Review Article
Biomarkers in Early Diagnosis of Alzheimer’s Disease: A Comprehensive Review
- Purohit Saraswati *
Assistant Professor, HOD, Department of Psychiatric Nursing, JSS College of Nursing, Mysuru, Karnataka, India.
*Corresponding Author: Purohit Saraswati, Assistant Professor, HOD, Department of Psychiatric Nursing, JSS College of Nursing, Mysuru, Karnataka, India.
Citation: Saraswati P. (2025). Biomarkers in Early Diagnosis of Alzheimer’s Disease: A Comprehensive Review, International Journal of Biomedical and Clinical Research, BioRes Scientia Publishers. 4(2):1-4. DOI: 10.59657/2997-6103.brs.25.082
Copyright: © 2025 Purohit Saraswati, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: June 19, 2025 | Accepted: July 21, 2025 | Published: July 28, 2025
Abstract
Alzheimer’s disease (AD) is a progressive neurodegenerative disorder characterized by cognitive decline and memory impairment. Early diagnosis remains a critical challenge in managing the disease effectively. Biomarkers have emerged as essential tools in identifying the disease during its preclinical stages, thereby offering opportunities for early therapeutic intervention. This review explores the current landscape of AD biomarkers, including cerebrospinal fluid (CSF) markers, imaging techniques, blood-based biomarkers, and novel genetic indicators. We also discuss their clinical applicability, challenges in implementation, and future directions for biomarker research in Alzheimer's disease.
Keywords: alzheimer’s disease; biomarkers; early diagnosis; cerebrospinal fluid; neuroimaging; blood-based markers
Introduction
Alzheimer’s disease is the most common cause of dementia globally, accounting for 60–80% of cases. The global burden is expected to rise significantly with an aging population. Traditional diagnosis based on clinical symptoms often occurs at later stages when neuronal damage is extensive. Hence, the identification and validation of reliable biomarkers are crucial for early detection, diagnosis, and monitoring of disease progression. The field of Alzheimer’s disease (AD) biomarker research is undergoing a significant transformation, with innovations driving the transition from research settings into routine clinical practice. While traditional CSF and neuroimaging biomarkers remain the benchmark for AD diagnosis, several recent developments are poised to improve the accuracy, accessibility, and predictive capabilities of early diagnostic tools.
One of the most promising innovations is the development of ultrasensitive blood-based biomarker platforms, such as Single Molecule Array (Simoa) and mass spectrometry-based assays, which allow quantification of minute protein concentrations (e.g., plasma p-tau217, Aβ42/Aβ40) with high specificity. These tools have demonstrated comparable diagnostic power to CSF analysis, and ongoing studies are validating their use in primary care screening and longitudinal monitoring.
Another cutting-edge development is the use of extracellular vesicles (EVs) and neuron-derived exosomes (NDEs) from peripheral blood. These vesicles, which cross the blood-brain barrier, carry brain-specific cargo, including phosphorylated tau and Aβ peptides, and provide a novel, non-invasive “liquid biopsy” of the brain.
Artificial Intelligence (AI) and machine learning (ML) algorithms are now being integrated into biomarker analysis and neuroimaging interpretation. AI-based platforms can synthesize data from multiple biomarker modalities (e.g., plasma, imaging, genetics) to generate highly individualized risk profiles. Tools such as the AT(N) classification framework are evolving to incorporate AI-driven scoring systems that adapt dynamically as more patient data becomes available.
Furthermore, researchers are exploring the role of digital biomarkers—including data from wearable sensors, speech analysis, typing patterns, and gait assessment—to detect subtle cognitive and behavioral changes before traditional symptoms appear. These digital signals, combined with biological biomarkers, could enable proactive intervention years before significant neurodegeneration occurs.
Emerging studies are also investigating inflammatory biomarkers (e.g., YKL-40, sTREM2) and metabolomic/lipidomic profiles, which may reflect the neuroimmune environment and cellular metabolism associated with early AD. This opens a new avenue for identifying individuals at risk due to systemic changes rather than only brain-specific pathology.
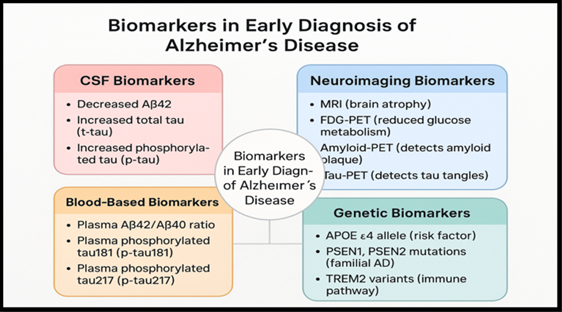
Figure 1: Biomarkers in early identification of Alzheimer’s disease.
Pathophysiology of Alzheimer’s Disease
AD pathology is defined by the accumulation of β-amyloid (Aβ) plaques and tau protein tangles, leading to synaptic dysfunction and neuronal loss. These changes begin years before clinical symptoms appear, underscoring the need for early biomarkers that reflect these underlying pathological processes.
Types of Biomarkers in AD Diagnosis
Cerebrospinal Fluid (CSF) Biomarkers
CSF biomarkers are currently the most validated for AD diagnosis.
- Aβ42: Levels are decreased in CSF due to deposition in brain plaques.
- Total tau (t-tau): Elevated in response to neuronal damage.
- Phosphorylated tau (p-tau): Increases specifically in AD, indicating tau pathology.
Clinical Utility: CSF biomarkers offer high sensitivity and specificity but are invasive due to lumbar puncture requirements.
Neuroimaging Biomarkers
- MRI: Shows brain atrophy, especially in the hippocampus and medial temporal lobe.
- FDG-PET: Detects reduced glucose metabolism in AD-affected brain regions.
- Amyloid-PET and Tau-PET: Directly visualize amyloid plaques and tau tangles.
Advantage: Non-invasive and valuable in visualizing disease progression.
Blood-Based Biomarkers
Recent advances have made blood-based biomarkers a promising area due to ease of collection and cost-effectiveness.
- Plasma Aβ42/Aβ40 ratio: Correlates with brain amyloid burden.
- Plasma p-tau181 and p-tau217: Strong indicators of tau pathology.
- Neurofilament light chain (NfL): Marker of neurodegeneration.
Genetic Biomarkers
APOE ε4 allele: The strongest genetic risk factor for sporadic AD.
TREM2, PSEN1/2 mutations: Associated with familial and early-onset AD.
Clinical Applications and Diagnostic Criteria
Biomarkers are now integrated into diagnostic criteria, such as the NIA-AA research framework, which classifies AD biologically based on amyloid (A), tau (T), and neurodegeneration (N) markers.
Challenges and Limitations
- Standardization: Variability in assays and techniques across labs.
- Access and Cost: Limited availability in low-resource settings.
- Ethical Considerations: Implications of early diagnosis in asymptomatic individuals.
Future Directions
- Multi-modal Biomarker Panels: Integration of blood, imaging, and digital biomarkers.
- Artificial Intelligence: Enhancing biomarker interpretation and predictive modeling.
- Longitudinal Studies: Understanding biomarker evolution over time.
- Point-of-Care Testing: Development of affordable and rapid tests for primary care settings.
Discussion
The early diagnosis of Alzheimer’s disease (AD) is crucial for timely intervention and slowing disease progression. Biomarkers have revolutionized the diagnostic landscape, providing objective measures that reflect underlying pathophysiological changes long before clinical symptoms emerge. Among these, cerebrospinal fluid (CSF) biomarkers—such as Aβ42, total tau (t-tau), and phosphorylated tau (p-tau)-remain the gold standard, offering high diagnostic accuracy. However, their invasive nature limits widespread use, particularly in primary care and low-resource settings.
Neuroimaging techniques such as MRI, FDG-PET, and amyloid-PET have enhanced the ability to detect structural and functional changes in the brain. While these techniques offer valuable insights into disease progression, their high cost and need for specialized infrastructure restrict routine application. The emergence of tau-PET imaging provides even greater specificity, allowing researchers to directly monitor tau pathology in vivo.
The most promising advancement in recent years is the development of blood-based biomarkers. These are minimally invasive, cost-effective, and feasible for large-scale population screening. Plasma p-tau217 and p-tau181 have shown remarkable correlation with CSF and imaging biomarkers, making them viable candidates for future diagnostic algorithms. Additionally, plasma Aβ42/Aβ40 ratio and neurofilament light chain (NfL) serve as robust markers for amyloid pathology and neurodegeneration, respectively.
Genetic markers such as APOE ε4 allele provide risk stratification, although their role is more predictive than diagnostic. Emerging research into polygenic risk scores and rare gene variants (e.g., TREM2) may further refine individualized risk assessments.
Despite these advances, several challenges remain. Standardization across platforms, inter-laboratory variability, and lack of universal thresholds hinder the clinical implementation of biomarkers. Moreover, ethical considerations around early diagnosis—especially in pre-symptomatic individuals—must be carefully addressed, particularly regarding psychological impact and implications for employment or insurance.
In this context, a multi-modal biomarker approach, integrating CSF, imaging, blood-based, and genetic data, may offer the most reliable strategy. Coupled with artificial intelligence and machine learning, such integrated models can improve predictive accuracy and clinical decision-making. Future research must also focus on diverse populations to ensure global applicability, as most current data are derived from Western cohorts.
Conclusion
Biomarkers play an indispensable role in the early diagnosis and management of Alzheimer’s disease. While CSF and imaging markers are well-established, recent advances in blood-based and genetic biomarkers offer hope for less invasive, accessible, and cost-effective diagnosis. Continued research and validation are essential to integrate these tools into routine clinical practice and enable timely intervention. In conclusion, while significant strides have been made, the journey toward universal, accessible, and reliable biomarker-based diagnosis of AD continues. Bridging the gap between research and routine practice will be essential for transforming Alzheimer’s care.
References
- Jack Jr, C. R., Bennett, D. A., Blennow, K., Carrillo, M. C., Dunn, B., et al. (2018). NIA-AA Research Framework: Toward a Biological Definition of Alzheimer's Disease. Alzheimer's & Dementia, 14(4):535-562.
Publisher | Google Scholor - Hansson, O. (2021). Biomarkers for Neurodegenerative Diseases. Nature Medicine, 27(6):954-963.
Publisher | Google Scholor - Nakamura, A., Kaneko, N., Villemagne, V. L., Kato, T., Doecke, J., et al. (2018). High Performance Plasma Amyloid-β Biomarkers for Alzheimer’s Disease. Nature, 554(7691):249-254.
Publisher | Google Scholor - Palmqvist, S., Janelidze, S., Quiroz, Y. T., Zetterberg, H., Lopera, F., et al. (2020). Discriminative Accuracy of Plasma Phospho-tau217 for Alzheimer Disease vs Other Neurodegenerative Disorders. JAMA, 324(8):772-781.
Publisher | Google Scholor - Hampel, H., O’Bryant, S. E., Molinuevo, J. L., Zetterberg, H., Masters, C. L., et al. (2018). Blood-Based Biomarkers for Alzheimer Disease: Mapping the Road to the Clinic. Nature Reviews Neurology, 14(11):639-652.
Publisher | Google Scholor - Mattsson, N., Zetterberg, H., Hansson, O., Andreasen, N., Parnetti, L., et al. (2009). CSF Biomarkers and Incipient Alzheimer Disease in Patients with Mild Cognitive Impairment. JAMA, 302(4):385-393.
Publisher | Google Scholor - Villemagne, V. L. (2016). Amyloid Imaging: Past, Present and Future Perspectives. Ageing Research Reviews, 30:95-106.
Publisher | Google Scholor - Mosconi, L. (2005). Brain Glucose Metabolism in the Early and Specific Diagnosis of Alzheimer’s Disease: FDG-PET Studies in MCI and AD. European Journal of Nuclear Medicine and Molecular Imaging, 32:486-510.
Publisher | Google Scholor - Teunissen, C. E., Verberk, I. M., Thijssen, E. H., Vermunt, L., Hansson, O., et al. (2022). Blood-Based Biomarkers for Alzheimer's Disease: Towards Clinical Implementation. The Lancet Neurology, 21(1):66-77.
Publisher | Google Scholor - Sancesario, G. M., Bernardini, S. (2018). Alzheimer's Disease in the Omics Era. Clinical Biochemistry, 59:9-16.
Publisher | Google Scholor