Research Article
Antioxidant Testing Using DPPH for Dadap Leaves Through Without Extraction Stage
- Muhammad Roy Asrori 1*
- Mailinda Ayu Hana Margaretha 1
- Siti Khoirunnisa 3
- Achmad Dzulfikri Almufti Asyhar 4
- Suharti Suharti 1,2
- Surjani Wonorahardjo 1,2
1 Laboratory of Minerals and Advanced Materials, Faculty of Mathematics and Natural Sciences, Universitas Negeri Malang, Indonesia.
2 Department of Chemistry, Faculty of Mathematics and Natural Sciences, Universitas Negeri Malang, Indonesia.
3 Department of Pharmacy, Universitas Muhammadiyah Yogyakarta, Jl. Brawijaya, Bantul, Yogyakarta, Indonesia.
4 MTsN 1 Malang, Jl. Basuki Rachmat No.194, Indonesia.
*Corresponding Author: Muhammad Roy Asrori, Laboratory of Minerals and Advanced Materials, Faculty of Mathematics and Natural Sciences, Universitas Negeri Malang, Indonesia.
Citation: Muhammad R. Asrori, Margaretha M.A.H., Khoirunnisa S., Asyhar A.D.A., Suharti S., et al. (2025). Antioxidant Testing Using DPPH for Dadap Leaves Through Without Extraction Stage, Journal of BioMed Research and Reports, BioRes Scientia Publishers. 8(4):1-3. DOI: 10.59657/2837-4681.brs.25.195
Copyright: © 2025 Muhammad Roy Asrori, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: August 12, 2025 | Accepted: August 26, 2025 | Published: September 03, 2025
Abstract
Antioxidant testing is widely needed in health aspects. Knowledge of antioxidant values can help in the selection and development of food products. The DPPH method is widely used because of its ease and efficiency. For plant samples, initial treatment is needed to ensure the accuracy of the solution concentration. Many plant samples must be extracted first. This process is time consuming and has mass validity issues. This study aims to: to evaluate a DPPH antioxidant test method without extraction that is efficient, effective, practical, and environmentally friendly, (2) to analyze the validity of the developed method. The result revealed that the IC50 value was obtained about 1028.85 ppm. The linearity and precision were 0.98 and 0.02% respectively. This method proved that without extraction stage, the IC50 can be determined.
Keywords: DPPH; antioxidant; testing; dadap leaves
Introduction
Food plays an important role in body health. Body health can be attacked by free radicals in the body that can trigger serious diseases [1]. Meanwhile, free radicals can be stopped by antioxidant compounds. These compounds are contained in food at certain levels. These levels are analyzed by antioxidant test results [2]. Quantitative analysis of antioxidants aims to obtain a value of the content that states how much its concentration is. Antioxidant testing methods include = DPPH, FRAP, ABS, etc. Many of these test applications are for health, food, and beverage products [3]. Antioxidant test using the DPPH method. DPPH has a chemical formula, namely 2,2-diphenyl-1-picrylhydrazyl. DPPH is used as a model of free radical compounds. The interaction of DPPH (a free radical model) with antioxidants produces neutral and safe products. This method has been widely used in previous studies [4].
Many studies before the DPPH test must prepare samples in the form of extraction [5]. The extraction carried out also varies in methods from the use of appropriate solvents, time, tools used, and other technical parameters (pressure, temperature, humidity). Then, sample preparation is made with a concentration variation with a minimum of 4-5 concentration variants to create a linear regression equation. The equation is important for determining the IC50 [6]. This study offers a DPPH test without the extraction stage for sample preparation. Without any extraction stage, this DPPH test will be analyzed for its validation. The development of this test will save a lot of time, materials, tools, and support the reduction of environmental impacts. In this study, Dadap leave sample were taken as a model of materials containing antioxidant compounds. The objectives of this study are: (1) to evaluate a DPPH antioxidant test method without extraction that is efficient, effective, practical, and environmentally friendly, (2) to analyze the validity of the developed method.
Materials and Methods
The tools used were a 10 mL measuring flask, stirrer, analytical balance (Kern), incubator (memmert), test tube (iwaki), test tube rack, dropper, 5 mL-10 mL measuring cup, Erlenmeyer flask, weighing paper, and label paper. The materials used were ascorbic acid (p.a.), DPPH (p.a., Himedia), methanol (Smartlab), Dadap leave from Malang. The instrument used was a UV-Vis spectrophotometer (Thermo Scientific, Evolution 201).
Antioxidant test with Dadap leaves without extraction stage
There are two types of sample preparation, namely sample preparation from extraction results and sample preparation without extraction. Both will be compared and become the subject of discussion. Sample preparation is made 1000 ppm by: mixing 10 mg of sample with methanol solvent in a 10 mL measuring flask. Specifically, for samples without extraction, the sample mass is obtained from the following formula:

Note: m = mass of sample required to equivalent 10 mg of extracted sample, w = mass is expected to be 1000 ppm from sample preparation, 100 = multiplying factor, %yield = percent yield of samples obtained from literature/extraction results. After making a 1000 ppm solution, concentration variations were made by dilution to 30, 60, 90, 120, 150 ppm.
Next, the preparation of the positive control solution (ascorbic acid) was made 100 ppm by: mixing 1 mg with methanol solvent in a 10 mL measuring flask. Then, the positive control solution was made with concentration variations of 4, 8, 12, 16, 20 ppm through dilution. Then, the preparation of the DPPH solution was prepared with a concentration of 0.76 mM (300 ppm) which was made by: 3 mg of powder with methanol solvent in a 10 mL measuring flask.
The mixture and incubation were carried out at room temperature, and the absorbance reading was carried out with a UV-Vis spectrophotometer instrument at a wavelength of 517 nm. Furthermore, the results of the UV-Vis data were in the form of absorbance values. Absorbance data from various concentration variations were used to calculate the % inhibition with the formula:
 (Equation 1)
(Equation 1)
Note: Abs = absorbance of instrument
Then, a graph is made with the x-axis (concentration) and the y-axis (% inhibition). Finally, the IC50 value is calculated using the linear regression equation obtained from the graph.
Results and Discussion
Antioxidant testing was carried out in a laboratory. After absorbance measurement, the analysis is used to result the % inhibition value. The results can be seen in Table 1.
Table 1: Results of the absorbance and the % inhibition
| Concentration (ppm) | Abs. (1) | Abs. (2) | Abs. (3) | Average | Abs, negative control | Abs, sample control | Average of % Inhibition |
| 250 | 1.705 | 1.707 | 1.708 | 1.707 | 1.956 | 0.103 | 18.00 |
| 200 | 1.747 | 1.749 | 1.752 | 1.749 | 1.956 | 0.103 | 15.85 |
| 150 | 1.779 | 1.771 | 1.774 | 1.775 | 1.956 | 0.103 | 14.52 |
| 100 | 1.838 | 1.837 | 1.826 | 1.834 | 1.956 | 0.103 | 11.50 |
| 50 | 1.86 | 1.863 | 1.862 | 1.862 | 1.956 | 0.103 | 10.07 |
Note: Abs = absorbance
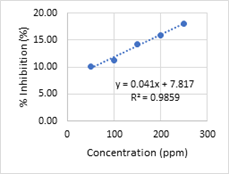
Subsequently, the linear regression curve between % inhibition and concentration can be seen in Figure 1.
Figure 1: Linear regression curve of first antioxidant testing of Dadap leaves
Based on Figure 1, the equation is y = 0.041x + 7.817 with R2 equals to 0.9859. the R2 value shows linearity because of the acceptable linearity is R2 ≥ 0.98 (7). Therefore, the IC50 of Dadap leaves is 1028.85 ppm. The value was categorized as low level of antioxidant activity. Furthermore, the precision of this method by RSD (Relative Standard Deviation) is 0.02 percentage based on Table 2. the precision is acceptable because of less than 2% limit [8]. Therefore, antioxidant of a sample without extraction stage can be tested by DPPH method.
Conclusion
Based on the results, the method of this study was efficient and effective. The IC50 obtained based on the calculation is 1028.85 ppm. This method was acceptable since the linearity and precision were acceptable. Therefore, without extraction stage, the antioxidant testing can be carried out. The future challenge is how to evaluate the free-extraction sample using other methods such as FIC, FRAP, and ABTS.
Acknowledgement
Thanks to Laboratory of Minerals and Advanced Materials for supporting this study.
References
- Chandimali, N., Bak, S. G., Park, E. H., Lim, H.-J., Won, Y.-S., Kim, E.-K., et al. (2025). Free radicals and their impact on health and antioxidant defenses: A review. Cell Death Discovery, 11(1):19.
Publisher | Google Scholor - Mendonça, J. D., Guimarães, R. D., Zorgetto-Pinheiro, V. A., Fernandes, C. D., Marcelino, G., Bogo, D., et al. (2022). Natural antioxidant evaluation: A review of detection methods. Molecules, 27(11):3563.
Publisher | Google Scholor - Neffe-Skocińska, K., Karbowiak, M., Kruk, M., Kołożyn-Krajewska, D., & Zielińska, D. (2023). Polyphenol and antioxidant properties of food obtained by the activity of acetic acid bacteria (AAB): A systematic review. Journal of Functional Foods, 107:105691.
Publisher | Google Scholor - Munteanu, I. G., & Apetrei, C. (2021). Analytical methods used in determining antioxidant activity: A review. International Journal of Molecular Sciences, 22(7):3380.
Publisher | Google Scholor - Zhang, L., Li, J., Huo, Y., Yang, W., Chen, J., Gao, Z., et al. (2024). Ultrasonic extraction and antioxidant evaluation of oat saponins. Ultrasonics Sonochemistry, 109:106989.
Publisher | Google Scholor - Reviana, R., Usman, A. N., Raya, I., Aliyah, Dirpan, A., Arsyad, A., et al. (2021). Analysis of antioxidant activity on cocktail honey products as female pre-conception supplements. Gaceta Sanitaria, 35:S202–S205.
Publisher | Google Scholor - Ferenczi-Fodor, K., Végh, Z., Nagy-Turák, A., Renger, B., & Zeller, M. (2001). Validation and quality assurance of planar chromatographic procedures in pharmaceutical analysis. Journal of AOAC International, 84(4):1265–1276.
Publisher | Google Scholor - Ermer, J., & Ploss, H.-J. (2005). Validation in pharmaceutical analysis: Part II: Central importance of precision to establish acceptance criteria and for verifying and improving the quality of analytical data. Journal of Pharmaceutical and Biomedical Analysis, 37(5):859–870.
Publisher | Google Scholor - Dzufakar, A. B. P., Wahyuningsih, C., Khotijah, P. R. S., Maharani, N. A., Dion, R., Putri, F. N., et al. (2023). Antioxidant potential of dadap serep (Erythrina subumbrans) leaves extract from Semarang, Central Java to prevent acne. AIP Conference Proceedings, 2738(1):040035.
Publisher | Google Scholor - Gulcin, İ., & Alwasel, S. H. (2023). DPPH radical scavenging assay. Processes, 11:2248.
Publisher | Google Scholor