent Advance in Electrochemical Chiral Recognition Based on Biomaterials (2019-2024). Molecules, 30(16):3386.
Publisher | Google Scholor - Panahi, O. (2025). Innovative Biomaterials for Sustainable Medical Implants: A Circular Economy Approach. European Journal of Innovative Studies and Sustainability, 1(2):20-29.
Publisher | Google Scholor - Jurczak, K. M., van Der Boon, T. A., Devia-Rodriguez, R., Schuurmann, R. C., Sjollema, J., et al. (2025). Recent regulatory developments in EU Medical Device Regulation and their impact on biomaterials translation. Bioengineering & Translational Medicine, 10(2):e10721.
Publisher | Google Scholor - Ebrahim, N. A., Soliman, S. M. (2026). Advanced biomaterials and biomedical devices for studying tumor-associated fibroblasts: current trends, innovations, and future prospects. Biomedical Materials & Devices, 4(1):287-301.
Publisher | Google Scholor - Amponsah, O., Nopuo, P. S. A., Manga, F. A., Catli, N. B., Labus, K. (2025). Future-Oriented Biomaterials Based on Natural Polymer Resources: Characteristics, Application Innovations, and Development Trends. International Journal of Molecular Sciences, 26(12):5518.
Publisher | Google Scholor - Akhtar, Z. B. (2025). Voices in the night: Sleep paralysis & The intersection of brain, trauma, traditions. Journal of Pain Research and Management, 1(1):62-74.
Publisher | Google Scholor - Akhtar, Z. B., Rozario, V. S. (2025). AI Perspectives Within Computational Neuroscience: EEG Integrations and the Human Brain. In Artificial Intelligence and Applications. 3(2):145-160.
Publisher | Google Scholor - Akhtar, Z. B. (2025). Exploring AI for pain research management: A deep dive investigative exploration. Journal of Pain Research and Management, 1(1):28-42.
Publisher | Google Scholor - Tajurahim, N. A. N., Mahmood, S., Ngadiman, N. H. A., Sing, S. L. (2025). Biomaterials for tissue engineering scaffolds: Balancing efficiency and eco-friendliness through life cycle assessment. Cleaner Environmental Systems, 16:100253.
Publisher | Google Scholor - Ingle, A. P., Saxena, S., Moharil, M. P., Rivaldi, J. D., Ramos, L., et al. (2025). Production of biomaterials and biochemicals from lignocellulosic biomass through sustainable approaches: current scenario and future perspectives. Biotechnology for Sustainable Materials, 2(1):3.
Publisher | Google Scholor - Davis Jr, R., Duggal, I., Peppas, N. A., Gaharwar, A. K. (2025). Designing the Next Generation of Biomaterials through Nanoengineering. Advanced Materials, 37(39):2501761.
Publisher | Google Scholor - Zhang, R., Lin, H., Wang, Z., Cheng, S., Li, C., et al. (2025). Research progress on antimicrobial biomaterials. Macromolecular Bioscience, 25(9):e00239.
Publisher | Google Scholor - Liu, S., Manshaii, F., Chen, J., Wang, X., Wang, S., et al. (2025). Unleashing the potential of electroactive hybrid biomaterials and self-powered systems for bone therapeutics. Nano-Micro Letters, 17(1):44.
Publisher | Google Scholor - Liao, Z., Liu, T., Yao, Z., Hu, T., Ji, X., et al. (2025,). Harnessing stimuli‐responsive biomaterials for advanced biomedical applications. In Exploration, 5(1):20230133.
Publisher | Google Scholor - Wang, F., Lei, H., Tian, C., Ji, Y., Wang, F., et al. (2025). An efficient biosynthetic system for developing functional silk fibroin‐based biomaterials. Advanced Materials, 37(7):2414878.
Publisher | Google Scholor - Zhu, Y., Guo, S., Ravichandran, D., Ramanathan, A., Sobczak, M. T., et al. (2025). 3D‐Printed Polymeric Biomaterials for Health Applications. Advanced Healthcare Materials, 14(1):2402571.
Publisher | Google Scholor - Wang, W., Wang, J., Hu, Z., Yan, X., Gao, Q., et al. (2025). Advancing aggregation‐induced emission‐derived biomaterials in viral, tuberculosis, and fungal infectious diseases. Aggregate, 6(3):e715.
Publisher | Google Scholor - Felix, R. B., Shabazz, A., Holeman, W. P., Han, S., Wyble, M., et al. (2025). From promise to practice: recent growth in 30 years of tissue engineering commercialization. Tissue Engineering Part A, 31(7-8):285-302.
Publisher | Google Scholor - Kobayashi, K., Horii, T., Wada, A., Yamanaka, K., Hagiwara, A., et al. (2025). Current Progress of Tissue Engineering with Stem Cells in Urology Updated Review in 2025. International Journal of Urology, 32(11):1535-1543.
Publisher | Google Scholor - Žiaran, S., Danišovič, Ľ., Hammer, N. (2025). Tissue engineering and regenerative medicine: advances, controversies, and future directions. Frontiers in Bioengineering and Biotechnology, 13:1568490.
Publisher | Google Scholor - Patel, K. (2025). Cardiac Regeneration: A Promising Future for Tissue Engineering and Cardiac Repair. Columbia Undergraduate Science Journal, 19(1).
Publisher | Google Scholor - Senyange, B., Wesana, J., Van Huylenbroeck, G., Gellynck, X., De Steur, H. (2025). Tissue engineering in the agri-food industry: current status, socio-economic overview and regulatory compliance. Current Opinion in Biotechnology, 91:103228.
Publisher | Google Scholor - Bagherpour, R., Bagherpour, G., Mohammadi, P. (2025). Application of artificial intelligence in tissue engineering. Tissue Engineering Part B: Reviews, 31(1):31-43.
Publisher | Google Scholor - Liu, J., Song, Q., Yin, W., Li, C., An, N., et al. (2025, February). Bioactive scaffolds for tissue engineering: a review of decellularized extracellular matrix applications and innovations. In Exploration, 5(1):20230078.
Publisher | Google Scholor - Sadraei, A., Naghib, S. M. (2025). 4D printing of physical stimuli-responsive hydrogels for localized drug delivery and tissue engineering. Polymer Reviews, 65(1):104-168.
Publisher | Google Scholor - Koeck, K. S., Trossmann, V. T., Scheibel, T. (2025). 3D‐Printed and Recombinant Spider Silk Particle Reinforced Collagen Composite Scaffolds for Soft Tissue Engineering. Advanced Functional Materials, 35(15):2407760.
Publisher | Google Scholor - Wang, H., Zu, Q., Chen, J., Yang, Z., Ahmed, M. A. (2021). Application of artificial intelligence in acute coronary syndrome: a brief literature review. Advances in Therapy, 38(10):5078-5086.
Publisher | Google Scholor - Infante, T., Cavaliere, C., Punzo, B., Grimaldi, V., Salvatore, M., et al. (2021). Radiogenomics and artificial intelligence approaches applied to cardiac computed tomography angiography and cardiac magnetic resonance for precision medicine in coronary heart disease: a systematic review. Circulation: Cardiovascular Imaging, 14(12):1133-1146.
Publisher | Google Scholor - Sotirakos, S., Fouda, B., Mohamed Razif, N. A., Cribben, N., Mulhall, C., et al. (2022). Harnessing Artificial Intelligence in Cardiac rehabilitation: a Systematic Review. Future Cardiology, 18(2):154-164.
Publisher | Google Scholor - Chen, W., Sun, Q., Chen, X., Xie, G., Wu, H., et al. (2021). Deep learning methods for heart sounds classification: A systematic review. Entropy, 23(6):667.
Publisher | Google Scholor - Chan, S., Reddy, V., Myers, B., Thibodeaux, Q., Brownstone, N., et al. (2020). Machine learning in dermatology: current applications, opportunities, and limitations. Dermatology and Therapy, 10(3):365-386.
Publisher | Google Scholor - COVID-19 Pandemic Impact: Global R&D Spend for AI in Healthcare and Pharmaceuticals will Increase US$1.5 Billion By 2025. Medical Letter on the CDC & FDA. May 3, 2020 - Via Gale Academic OneFile.
Publisher | Google Scholor - Sharief, S. A., Chahal, P., Alocilja, E. (2021). Application of DNA sequences in anti-counterfeiting: Current progress and challenges. International Journal of Pharmaceutics, 602:120580.
Publisher | Google Scholor - Ullrich, C., Luescher, A. M., Koch, J., Grass, R. N., Sax, H. (2022). Silica nanoparticles with encapsulated DNA (SPED) to trace the spread of pathogens in healthcare. Antimicrobial Resistance & Infection Control, 11(1):4.
Publisher | Google Scholor - Denimsandjeans. (2020). How soorty is leveraging haelixa DNA to get brands into traceability-Denimandjeans, Global Trends, News and Reports. Worldwide.
Publisher | Google Scholor - Brand Integrity International. (2023). DNA SmartMarks, Brand Integrity.
Publisher | Google Scholor - Holoptica. (2023). DNA Technology.
Publisher | Google Scholor