Research Article
Antibiotic-Related Microbiome Disruption as a Modifiable Driver of NAFLD: Pathogenesis and Therapeutic Perspectives
1 Assistant Professor, Department of Pharmacy Practice, Thanthai Roever College of Pharmacy, Tamil Nadu, India.
2 Doctor of Pharmacy Student, Thanthai Roever College of Pharmacy, Perambalur, Tamil Nadu, India.
3 Principal, Thanthai Roever College of Pharmacy, Perambalur, Tamil Nadu, India.
*Corresponding Author: Mahavidhya R, Doctor of Pharmacy Student, Thanthai Roever College of Pharmacy, Perambalur, Tamil Nadu, India.
Citation: Ramesh K., Swathi G., Mahavidhya R., Aishwar J. P., Nepolean R. [2026]. Antibiotic- Related Microbiome Disruption as a Modifiable Driver of NAFLD: Pathogenesis and Therapeutic Perspectives, Journal of BioMed Research and Reports, BioRes Scientia Publishers. 10(3):1-11. DOI: 10.59657/2837-4681.brs.26.235
Copyright: © 2026 Mahavidhya R, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: February 10, 2026 | Accepted: February 24, 2026 | Published: March 09, 2026
Abstract
Non-alcoholic fatty liver disease (NAFLD) is the most common chronic liver disease globally, and its incidence is increasing even among people who do not have typical metabolic risk factors. While NAFLD is closely associated to obesity, insulin resistance, and type 2 diabetes, recent research points to the gut microbiota and its disturbance, specifically through antibiotic usage, as a major factor to disease initiation and progression. The gut microbiota controls immune activity, food metabolism, and intestinal barrier integrity. Antibiotics, particularly broad-spectrum drugs, can disturb microbial diversity, deplete beneficial bacteria, and cause dysbiosis. This imbalance increases gut permeability, allowing bacterial endotoxins like lipopolysaccharide [LPS] to enter the portal circulation, triggering hepatic immune responses and promoting inflammation, steatosis, and fibrosis. Therapeutic techniques that target the gut-liver axis are gaining interest. Probiotics and prebiotics have been demonstrated to help restore microbial balance, enhance liver enzymes, and decrease inflammation. Faecal microbiota transplantation [FMT], as well as fibre and polyphenol-rich dietary treatments, have the potential to improve gut health and metabolic outcomes. Additionally, gut-targeted pharmacologic treatments such as FXR and TGR5 agonists are being investigated. Clinical pharmacists play an important role in the management of antibiotics, assuring reasonable prescribing practices and promoting microbiome preservation initiatives. In conclusion, antibiotic-induced gut dysbiosis is a modifiable risk factor for NAFLD. Microbiota-targeted medicines are a potential approach to prevention and treatment. Future research should concentrate on personalized microbiome-informed methods and robust clinical trials to demonstrate their effectiveness in improving liver health.
Keywords: non-alcoholic fatty liver disease; gut microbiota; antibiotic use; gut-liver axis; dysbiosis; intestinal permeability; probiotics; clinical pharmacist; NAFLD treatment
Introduction
Nonalcoholic fatty liver disease (NAFLD) is the most common cause of chornic liver disease, affecting 14-30% of people around the world [1, 2]. It includes a lot of different condition, like simple steatosis, non –alcoholic steatohepatitis [NASH], progressive fibrosis, and finally cirrhosis [3], NAFLD is strongly linked to metabolic syndrome, obesity, insulin resistance, and type 2 diabetes, which is why it’s becoming a major health issue around the world [4]. There are many things that can cause NAFLD, such as oxidative stress, mitochondrial dysfunction, lipotoxicity, insulin resistance and inflammatory reaction. The "multiple-hit” hypothesis says that disease progression happens when multiple insults happen at the same time on people who are genetically predisposed to them [5]. Non-invasive imaging and liver function tests are commonly used to diagnose NAFLD, but liver biopsy is still the best way to stage the disease and tell it apart from NASH [6].
Globally, NAFLD prevalence and mortality rates have increased dramatically. Between 1990 to 2019 disease rate rised by 95.4% with the global prevalence rising from from 10.5% to16.0% [7, 8]. The highest disease burden, including disability-adjusted life years, is found in populations over 50 and in regions with lower socio-demographic indices [9], particularly in asia accounts for the greatest number of NADLD cases [8]. Futher more, NAFLD is not limited to hepatic manifestations, it’s also a risk factor for cardiovascular disease and extra-hepatic cancer [2].
Recent research has focused on the gut microbiota as a major player in NAFLD pathophysiology. The human gut contains trillions of microorganisms, primarily from the Bacteroidetes and Firmicutes phyle, which are required for immune modulation, nutrients metabolisum and intestinal barrier integrity [10, 11]. Environmental exposures, age, diet and pharmacologic agents all influences the composition of the gut microbiota, which changes over time [12]. Dysbiosis or disruption of microbial hemoostasis, is becoming more widely recognized in diabetes mellitus, obesity, cardiovascular disease, and neuropsychiatric disorders [13]. In addition, the gut –liver axis, a bidirectional communication system mediated by portal circulation and bile acids [14]. This axis is crucial for liver homeostasis and disease progression [15], intestinal dysbiosis and increased intestinal permeability have been linked to a variety of liver disease, including NAFLD and cirrhosis [16]. The translocation of gut- derived bacteria, metabolites and other products can cause fibrosis and liver inflammation by activating immmue responses [17].
Antibiotics, while life-saving, can significantly modifying the composition and diversity of the gut microbiota, potentially leading to various health problem [18]. Antibiotic have a significant impact on gut microbiota [19]. These changes are influenced by antibiotic class, administration routes, dosage, and host –related factors [20], all of which can affect the development and progression of NAFLD. Excessive antibiotic use can potentially contribute to the onset of NAFLD [19]. Chronic antibiotic use has been shown in studies to suppress gut bacteria, reduce liver inflammation, lower portal secondary bile acid levels and fibrosis in NAFLD in mouse models [21]. However, antibiotics effects on NAFLD are mixed, with some research reporting both positive and negative outcome [22]. Probiotic, prebiotics, dietary changes and medication associated comorbidities (eg. metformin, GLP-1 agonists) have shown promise in modulating gut microbiota and improving NAFLD [23].
Recent research emphasises how important gut microbiota dysbiosis is to the pathophysiology of non-alcoholic fatty liver disease (NAFLD), as microbial imbalance can cause inflammation, increase intestinal permeability, and interfere with metabolism [24]. One important mechanism connecting dysbiosis and liver damage is the gut-liver axis [25]. To restore microbial balance, therapeutic approaches like faecal microbiota transplantation, probiotics, prebiotics, and antibiotics are being researched [26]. Still, additional human research is required to validate their clinical relevance, even in light of encouraging outcomes in animal models [24].
This review aims to explore the relationship between antibiotics, disruption of the gut microbiota, and the pathogenesis of NAFLD, with a focus on potential therapeutic interventions targeting the gut-liver axis. Given the increasing global burden of NAFLD and the widespread use of antibiotics, it is becoming more and more clinically important to understand how antibiotic-induced dysbiosis contributes to liver disease.
Gut Microbiotab: Composition and Fuction
The human gut microbiota, which is primarily made up of the Bacteroidetes and firmicutes phyla is essential for maintaining host health through a variety of mechanisms such as protection against pathogenic microbes, immune regulation, nutrients metabolism [27]. A variety of factors influence the composition og the gut microbiota, including antibiotic use, host genetic background, mode of delivery birth, dietary habits [28]. Researches have gained deeper insights into the gut’s microbial diversity and its dynamic interaction with the human host as advanced sequencing technologies have become available [29].
When compared to healthy individuals, NAFLD patients have significantly lower microbial diversity and richness [30]. This decline in microbial diversity is frequently accompanied by increased intestinal inflammation and impaired mucosal immune function, which contribute to disease progression [31]. Other than associatios, the gut microbiota influences heaith outcome by assisting with nutrients digestion and absorption and modulating immune, producing essential metabolites and modulating immune response [32].
One of the primary metaolic output of the gut microbiota is the fermentation of dietary fibres ton produces short-chain-fatty acids [SCFDs], primarily acetate, propionate and butyrate [34]. These SCFA s provide vital energy to intestinal epithelial cells, aid in gut motitlity, strengthen the intestinal barrier, and control cellular processes such as profileration and differentiation [35, 36].
Futhermore, SCAFs have inmmunomodulatory properties that help maintain intestinal homeostasis and prevent systemic inflammation [36]. The gut microbiota also contributes to the processing of dietary carbohydrates, proteins, and polyphenols and it is known to influence host gene expression involved in metabolism and nutrient uptake, which may directly affecr adiposity and metabolic outcomes [32, 33].
The gut –liver is an important pathway in liver physiology and pathology, with bi-directional communication via the portal vein, billiary tract, and systemic circulation [37]. Disruption of gut microbial balance, such as from chronic alcohol consumption or other environmental factors, can liver [38]. These gut –derived substances, which include SCFAs, secondary bile acids and bacterial cell wall elements have the potential to disrupts liver metabolism, promote inflammation and contributes to the onset and progression of liver disease such as NAFLD and alcoholic liver disease [39]. Also, the gut microbiota modulates liver inflammation, induces DNA damage and alters the tumour microenvironment [40].
Antibiotics And Gut Dysbiosis
Dysbiosis a term increasingly used in microbiome research, refers to alterations in the gut microbiota associated with disease states [41]. Dysbiosis has been linked to chronic and metabolic disease, in addition to gastrointestinal symptoms like diarrhea and abdominal pain [42]. There is no standard definition for the concept, which makes diagnosis and treatment difficult, Researchers have attempted to refine the definition via concept analysis emphasizing risk factors and outcomes for broad nursing practices [43].
Antibiotics commonly prescribed in primary care can significantly disrupt the gut microbiota resulting in decreased bacterial diversity and altered species abundances [44]. Clindamycin fluoroquinolones composition, while penicillin V and nitrofurantoin have minimal impact [45]. Beta-lactams, glycopeptides and macrolides can deplete beneficial Bifidobacterium and Lactobacillius bacteria [46]. Macrolides and tetracyclines, which were previously thought to be bacteriostatics have species-specific killing activity against a variety of commensal bacteria, which could explain their significant impact on gut microbiomes [47]. Although gut bacteria typically recover within weeks of antibiotic discontinuation, some studies suggest lasting up to 6 months [44].
The gut microbiome may take weeks or months to recover after treatment [44]. Persistent dysbiosis can encourage the growth of potentially pathogenic bacteria like Enterobacteria cease promoting horizontal gene transfer and the spread of virulence and antibiotics resistances genes [48, 49]. High microbial diversity acts as a natural barrier to resistances spread, but modern lifestyle factors, particularly antimicrobial over use, have contributed to long-term microbial loss, increased vulnerability to infection, and potentially rising disease prevalence in industrialized countries [50, 51].
Duration And Frequency of Antibiotic Use as Risk Factors
A large-scale investigation by Ebrahimi et al. [2023] involving 2, 584 histologically verified NAFLD patients and 12, 646 matched controls found an important association between antibiotic use and the development of NAFLD. The study found that any prior antibiotic usage was associated with a 35% increased risk of NAFLD [adjusted odds ratio (aOR) 1.35; 95% CI 1.21-1.51]. Short-term antibiotic use [≤7 days] was associated with a greater risk (aOR 1.63), while longer exposure [≥43 specified daily doses, over six weeks] increased the chances to 1.72.. The frequency of antibiotic dispensation exhibited a dose-response association, with an aOR of 1.30 for a single dispensation, 1.38 for two dispensations, and 1.39 for three or more, with a significant trend (p<0.001). Importantly, a time-lagged study revealed that this connection was still strong even when antibiotic usage occurred three to five years before the NAFLD diagnosis. The relationship was constant across histological phases, including simple steatosis, NASH, and fibrosis [52].
The Swedish Histology Substudy (2007-2017) indicated that NAFLD patients were more likely to have used antibiotics before. Fluoroquinolones had the strongest link (aOR 1.38). Interestingly, this link was much greater in persons without metabolic syndrome (aOR 1.63) than in those with metabolic syndrome (aOR 1.09), indicating that antibiotics may contribute to NAFLD development in metabolically healthy individuals [54].
Tarantino and Citro's (2024) review validated the relationship between antibiotic usage and NAFLD, particularly associations with gut barrier disturbance and microbiota alterations [54]. In Table 1, the key risk factors associated with NAFLD are outlined and impact on NAFLD Risk increase the likelihood of disease development and progression.
Table 1: Risk Factors for NAFLD and Their Relative Impact on Disease Development
| Risk Factor | Impact on NAFLD Risk |
| Any antibiotic use | ↑ ~35 % [aOR 1.35] |
| 1–≤7 days of antibiotics | aOR 1.63 |
| >6 weeks exposure | aOR 1.72 |
| ≥3 antibiotic courses | aOR ~1.39 |
| Fluoroquinolone courses | aOR ~1.38 |
| Without metabolic syndrome | aOR ~1.63 |
Link Between Gut Dysbiosis and NAFLD
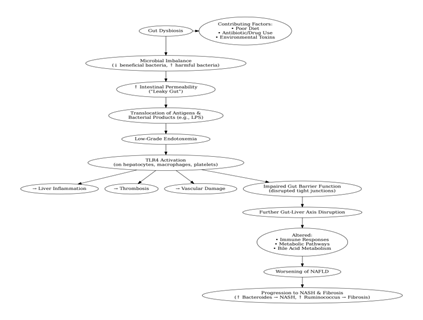
Recent study shows a strong association between gut dysbiosis and non-alcoholic fatty liver disease (NAFLD). Gut dysbiosis, defined as an imbalance in gut microbiota, can contribute to metabolic problems and promote NAFLD [55]. Bacteroides and Ruminococcus have been related to non-alcoholic steatohepatitis (NASH) and severe fibrosis, respectively [56]. In Figure 1, depicts the gut–liver axis dysregulation in NAFLD, demonstrating how gut dysbiosis increases intestinal permeability, promotes endotoxemia and TLR4-mediated inflammatory responses, and contributes to progression from simple steatosis to NASH and fibrosis.
Figure 1: illustrates the mechanistic pathway linking gut dysbiosis to the progression of NAFLD.
Dysbiosis, or microbial imbalance in the gut, is a primary contributor to increased intestinal permeability, which is also known as "leaky gut." This syndrome permits dangerous antigens to enter the body, potentially inducing autoimmune disorders such as type 1 diabetes and multiple sclerosis [57]. Increased permeability has been linked to a variety of diseases, including inflammatory bowel disease [IBD] and diabetes, where it is associated with higher amounts of proinflammatory cytokines and endotoxins [58]. Diet, pharmaceuticals, and environmental toxins can exacerbate dysbiosis by causing inflammation and disruption of tight junction proteins, increasing gut permeability [59]. They can also impair gut barrier integrity, resulting in increased translocation of bacterial products, particularly lipopolysaccharide [LPS], into the portal circulation [60]. The resulting low-grade endotoxemia causes liver inflammation, thrombosis, and vascular damage by interacting with the Toll-like receptor 4 [TLR4] found on hepatocytes, platelets, and macrophages [61]. Also, gut dysbiosis impairs immune responses, metabolic pathways, and bile acid profiles, worsening NAFLD [55] [62]. Gut microbiome dysbiosis disrupts the gut-liver axis, resulting in a vicious cycle of dysfunctions that contribute to the development of NAFLD [62].
Evidence From Animal Studies and Clinical Trials
Exercise training has been reported to reverse intestinal dysbiosis in NASH patients [63]. In animal models, high-fat diets cause gut dysbiosis, increased intestinal permeability, and liver inflammation, which leads to NAFLD progression [64]. Dietary therapies and antibiotic treatments can alter the gut flora, potentially relieving NAFLD symptoms [65]. Maternal nutrition also influences child gut microbiome and vulnerability to hepatic steatosis. Maternal sucralose use has been shown to impair intestinal development, promote gut dysbiosis, and worsen high-fat diet-induced hepatic steatosis in offspring via down-regulating GPR43 [66].
Therapeutic Perspectives
Recent research has revealed various possible therapy methods for non-alcoholic fatty liver disease (NAFLD), emphasising the condition's complexity and diversity. One promising approach is to target bile acid signaling pathways via nuclear and membrane-bound receptors such as the farnesoid X receptor [FXR] and Takeda G-protein-coupled receptor 5 [TGR5], both of which have been shown to improve insulin sensitivity and hepatic lipid metabolism [67]. An increasing pipeline of pharmacologic drugs is being developed to address a variety of pathophysiological conditions. These include fibroblast growth factor [FGF] analogues, peroxisome proliferator-activated receptor [PPAR] agonists, glucagon-like peptide-1 [GLP-1] receptor agonists, and sodium-glucose co-transporter 2 [SGLT-2] inhibitors, which each target a different aspect of NAFLD pathogenesis, such as lipotoxicity, inflammation, glucose metabolism, and hepatic fibrosis [68].
Therapeutic approaches are broadly classified into four categories: metabolic regulators, anti-inflammatory medicines, gut-liver axis modulators, and antifibrotic therapies [69]. Given the growing importance of the gut microbiome in NAFLD, microbiota-directed therapies such as probiotics, prebiotics, and faecal microbiota transplantation are being studied for their ability to restore microbial balance and decrease hepatic inflammation [70]. While dietary and physical activity changes remain the cornerstones of NAFLD care, pharmaceutical treatment is generally suggested for individuals with biopsy-confirmed nonalcoholic steatohepatitis [NASH] or severe fibrosis [69].
Antibiotic stewardship: rational use to minimize dysbiosis
Antibiotic usage and misuse can lead to the beginning and development of nonalcoholic fatty liver disease via gut dysbiosis [54].
1. Rational antibiotic prescribing
Antibiotics should only be administered when bacterial infection is proven or strongly suspected, in order to protect gut microbial diversity and prevent dysbiosis-related liver damage. Excessive or incorrect antibiotic usage alters key microbial taxa, such as Bacteroides and Ruminococcus, leading to dysbiosis and NAFLD development [72].
2. Target narrow-spectrum treatment
Choosing narrow-spectrum antibiotics decreases collateral damage to beneficial gut commensals, hence promoting microbiome resilience. Broad-spectrum drugs, such as fluoroquinolones, have been linked to an increased risk of NAFLD due to gut flora alteration [53].
3. Shortest effective duration
Limiting the length to the lowest clinically effective course reduces the influence on gut flora while protecting metabolic health. Extended antibiotic courses in animal models have been shown to enhance hepatic lipid buildup, indicating a direct influence on NAFLD [73].
4. Use antibiotics that have eubiotic effects
Non-absorbable drugs, such as rifaximin, modify gut flora without systemic exposure, which helps restore barrier integrity. Rifaximin has been shown in preclinical and clinical investigations to increase tight junction expression, decrease LPS translocation, and inhibit TLR4/NF-κB inflammatory pathways [74].
5. Combine with microbiota-supportive therapy
Concurrent use of probiotics and/or prebiotics helps to combat antibiotic-induced dysbiosis and promotes SCFA synthesis. Researchers suggest that probiotic/prebiotic therapy decreases ALT/AST and BMI in NAFLD patients [75].
6. Monitor metabolic and hepatic indicators
Regular monitoring of ALT, AST, insulin resistance, and imaging during antibiotic treatment can detect early NAFLD symptoms [76].
8. Personalise therapy with microbiota profiling
New evidence suggests that baseline microbiota composition [e.g., Bacteroides abundance] may predict NAFLD severity. Future antibiotic regimens might be customised to individual microbiota to improve eubiotic results [77] [56].
Probiotics and prebiotics: Restoring gut balance
Probiotics and prebiotics help restore intestinal homeostasis. Probiotics, prebiotics, and synbiotics have shown potential in treating NAFLD by altering gut microbiota and increasing intestinal barrier function. NAFLD is associated with intestinal dysbiosis and increased permeability [78]. Lactobacillus and Bifidobacterium strains promote microbial balance and prevent liver inflammation [79]. These treatments may improve insulin resistance, lipid profiles, and liver enzymes, making them useful as NAFLD support therapy [80, 81].
Fecal microbiota transplantation
FMT is a possible treatment for NAFLD that aims to repair gut dysbiosis and enhance intestinal barrier function [82]. While it may decrease liver fat and improve gut flora, the effects differ, particularly in lean and obese patients [83]. Some studies demonstrate that FMT has minimal impact on insulin resistance, but it can lower intestinal permeability in certain circumstances [84]. Its mechanism includes restoring gut-liver axis balance, even though further clinical data is required to validate effectiveness and identify suitable procedures [85].
Dietary Interventions: High Fibre, Polyphenols, And Fermented Foods
Individuals with irritable bowel syndrome may benefit from dietary components such as polyphenols, minerals, fibres, and fruits, with soluble fibre being especially useful in decreasing bloating and increasing stool consistency. A functional food-based diet high in fibre, polyphenols, and vegetable protein can alter faecal microbiota in type 2 diabetes patients, boosting beneficial bacteria such as Faecalibacterium prausnitzii and Akkermansia muciniphila. This dietary regimen significantly improved glycaemic management, lipid profiles, and diminished inflammation [87].
Emerging Pharmacological Therapies for Nafld and Nash
Recent investigations have identified promising pharmacological treatments for NAFLD and NASH. Obeticholic acid, a farnesoid X receptor [FXR] agonist, has been demonstrated to help reduce liver fibrosis [88]. Therapies targeting hepatic fat accumulation, oxidative stress, inflammation, and fibrosis are being investigated [89]. Liraglutide and other GLP-1 receptor agonists have been shown to be effective in the treatment of NASH [90]. Bile acid modulators, elafibranor, ASK1 inhibitors, and cenicriviroc are all potential possibilities, with many in phase 3 clinical studies [91].
Various therapy techniques focused on improving NAFLD outcomes, such as dietary changes, exercise, prebiotics, probiotics, synbiotics, antibiotics, and faecal microbiota transplantation, have showed effectiveness [70]. While probiotics have been evaluated in human randomised controlled trials, several putative therapies have yet to be confirmed in human investigations [92]. Notably, antibiotic usage has been associated to NAFLD development, with preclinical studies indicating a relationship between antibiotic-induced gut dysbiosis and NAFLD [52]. However, further study is required to determine the appropriate techniques, long-term safety, and efficacy of microbiota-targeted therapy for NAFLD [93].
Role of Clinical Pharmacist
Clinical pharmacists play an important role in encouraging rational antibiotic usage and teaching patients about gut health, with an emphasis on microbiota preservation and proper probiotic administration. Their engagement considerably improves the management of antimicrobial efforts, particularly in high-risk environments such as surgical units [94]. Pharmacists, with their extensive knowledge of antimicrobial pharmacotherapy, play an important role in reducing antibiotic resistance. According to research, there is a good association between pharmacists' microbiome knowledge and their antibiotic and probiotic dispensing practices [95]. As probiotics become more popular and widely accessible, pharmacists are expected to give evidence-based recommendations for their usage [96]. Pharmacists are advised to undertake prescription audits and patient counselling in order to enhance patient outcomes and reduce unnecessary antibiotic usage [94]. Simultaneously, the increased prevalence of non-alcoholic fatty liver disease (NAFLD), particularly among teenagers, emphasizes the importance of a multidisciplinary approach. Liver fibrosis is often regarded as the leading predictor of poor outcomes, including death and liver-related complications [97]. Although the transition from basic steatosis to later phases such as NASH or fibrosis is rather slow, the growing frequency of NAFLD is alarming [98]. As NAFLD becomes a prominent cause of cirrhosis and liver transplantation, healthcare expenditures and clinical problems rise [99]. Currently, care is on long-term lifestyle adjustments and weight loss, while various pharmaceutical medicines aimed at disease progression are under research [100].
Conclusion
Non-alcoholic fatty liver disease has quickly arisen as a major worldwide health concern, linked to metabolic diseases and impacted by a wide range of variables, including gut microbiota imbalance. Gut dysbiosis is increasingly seen as a major factor to the onset and progression of NAFLD, rather than only a side effect. Alterations in microbial populations, particularly those caused by antibiotic miuse, can alter the gut-liver axis, causing inflammation, increased permeability, and metabolic dysfunctions that contribute to liver damage. What makes this even more troubling is the growing use of antibiotics, frequently beyond therapeutic necessity, which may accidentally increase the risk of NAFLD, even in people who lack classic metabolic risk factors. However, this problem also provides opportunities for therapeutic innovation. Restoring gut health with probiotics, prebiotics, faucal microbiota transplantation, and precision-based dietary or pharmaceutical therapies offers potential disease management and preventive options. Given these findings, a move towards more careful antibiotic prescribing, microbiome-conscious treatment strategies, and proactive patient education is critical. Clinical pharmacists, doing key role antimicrobial stewardship, can help advise optimal antibiotic usage while also promoting gut-friendly practices.
Future research should focus on improving and confirming these strategies in strong clinical trials, paving the path for more personalized and effective NAFLD treatments based on gut microbiome science
References
- Khan, A., Ross, H., Parra, N., Chen, S., Chauhan, K., et al. (2022). Risk prevention and health promotion for non-alcoholic fatty liver diseases (NAFLD). Livers, 2(4):264–282.
Publisher | Google Scholor - Than, N., & Newsome, P. (2015). A concise review of non-alcoholic fatty liver disease. Atherosclerosis, 239(1):192–202.
Publisher | Google Scholor - Abd, E. S., & El-Den, A. E. (2015). Non-alcoholic fatty liver disease: The diagnosis and management. World journal of hepatology, 7(6):846.
Publisher | Google Scholor - Rector, R., Thyfault, J., Wei, Y., & Ibdah, J. (2008). Non-alcoholic fatty liver disease and the metabolic syndrome: an update. World journal of gastroenterology: WJG, 14(2):185.
Publisher | Google Scholor - Buzzetti, E., Pinzani, M., & Tsochatzis, E. (2016). The multiple-hit pathogenesis of non-alcoholic fatty liver disease (NAFLD). Metabolism, 65(8):1038–1048.
Publisher | Google Scholor - Salt, W. (2004). Nonalcoholic fatty liver disease (NAFLD): a comprehensive review. JOURNAL OF INSURANCE MEDICINE-NEW YORK THEN DENVER--, 36(1):27–41.
Publisher | Google Scholor - Tian, H., Zhang, K., Hui, Z., Ren, F., Ma, Y., Han, F., Sun, X., Kan, C., & Hou, N. (1990). Global burden of non-alcoholic fatty liver disease in 204 countries and territories from 1990 to 2019. Clinics and Research in Hepatology and Gastroenterology, 47(1):102068.
Publisher | Google Scholor - Paik, J., Henry, L., Younossi, Y., Ong, J., Alqahtani, S., & Younossi, Z. (1990). The burden of nonalcoholic fatty liver disease (NAFLD) is rapidly growing in every region of the world from 1990 to 2019. Hepatology communications.
Publisher | Google Scholor - Golabi, P., Paik, J., AlQahtani, S., Younossi, Y., Tuncer, G., & Younossi, Z. (2009). Burden of non-alcoholic fatty liver disease in Asia, the Middle East and North Africa: Data from Global Burden of Disease 2009-2019. Journal of hepatology, 75(4):795–809.
Publisher | Google Scholor - Thursby, E., & Juge, N. (2017). Introduction to the human gut microbiota. Biochemical journal, 474(11):1823–1836.
Publisher | Google Scholor - Pushpanathan, P., Mathew, G., Selvarajan, S., Seshadri, K., & Srikanth, P. (2019). Gut microbiota and its mysteries. Indian Journal of Medical Microbiology, 37(2):268–277.
Publisher | Google Scholor - Sekirov, I., Russell, S., Antunes, L., & Finlay, B. (2010). Gut microbiota in health and disease. Physiological reviews.
Publisher | Google Scholor - Comess, J., & Abad-Jorge, A. (2023). Introduction to the gut microbiome and its impact on health and disease. Topics in Clinical Nutrition, 38(3):183–195.
Publisher | Google Scholor - Albillos, A., De, G. A., & Rescigno, M. (2020). The gut-liver axis in liver disease: Pathophysiological basis for therapy. Journal of hepatology, 72(3):558–577.
Publisher | Google Scholor - Wang, J., Wang, X., Zhuo, E., Chen, B., & Chan, S. (2024). Gut‑liver axis in liver disease: From basic science to clinical treatment. Molecular Medicine Reports, 31(1):10.
Publisher | Google Scholor - Ohtani, N., & Kawada, N. (2019). Role of the gut–liver axis in liver inflammation, fibrosis, and cancer: a special focus on the gut microbiota relationship. Hepatology communications, 3(4):456–470.
Publisher | Google Scholor - Bruneau, A., Hundertmark, J., Guillot, A., & Tacke, F. (2021). Molecular and cellular mediators of the gut-liver axis in the progression of liver diseases. Frontiers in Medicine.
Publisher | Google Scholor - Yang, L., Bajinka, O., Jarju, P., Tan, Y., Taal, A., & Ozdemir, G. (2021). The varying effects of antibiotics on gut microbiota. AMB express, 11(1):116.
Publisher | Google Scholor - Tarantino, G., & Citro, V. (2024). Could adverse effects of antibiotics due to their use/misuse be linked to some mechanisms related to nonalcoholic fatty liver disease? International Journal of Molecular Sciences, 25(4):1993.
Publisher | Google Scholor - Ianiro, G., Tilg, H., & Gasbarrini, A. (2016). Antibiotics as deep modulators of gut microbiota: between good and evil. Gut, 65(11):1906–1915.
Publisher | Google Scholor - Janssen, A., Houben, T., Katiraei, S., Dijk, W., Boutens, L., van, D. B. N., Wang, Z., Brown, J., Hazen, S., Mandard, S., & Shiri-Sverdlov, R. (2017). Modulation of the gut microbiota impacts nonalcoholic fatty liver disease: a potential role for bile acids. Journal of lipid research, 58(7):1399–1416.
Publisher | Google Scholor - Singh, R., Sripada, L., & Singh, R. (2014). Side effects of antibiotics during bacterial infection: mitochondria, the main target in host cell. Mitochondrion.
Publisher | Google Scholor - Maestri, M., Santopaolo, F., Pompili, M., Gasbarrini, A., & Ponziani, F. (2023). Gut microbiota modulation in patients with non-alcoholic fatty liver disease: Effects of current treatments and future strategies. Frontiers in Nutrition.
Publisher | Google Scholor - Jacob, J., Ahmed, A., & Cholankeril, G. (2021). The impact of alteration in gut microbiome in the pathogenesis of nonalcoholic fatty liver disease. Current opinion in infectious diseases, 34(5):477–482.
Publisher | Google Scholor - Hu, H., Lin, A., Kong, M., Yao, X., Yin, M., Xia, H., Ma, J., & Liu, H. (2020). Intestinal microbiome and NAFLD: molecular insights and therapeutic perspectives. Journal of gastroenterology, 55(2):142–158.
Publisher | Google Scholor - Bellucci, E., Chiereghin, F., Pacifici, F., Donadel, G., De, S. A., Malatesta, G., Valente, M., Guadagni, F., Infante, M., Rovella, V., & Noce, A. (2023). Novel therapeutic approaches based on the pathological role of gut dysbiosis on the link between nonalcoholic fatty liver disease and insulin resistance. European Review for Medical and Pharmacological Sciences, 27(5):1921–1944.
Publisher | Google Scholor - Ganguly, S., & Chandrasekhara, P. (2019). Gut microbiota in health and disease. APIK Journal of Internal Medicine, 7(1):19–30.
Publisher | Google Scholor - Collado, M., Rautava, S., Aakko, J., Isolauri, E., & Salminen, S. (2016). Human gut colonisation may be initiated in utero by distinct microbial communities in the placenta and amniotic fluid. Scientific reports, 6(1):1–3.
Publisher | Google Scholor - Izakson, M., & Zalsman, G. (2023). The Psychic Gut: Do Gut Flora Constitute the Next Psychiatric Treatment? Journal of Psychiatry and Psychiatric Disorders, 7(1):25–29.
Publisher | Google Scholor - Tsai, M., Liu, Y., Lin, C., Wang, C., Wu, Y., Yong, C., Chen, K., Chuah, S., Yao, C., Huang, P., & Chen, C. (2020). Gut microbiota dysbiosis in patients with biopsy-proven nonalcoholic fatty liver disease: a cross-sectional study in Taiwan. Nutrients, 12(3):820.
Publisher | Google Scholor - Jiang, W., Wu, N., Wang, X., Chi, Y., Zhang, Y., Qiu, X., Hu, Y., Li, J., & Liu, Y. (2015). Dysbiosis gut microbiota associated with inflammation and impaired mucosal immune function in intestine of humans with non-alcoholic fatty liver disease. Scientific reports, 5(1):8096.
Publisher | Google Scholor - Rowland, I., Gibson, G., Heinken, A., Scott, K., Swann, J., Thiele, I., & Tuohy, K. (2018). Gut microbiota functions: metabolism of nutrients and other food components. European journal of nutrition, 57(1):1–24.
Publisher | Google Scholor - Gao, , & M, (2020). K. , Pei-wen Hu and Mumine Senturk.
Publisher | Google Scholor - Mirzaei, R., Dehkhodaie, E., Bouzari, B., Rahimi, M., Gholestani, A., Hosseini-Fard, S., Keyvani, H., Teimoori, A., & Karampoor, S. (2022). Dual role of microbiota-derived short-chain fatty acids on host and pathogen. Biomedicine & Pharmacotherapy.
Publisher | Google Scholor - Kim, C. (2021). Control of lymphocyte functions by gut microbiota-derived short-chain fatty acids. Cellular & molecular immunology, 18(5):1161–1171.
Publisher | Google Scholor - Martin-Gallausiaux, C., Marinelli, L., Blottière, H., Larraufie, P., & Lapaque, N. (2021). SCFA: mechanisms and functional importance in the gut. Proceedings of the Nutrition Society, 80(1):37–49.
Publisher | Google Scholor - Lang, S., & Schnabl, B. (2020). Microbiota and fatty liver disease—the known, the unknown, and the future. Cell host & microbe, 28(2):233–244.
Publisher | Google Scholor - Stärkel, P., & Schnabl, B. (2016). Bidirectional communication between liver and gut during alcoholic liver disease. InSeminars in liver disease 2016 Sep (Vol.
Publisher | Google Scholor - Arab, J., Karpen, S., Dawson, P., Arrese, M., & Trauner, M. (2017). Bile acids and the gut–liver axis: emerging roles in liver physiology and disease. Nat Rev Gastroenterol Hepatol, 14(9):503–516.
Publisher | Google Scholor - Ohtani, N., & Hara, E. (2021). Gut‐liver axis‐mediated mechanism of liver cancer: a special focus on the role of gut microbiota. Cancer science, 112(11):4433–4443.
Publisher | Google Scholor - Blaser, M. (2019). Fecal microbiota transplantation for dysbiosis—predictable risks. New England Journal of Medicine, 381(21):2064–2066.
Publisher | Google Scholor - Carías, D. A., de, J. R. S. D., Stefanolo, J., Cruz, S. M., Casas, I., & Zuluaga, P. J. (2025). Intestinal dysbiosis: exploring definition, associated symptoms, and perspectives for a comprehensive understanding—a scoping review. Probiotics and Antimicrobial Proteins, 17(1):440–449.
Publisher | Google Scholor - Perez, N., Dorsen, C., & Squires, A. (2020). Dysbiosis of the gut microbiome: a concept analysis. Journal of Holistic Nursing, 38(2):223–232.
Publisher | Google Scholor - Elvers, K., Wilson, V., Hammond, A., Duncan, L., Huntley, A., Hay, A., & Van, D. W. E. (2020). Antibiotic-induced changes in the human gut microbiota for the most commonly prescribed antibiotics in primary care in the UK: a systematic review. BMJ open.
Publisher | Google Scholor - Schwartz, B., Pollak, J., Bailey-Davis, L., Hirsch, A., Cosgrove, S., Nau, C., Kress, A., Glass, T., & Bandeen-Roche, K. (2016). Antibiotic use and childhood body mass index trajectory. International journal of obesity, 40(4):615–621.
Publisher | Google Scholor - Duan, H., Yu, L., Tian, F., Zhai, Q., Fan, L., & Chen, W. (2022). Antibiotic-induced gut dysbiosis and barrier disruption and the potential protective strategies. Critical reviews in food science and nutrition, 62(6):1427–1452.
Publisher | Google Scholor - Maier, L., Goemans, C., Wirbel, J., Kuhn, M., Eberl, C., Pruteanu, M., Müller, P., Garcia-Santamarina, S., Cacace, E., Zhang, B., & Gekeler, C. (2021). Unravelling the collateral damage of antibiotics on gut bacteria. Nature, 599(7883):120–124.
Publisher | Google Scholor - Zeng, M., Inohara, N., & Nuñez, G. (2017). Mechanisms of inflammation-driven bacterial dysbiosis in the gut. Mucosal immunology, 10(1):18–26.
Publisher | Google Scholor - Stecher, B., Maier, L., & Hardt, W. (2013). 'Blooming'in the gut: how dysbiosis might contribute to pathogen evolution. Nature Reviews Microbiology, 11(4):277–284.
Publisher | Google Scholor - Chen, Q., An, X., Zheng, B., Gillings, M., Peñuelas, J., Cui, L., Su, J., & Zhu, Y. (2019). Loss of soil microbial diversity exacerbates spread of antibiotic resistance. Soil Ecology Letters, 1(1):3–13.
Publisher | Google Scholor - Gillings, M., Paulsen, I., & Tetu, S. (2015). Ecology and evolution of the human microbiota: fire, farming and antibiotics. Genes, 6(3):841–857.
Publisher | Google Scholor - Ebrahimi, F., Simon, T., Hagström, H., Sun, J., Bergman, D., Forss, A., Roelstraete, B., Engstrand, L., & Ludvigsson, J. (2023). Antibiotic use and development of nonalcoholic fatty liver disease: a population‐based case–control study. Liver International, 43(10):2186–2197.
Publisher | Google Scholor - Sinha, R., Lockman, K., Homer, N., Bower, E., Brinkman, P., Knobel, H., Fallowfield, J., Jaap, A., Hayes, P., & Plevris, J. (2020). Volatomic analysis identifies compounds that can stratify non-alcoholic fatty liver disease. JHEP Reports, 2(5):100137.
Publisher | Google Scholor - Tarantino, G., & Citro, V. (2024). Could adverse effects of antibiotics due to their use/misuse be linked to some mechanisms related to nonalcoholic fatty liver disease?. International Journal of Molecular Sciences, 25(4):1993.
Publisher | Google Scholor - Jadhav, P., Thomas, A., Nanda, R., & Chitlange, S. (2023). Unveiling the role of gut dysbiosis in non-alcoholic fatty liver disease. European Journal of Gastroenterology & Hepatology, 35(12):1324–1333.
Publisher | Google Scholor - Boursier, J., Mueller, O., Barret, M., Machado, M., Fizanne, L., Araujo‐Perez, F., Guy, C., Seed, P., Rawls, J., David, L., & Hunault, G. (2016). The severity of nonalcoholic fatty liver disease is associated with gut dysbiosis and shift in the metabolic function of the gut microbiota. Hepatology, 63(3):764–775.
Publisher | Google Scholor - Kinashi, Y., & Hase, K. (2021). Partners in leaky gut syndrome: intestinal dysbiosis and autoimmunity. Frontiers in immunology.
Publisher | Google Scholor - Fukui, H. (2016). Increased intestinal permeability and decreased barrier function: does it really influence the risk of inflammation?. Inflammatory intestinal diseases, 1(3):135–145.
Publisher | Google Scholor - Sarkar, K., & Sil, P. (2021). Effect of diet, pharmaceuticals, and environmental toxicants on gut microbiota imbalance and increased intestinal membrane permeability. InToxicological Risk Assessment and Multi-System Health Impacts from Exposure 2021 Jan 1.
Publisher | Google Scholor - Wu, M., & Fan, J. (2023). Gut microbiome and nonalcoholic fatty liver disease. Hepatobiliary & Pancreatic Diseases International, 22(5):444–451.
Publisher | Google Scholor - Violi, F., Nocella, C., Bartimoccia, S., Castellani, V., Carnevale, R., Pignatelli, P., & Cammisotto, V. (2023). Gut dysbiosis-derived low-grade endotoxemia: A common basis for liver and cardiovascular disease. Polish Heart Journal (Kardiologia Polska):81(6):563–571.
Publisher | Google Scholor - Pezzino, S., Sofia, M., Faletra, G., Mazzone, C., Litrico, G., La, G. G., & Latteri, S. (2022). Gut–Liver axis and non-alcoholic fatty liver disease: A vicious circle of dysfunctions orchestrated by the gut microbiome. Biology, 11(11):1622.
Publisher | Google Scholor - Hughes, A., Dahmus, J., Rivas, G., Hummer, B., See, J., Wright, J., Lamendella, R., Schmitz, K., Sciamanna, C., Ruffin, M., & Patterson, A. (2021). Exercise training reverses gut dysbiosis in patients with biopsy-proven nonalcoholic steatohepatitis: a proof of concept study. Clinical Gastroenterology and Hepatology, 19(8):1723–1725.
Publisher | Google Scholor - Longo, L., Tonin, F. J., Rampelotto, P., Hirata, D. G., Pasqualotto, A., P, O. C., Thadeu, S. C. C., Reverbel, d. S. T., Uribe-Cruz, C., & Álvares-da-Silva, M. (2020). Gut dysbiosis and increased intestinal permeability drive microRNAs, NLRP-3 inflammasome and liver fibrosis in a nutritional model of non-alcoholic steatohepatitis in adult male sprague dawley rats. Clinical and experimental gastroenterology.
Publisher | Google Scholor - Yuan, Y., Sun, Z., Zhang, Y., Liang, F., & He, X. (2016). Influence of gut microecology on the pathogenesis and treatment of nonalcoholic fatty liver disease. Zhonghua gan zang bing za zhi= Zhonghua ganzangbing zazhi= Chinese journal of hepatology, 24(5):375–379.
Publisher | Google Scholor - Dai, X., Guo, Z., Chen, D., Li, L., Song, X., Liu, T., Jin, G., Li, Y., Liu, Y., Ajiguli, A., & Yang, C. (2020). Maternal sucralose intake alters gut microbiota of offspring and exacerbates hepatic steatosis in adulthood. Gut Microbes, 11(4):1043–1063.
Publisher | Google Scholor - Arab, J., Karpen, S., Dawson, P., Arrese, M., & Trauner, M. (2017). Bile acids and nonalcoholic fatty liver disease: molecular insights and therapeutic perspectives. Hepatology, 65(1):350–362.
Publisher | Google Scholor - Huang, X., Chen, H., Wen, S., Dong, M., Zhou, L., & Yuan, X. (2023). Therapeutic approaches for nonalcoholic fatty liver disease: established targets and drugs. Diabetes, Metabolic Syndrome and Obesity.
Publisher | Google Scholor - Brown, E., Hydes, T., Hamid, A., & Cuthbertson, D. (2021). Emerging and established therapeutic approaches for nonalcoholic fatty liver disease. Clinical therapeutics, 43(9):1476–1504.
Publisher | Google Scholor - Chen, H., Huang, H., Li, Y., Xu, H., & Zhou, Y. (2020). Therapeutic advances in non-alcoholic fatty liver disease: A microbiota-centered view. World journal of gastroenterology, 26(16):1901.
Publisher | Google Scholor - Fang, J., Yu, C., Li, X., Yao, J., Fang, Z., Yoon, S., & Yu, W. (2022). Gut dysbiosis in nonalcoholic fatty liver disease: pathogenesis, diagnosis, and therapeutic implications. Frontiers in cellular and infection microbiology.
Publisher | Google Scholor - Cai, W., Qiu, T., Hu, W., & Fang, T. (2024). Changes in the intestinal microbiota of individuals with non-alcoholic fatty liver disease based on sequencing: An updated systematic review and meta-analysis. PLoS One.
Publisher | Google Scholor - Kau, A., Ahern, P., Griffin, N., Goodman, A., & Gordon, J. (2011). Human nutrition, the gut microbiome and the immune system. Nature, 474(7351):327–336.
Publisher | Google Scholor - Qiu, T., Zhu, X., Wu, J., Hong, W., Hu, W., & Fang, T. (2025). Mechanisms of rifaximin inhibition of hepatic fibrosis in mice with metabolic dysfunction associated steatohepatitis through the TLR4/NFκB pathway. Scientific Reports, 15(1):9815.
Publisher | Google Scholor - Da, S. H., Teterina, A., Comelli, E., Taibi, A., Arendt, B., Fischer, S., Lou, W., & Allard, J. (2018). Nonalcoholic fatty liver disease is associated with dysbiosis independent of body mass index and insulin resistance. Scientific reports, 8(1):1466.
Publisher | Google Scholor - Swamikkannu, D., Dasarapu, S., Siva, R., Nallam, J., & Pabba, S. (2024). The gut-liver nexus: exploring gut microbiota dysbiosis in non-alcoholic fatty liver disease and its therapeutic implications. Egyptian Liver Journal, 14(1):28.
Publisher | Google Scholor - Li, F., Ye, J., Shao, C., & Zhong, B. (2021). Compositional alterations of gut microbiota in nonalcoholic fatty liver disease patients: a systematic review and meta-analysis. Lipids in Health and Disease, 20(1):22.
Publisher | Google Scholor - Mijangos-Trejo, A., Nuño-Lambarri, N., Barbero-Becerra, V., Uribe-Esquivel, M., Vidal-Cevallos, P., & Chávez-Tapia, N. (2023). Prebiotics and probiotics: therapeutic tools for nonalcoholic fatty liver disease. International Journal of Molecular Sciences, 24(19):14918.
Publisher | Google Scholor - Carpi, R., Barbalho, S., Sloan, K., Laurindo, L., Gonzaga, H., Grippa, P., Zutin, T., Girio, R., Repetti, C., Detregiachi, C., & Bueno, P. (2022). The effects of probiotics, prebiotics and synbiotics in non-alcoholic fat liver disease (NAFLD) and non-alcoholic steatohepatitis (NASH): a systematic review. International journal of molecular sciences, 23(15):8805.
Publisher | Google Scholor - Castillo, V., Figueroa, F., González-Pizarro, K., Jopia, P., & Ibacache-Quiroga, C. (2021). Probiotics and prebiotics as a strategy for non-alcoholic fatty liver disease, a narrative review. Foods, 10(8):1719.
Publisher | Google Scholor - Wastyk, H., Fragiadakis, G., Perelman, D., Dahan, D., Merrill, B., Yu, F., Topf, M., Gonzalez, C., Van, T. W., Han, S., Robinson, J., Elias, J., Sonnenburg, E., Gardner, C., & Sonnenburg, J. (2021). Gut-microbiota-targeted diets modulate human immune status. Cell, 184(16):4137–4153.
Publisher | Google Scholor - Xue, L., Luo, W., Wu, L., He, X., Xia, H., & Chen, Y. (2019). Fecal microbiota transplantation for the treatment of nonalcoholic fatty liver disease. Exploratory Research and Hypothesis in Medicine, 4(1):12–18.
Publisher | Google Scholor - Xue, L., Deng, Z., Luo, W., He, X., & Chen, Y. (2022). Effect of fecal microbiota transplantation on non-alcoholic fatty liver disease: A randomized clinical trial. Frontiers in cellular and infection microbiology.
Publisher | Google Scholor - Craven, L., Rahman, A., Parvathy, S., Beaton, M., Silverman, J., Qumosani, K., Hramiak, I., Hegele, R., Joy, T., Meddings, J., & Urquhart, B. (2020). Allogenic fecal microbiota transplantation in patients with nonalcoholic fatty liver disease improves abnormal small intestinal permeability: a randomized control trial. Official journal of the American College of Gastroenterology| ACG, 115(7):1055–1065.
Publisher | Google Scholor - Qiu, X., Cheng, S., Liu, Y., Li, Y., Zhang, R., Li, N., & Li, Z. (2024). Fecal microbiota transplantation for treatment of non-alcoholic fatty liver disease: Mechanism, clinical evidence, and prospect. World Journal of Gastroenterology, 30(8):833.
Publisher | Google Scholor - Wastyk, H., Fragiadakis, G., Perelman, D., Dahan, D., Merrill, B., Yu, F., Topf, M., Gonzalez, C., Van, T. W., Han, S., & Robinson, J. (2021). Gut-microbiota-targeted diets modulate human immune status. Cell, 184(16):4137–4153.
Publisher | Google Scholor - Medina-Vera, I., Sanchez-Tapia, M., Noriega-López, L., Granados-Portillo, O., Guevara-Cruz, M., Flores-López, A., Avila-Nava, A., Fernández, M., Tovar, A., & Torres, N. (2019). A dietary intervention with functional foods reduces metabolic endotoxaemia and attenuates biochemical abnormalities by modifying faecal microbiota in people with type 2 diabetes. Diabetes & metabolism, 45(2):122–131.
Publisher | Google Scholor - Abenavoli, L., Falalyeyeva, T., Boccuto, L., Tsyryuk, O., & Kobyliak, N. (2018). Obeticholic acid: a new era in the treatment of nonalcoholic fatty liver disease. Pharmaceuticals, 11(4):104.
Publisher | Google Scholor - Rotman, Y., & Sanyal, A. (2017). Current and upcoming pharmacotherapy for non-alcoholic fatty liver disease. Gut, 66(1):180–190.
Publisher | Google Scholor - Attia, S., Softic, S., & Mouzaki, M. (2021). Evolving role for pharmacotherapy in NAFLD/NASH. Clinical and translational science, 14(1):11–19.
Publisher | Google Scholor - Sumida, Y., & Yoneda, M. (2018). Current and future pharmacological therapies for NAFLD/NASH. Journal of gastroenterology, 53(3):362–376.
Publisher | Google Scholor - Aron-Wisnewsky, J., Warmbrunn, M., Nieuwdorp, M., & Clement, K. (2020). Nonalcoholic fatty liver disease: modulating gut microbiota to improve severity?. Gastroenterology, 158(7):1881–1898.
Publisher | Google Scholor - Jadhav, P., Thomas, A., Nanda, R., & Chitlange, S. (2024). Correlation of non-alcoholic fatty liver disease and gut microflora: clinical reports and treatment options. Egyptian Liver Journal, 14(1):21.
Publisher | Google Scholor - Kuruvilla, A., Madhan, R., & Puttaswamy, M. (2023). Clinical pharmacist-initiated assessment and amelioration of appropriate antibiotic use in surgical units at a South Indian tertiary care hospital-A handshake approach. The Journal of Infection in Developing Countries, 17(01):66–72.
Publisher | Google Scholor - Sawan, H., Shroukh, W., Abutaima, R., Al, O. S., Abdel-Qader, D., & Binsuwaidan, R. (2024). Impact of Jordanian Pharmacists’ Knowledge of the Human Microbiome: Has the Practice of Antibiotics and Probiotics Dispensing Been Affected? A Cross-Sectional Study. Infection and Drug Resistance.
Publisher | Google Scholor - Prescott, S. (2019). Vitamin and mineral supplements: Gut health and probiotics: Probiotics for a healthy microbiome: An evidence-based approach to appropriate product choice. AJP: The Australian Journal of Pharmacy, 100(1182):46–55.
Publisher | Google Scholor - Angulo, P., Kleiner, D., Dam-Larsen, S., Adams, L., Bjornsson, E., Charatcharoenwitthaya, P., Mills, P., Keach, J., Lafferty, H., Stahler, A., & Haflidadottir, S. (2015). Liver fibrosis, but no other histologic features, is associated with long-term outcomes of patients with nonalcoholic fatty liver disease. Gastroenterology, 149(2):389–397.
Publisher | Google Scholor - Adams, L., & Ratziu, V. (2015). Non-alcoholic fatty liver–perhaps not so benign. Journal of hepatology, 62(5):1002–1004.
Publisher | Google Scholor - Ratziu, V., Bellentani, S., Cortez-Pinto, H., Day, C., & Marchesini, G. (2009). A position statement on NAFLD/NASH based on the EASL 2009 special conference. Journal of hepatology, 53(2):372–384.
Publisher | Google Scholor - Berenguer, M. (2020). Fat, cancer, the gut-liver axis and rare liver diseases. JHEP Reports, 2(6):100189.
Publisher | Google Scholor