Case Report
Bending the Boundaries of Lineage: An Uncommon Situation of B/T-Lineage MPAL in an Elderly Patient with BCR-ABL1 p190 Fusion
1 Resident, Section of Hematology & Transfusion Medicine, Department of Pathology and Laboratory Medicine, Aga Khan University, Pakistan.
2 Assistant Professor, Section of Hematology & Transfusion Medicine, Department of Pathology and Laboratory Medicine, Aga Khan University Pakistan.
*Corresponding Author: Muhammad Shayan Ashfaq, Resident, Section of Hematology & Transfusion Medicine, Department of Pathology and Laboratory Medicine, Aga Khan University, Pakistan.
Citation: Muhammad S. Ashfaq, Muhammad H. Hayat. (2026). Bending the Boundaries of Lineage: An Uncommon Situation of B/T-Lineage MPAL in an Elderly Patient with BCR-ABL1 p190 Fusion, Clinical Case Reports and Studies, BioRes Scientia Publishers. 12(3):1-5. DOI: 10.59657/2837-2565.brs.26.314
Copyright: © 2026 Muhammad Shayan Ashfaq, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: March 03, 2026 | Accepted: March 17, 2026 | Published: March 25, 2026
Abstract
Mixed phenotype acute leukemia (MPAL) with both B and T cell features is incredibly rare and tough to pin down. In this form of acute leukemia, the cancerous cells carry markers from more than one blood cell type. MPAL makes up less than 5% of all acute leukemia cases, but it’s more aggressive than the usual types. It often shrugs off standard chemo, which makes things even harder for patients.
Here’s the case: a 77-year-old man came in feeling weak, and his white blood cell count wouldn’t come down. When doctors checked his bone marrow, they found two separate groups of abnormal cells—one with B-cell markers, the other with T-cell markers. Molecular tests showed he had the BCR-ABL1 p190 fusion transcript.
This case really shows why it’s so important to use detailed cell marker testing and molecular analysis for MPAL. Without that, it’s easy to miss or misdiagnose. Getting the diagnosis right matters—it’s the only way to pick the right, targeted treatment for patients facing this rare and stubborn leukemia.
Keywords: leukemia; MAPL; cancer
Introduction
Mixed phenotype acute leukemia, or MPAL, is a rare type of acute leukemia—only about 2% to 5% of cases fall into this category. What makes MPAL different is the way the cancerous cells, or blasts, show antigens from more than one blood cell lineage: myeloid, B-lymphoid, or T-lymphoid [1]. The World Health Organization calls MPAL “acute leukemia of ambiguous lineage.” That’s just a way of saying the blasts don’t fit neatly into one group. Sometimes, you’ll see two separate groups of blasts, each sticking to its own lineage (that’s bilineage leukemia) [2]. Other times, one group of blasts shows markers from multiple lineages (biphenotypic leukemia). And sometimes, you get a mix of both [3]. We almost never see both B- and T-lineage antigens together, either in the same group of blasts or in separate ones [4]. This particular B/T lineage version of MPAL pops up so rarely in medical literature that we don’t know much about how it looks in patients, how it behaves, or what it means for prognosis [5].
Diagnosing MPAL—especially the B/T cell kind—relies heavily on detailed immunophenotyping using flow cytometry [6]. Molecular testing helps too, especially when you’re looking for genetic quirks like the BCR-ABL1 fusion gene. The p190 version of this gene, which comes from the e1a2 transcript, usually shows up in acute lymphoblastic leukemia and is rarely found in MPAL [7].
Here, we describe a rare case of MPAL in an older man, where blasts from both B- and T-cell lineages appeared. Flow cytometry picked up two separate groups of lymphoblasts, and molecular tests confirmed the presence of the BCR-ABL1 p190 transcript. Cases like this really drive home how tricky MPAL can be to diagnose and why it’s so important to use both immunophenotypic and molecular tools to get the full picture [1].
Case Presentation
Mixed phenotype acute leukemia (MPAL) with both B and T cell features isn’t something you see every day. Here’s what happened with this patient: a 77-year-old man, already dealing with diabetes, high blood pressure, osteoarthritis, and an enlarged prostate, came in after a month of just feeling weak all over and fighting off cellulitis in his left leg.
When the doctors ran bloodwork, he turned out to be anemic and had high white blood cell counts, but his platelets were fine. The cellulitis cleared up once he started antibiotics, but the high white count stuck around. A closer look at his peripheral blood smear showed a leukoerythroblastic pattern with extra basophils and eosinophils, and some atypical mononuclear cells. On examination, there were no swollen lymph nodes, and his liver and spleen felt normal.
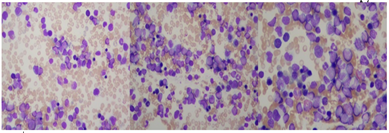
Bone marrow aspiration told more of the story: about 33%of the cells were blasts—small to medium, big nuclei, not much cytoplasm, fine chromatin, and the nucleoli were hard to spot. The normal blood cell production was clearly suppressed. (Figure:1)
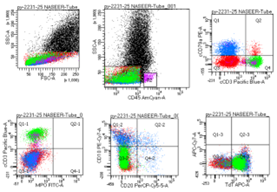
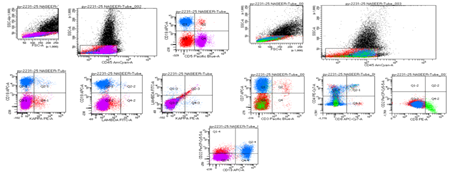
They ran flow cytometry on the bone marrow. It picked up two different blast populations based on CD45 levels and side scatter. One group (22%) had dim CD45 and showed T-cell markers: cytoplasmic CD3, CD5, CD7, dim CD4, CD34, and TdT. The other group (19%) also had dim CD45, but these cells had B-cell markers: CD19, CD22, cytoplasmic CD79a, CD10, TdT, CD34, HLA-DR, CD9, CD38, and CD58. (Figure:2)
On top of that, molecular tests found the BCR-ABL1 p190 fusion transcript (the e1a2 variant), which is the minor breakpoint cluster. All of this pointed to an MPAL, B/T cell type, with the BCR-ABL1 p190 fusion. The team moved fast and started him on intensive chemotherapy.
Figure 1: Bone marrow aspirate cytology showed that primitive immature lymphocytes.
Figure 2(a): B/T lymphoid MPAL: The blasts are CD 45 negative and expressed Stem cell marker TdT; B cell markers cCD79a and T cell markers cCD3
Figure 2(b): B/T lymphoid MPAL: The blasts are CD 45 negative and expressed Stem cell marker TdT, CD34; B cell markers CD19 and T cell markers cCD3
Figure 2(c): B/T lymphoid MPAL: The blasts are CD 45 negative and expressed; B cell markers cCD79a, CD19, CD22 and T cell markers cCD3, CD7, dim CD4.
Discussion
Mixed-phenotype acute leukemia (MPAL) remains a diagnostically challenging and biologically heterogeneous entity [8]. The current WHO/ICC frameworks explicitly recognize MPAL (acute leukemia of ambiguous lineage) as a group in which lineage assignment requires integrated interpretation of immunophenotype, cytogenetics, and molecular findings — and they single out MPAL with defining genetic abnormalities (for example, BCR-ABL1) as discrete, clinically important subtypes [9]. This modern, genetics-forward classification underscores why the finding of BCR-ABL1 in a case of ambiguous lineage should drive both diagnostic labeling and therapeutic planning [10].
Our patient’s marrow demonstrated two discrete lymphoid blast populations — one T-lineage and one B-lineage — meeting bilineage MPAL criteria. B/T-lineage MPAL (either biphenotypic or bilineage with separate populations) is very uncommon; most reported MPALs are myeloid plus lymphoid (B/myeloid or T/myeloid) [11].
The BCR::ABL1 fusion in MPAL represents less than 0.5% of all acute leukemia cases and occurs in 15% to 20% of MPAL cases, making it the most prevalent cytogenetic alteration found in MPAL.(12) Most of these transcripts arise from chromosomal breakpoints located in BCR introns 1, 13, or 14 and in ABL1 intron 1 (identified as e1a2, e13a2, or e14a2, respectively). Different BCR::ABL1 fusion proteins, including p210, p190, p230, and atypical forms, can be produced depending on the precise breakpoints; MPAL patients are more likely to have the p190 variant of BCR::ABL1 than the p210 variant [13]. The cytogenetic and molecular abnormalities of MPAL with atypical BCR::ABL1 are extremely poorly studied [14]. There is no prior documentation of the atypical BCR::ABL1 e13a3 fusion found in our case. Exon e13 (b2) from the BCR gene and exon a3 from the ABL1 gene directly fuse to produce the e13a3 transcript, which results in the ABL1 gene's exon a2 being omitted. The SH3 domain, which is encoded by this exon and is essential for the negative regulation of BCR::ABL1 kinase activity, suggests that the e13a3 transcript may modify BCR::ABL1 mediated signaling pathways [15]. The absence of the SH3 domain may have an impact on downstream signaling pathways, like the STAT5 pathway, which may then have an impact on leukemic cells' proliferative behavior. According to recent research, this transcript may affect molecular remission rates, the response to tyrosine kinase inhibitors, and the possibility of treatment-free remission following therapy discontinuation [16].
Currently, there is no established consensus on treating Philadelphia chromosome-positive (Ph+ MPAL), and reports on cases with the atypical e13a3 BCR::ABL1 fusion are especially rare. Treatment for MPAL with BCR::ABL1 typically includes tyrosine kinase inhibitors combined with ALL chemotherapy, followed by allogeneic stem cell transplantation when feasible.(15) Studies show that MPAL patients with the Ph+ chromosome generally have a poor prognosis, with a median survival of 8 months compared to 139 months for those with normal karyotypes. Ph+ MPAL displays features of both acute lymphoblastic leukemia (ALL) and AML, so leukemia cells may not respond fully to regimens targeting only ALL or AML [17].
There are currently few systematic studies on the transcriptome and genomic features of Ph + MPAL [18]. The presence of mutations in IKZF1, CUX1, and BCORL1, each of which has definite or potential clinical significance, suggests a poor prognosis in this case [19].
Conclusion
To sum up, accurate diagnosis and categorization of MPAL are crucial. To enable accurate clinical therapy and prognosis assessment, thorough investigation utilizing a variety of techniques, including morphology, immunophenotype, genetics, and molecular biology, is crucial. These finding provides fresh insights into the diagnosis and treatment of Ph + MPAL.
Declarations
Conflict of Interest
None
Patient’s Consent
Telegraphic Informed Consent Was Obtained from The Patient.
Competing Interest
The Authors Declared No Conflict Interest.
Author’s Contribution
Ms, Mh: Contributed to The Design, Drafting and Critical Revision of The Manuscript.
All Authors Approved the Final Version of The Manuscript to Be Published.
Disclaimer (Artificial Intelligence)
Author(S) Hereby Declare That No Generative Ai Technologies Such as Large Language Models (Chatgpt, Copilot, Etc.) And Text-To-Image Generators Have Been Used During the Writing or Editing of This Manuscript.
References
- George, B. S., Yohannan, B., Gonzalez, A., & Rios, A. (2022). Mixed-phenotype acute leukemia: Clinical diagnosis and therapeutic strategies. Biomedicines, 10(8):1974.
Publisher | Google Scholor - Béné, M. C., & Porwit, A. (2012). Acute leukemias of ambiguous lineage. Seminars in Diagnostic Pathology, 29(1):12–18.
Publisher | Google Scholor - Campo, E., Swerdlow, S. H., Harris, N. L., Pileri, S., Stein, H., & Jaffe, E. S. (2011). The 2008 WHO classification of lymphoid neoplasms and beyond: Evolving concepts and practical applications. Blood, 117(19):5019–5032.
Publisher | Google Scholor - Steensma, D. P. (2011). Oddballs: Acute leukemias of mixed phenotype and ambiguous origin. Hematology/Oncology Clinics of North America, 25(6):1235–1253.
Publisher | Google Scholor - Kim, H. J. (2016). Mixed-phenotype acute leukemia (MPAL) and beyond. Blood Research, 51(4):215–216.
Publisher | Google Scholor - Mishra, D. N., Singh, G., Meher, D., Jena, R. K., & Mohanty, D. P. (2025). Flow cytometric immunophenotyping of mixed phenotype acute leukemia in a tertiary care hospital of Eastern Odisha. International Journal of Hematology-Oncology and Stem Cell Research, 19(1):29–36.
Publisher | Google Scholor - Lingzhi, Y., Nana, P., Mingqing, Z., Aining, S., Yongquan, X., Changgeng, R., et al. (2012). Clinical, immunophenotypic, cytogenetic, and molecular genetic features in 117 adult patients with mixed-phenotype acute leukemia defined by WHO-2008 classification. Haematologica, 97(11):1708–1712.
Publisher | Google Scholor - Porwit, A., & Béné, M. C. (2015). Acute leukemias of ambiguous origin. American Journal of Clinical Pathology, 144(3):361–376.
Publisher | Google Scholor - Mulet-Lazaro, R., van Herk, S., Nuetzel, M., Sijs-Szabo, A., Díaz, N., Kelly, K., et al. (2024). Epigenetic alterations affecting hematopoietic regulatory networks as drivers of mixed myeloid/lymphoid leukemia. Nature Communications, 15(1):5693.
Publisher | Google Scholor - Kim, R., & Clappier, E. (2024). Uncovering new layers of Ph+ ALL biology. Blood, 143(14):1322–1323.
Publisher | Google Scholor - Porwit, A., & Béné, M. C. (2019). Multiparameter flow cytometry applications in the diagnosis of mixed phenotype acute leukemia. Cytometry Part B: Clinical Cytometry, 96(3):183–194.
Publisher | Google Scholor - Li, S., Ilaria, R. L., Jr., Million, R. P., Daley, G. Q., & Van Etten, R. A. (1999). The P190, P210, and P230 forms of the BCR/ABL oncogene induce a similar chronic myeloid leukemia-like syndrome in mice but have different lymphoid leukemogenic activity. Journal of Experimental Medicine, 189(9):1399–1412.
Publisher | Google Scholor - Score, J., Calasanz, M., Ottman, O., Pane, F., Yeh, R. F., Sobrinho-Simões, M. A., et al. (2010). Analysis of genomic breakpoints in p190 and p210 BCR–ABL indicate distinct mechanisms of formation. Leukemia, 24:1742–1750.
Publisher | Google Scholor - Kandeel, E. Z., Hassan, N. M., & El Ashry, M. S. (2023). Clinical and molecular characteristics of patients with mixed phenotype acute leukemia. Asian Pacific Journal of Cancer Prevention, 24:1217–1223.
Publisher | Google Scholor - Jean, J., Sukhanova, M., Dittmann, D., Gao, J., & Jennings, L. J. (2024). A novel BCR: ABL1 variant detected with multiple testing modalities. Case Reports in Hematology, 2024:8486267.
Publisher | Google Scholor - Su, Y. J., Kuo, M. C., Chen, T. Y., Wang, M. C., Yang, Y., Ma, M. C., et al. (2022). Comparison of molecular responses and outcomes between BCR: ABL1 e14a2 and e13a2 transcripts in chronic myeloid leukemia. Cancer Science, 113(10):3518–3527.
Publisher | Google Scholor - George, B. S., Yohannan, B., Gonzalez, A., & Rios, A. (2022). Mixed-phenotype acute leukemia: Clinical diagnosis and therapeutic strategies. Biomedicines, 10(8):1974.
Publisher | Google Scholor - Shi, S., Zhou, Q., Zhao, D., Zarif, M., Wei, C., Sibai, H., et al. (2025). Molecular genetic characterization of mixed-phenotype acute leukemia (MPAL) with BCR: ABL1 fusion. Leukemia Research, 151:107665.
Publisher | Google Scholor - Georgopoulos, K., Bigby, M., Wang, J. H., Molnar, A., Wu, P., Winandy, S., et al. (1994). The Ikaros gene is required for the development of all lymphoid lineages. Cell, 79(1):143–156.
Publisher | Google Scholor