Research Article
The Stage-Specific Effects of MTNR1B Variants like rs10830963 on the Progression from Pre-diabetes to Type 2 Diabetes
- Zainaba Naeema *
- Galau Audrey Jasmine Kamilia
- Beke-Szabo Hanna, Aiteanu Clara
- Hamza Naeem
Iuliu Hațieganu University of Medicine and Pharmacy, Cluj-Napoca, Romania.
*Corresponding Author: Zainaba Naeema, Iuliu Hațieganu University of Medicine and Pharmacy, Cluj-Napoca, Romania.
Citation: Naeema Z., Kamilia G.A.J., Beke S. Hanna, Clara A., Naeem H. (2026). The Stage-Specific Effects of MTNR1B Variants like rs10830963 on the Progression from Pre-diabetes to Type 2 Diabetes. Clinical Research and Reports, BioRes Scientia Publishers. 5(1):1-7. DOI: 10.59657/2995-6064.brs.26.058
Copyright: © 2026 Zainaba Naeema, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: December 26, 2025 | Accepted: January 20, 2026 | Published: February 16, 2026
Abstract
The MTNR1B gene codes for melatonin receptor 1B (MT1B), a G-protein-coupled receptor in pancreatic beta cells that bridges the circadian melatonin signals to insulin output and fasting glucose control. The intronic SNP rs10830963 is prominent as a confirmed T2D risk factor, with the G allele enhancing MTNR1B expression and strengthening melatonin's restriction on insulin release, which further increases fasting glucose. Prediabetes, marked by IFG or IGT, lies between normal glucose balance and full T2D, where beta-cell issues and early insulin resistance predict advancement. GWAS and functional work show the rs10830963-G allele links closely to weakened early insulin response, elevated fasting glucose, and higher IFG odds in those without diabetes. This shows its primary effect on beta-cell function early on, unlike the insulin resistance dominant in later T2D. This review explores the variant's phase-dependent impacts, comparing its beta-cell role in prediabetes versus overt T2D development. Melatonin pathways adjust glucose handling via sleep and light cues, and rs10830963 intensifies vulnerability to circadian disturbance. Investigating its push on beta-cell drop in prediabetes could heighten risk grading and direct personalised measures—such as chronotype-based habits or drug picks—to delay or stop T2D in at-risk groups. At its core, this review argues rs10830963 has its greatest effects in prediabetes by speeding beta-cell breakdown, causing the progression from glucose glitches to outright T2D.
Keywords: MTNR1B; rs10830963; prediabetes; type 2 diabetes; beta-cell dysfunction; melatonin signalling; circadian rhythm; genetic variant; impaired fasting glucose; insulin secretion; stage-specific effects; progression risk
Introduction
The MTNR1B gene codes for the melatonin receptor 1B (MT1B), a G-protein-coupled receptor found mostly in pancreatic beta cells. This receptor connects circadian melatonin signalling to insulin secretion and fasting glucose regulation. The intronic variant rs10830963 is located here, a well-established and widely confirmed risk single nucleotide polymorphism (SNP) for type 2 diabetes (T2D). The risk allele G increases MTNR1B expression, thus strengthening melatonin's curbing effect on insulin release and raising fasting plasma glucose levels [1,3,5]. Prediabetes—defined by impaired fasting glucose (IFG) and/or impaired glucose tolerance (IGT)—serves as an intermediate state between normal glucose homeostasis and overt T2D. In this phase, beta-cell dysfunction appears with early insulin resistance, both of which foreshadow disease progression [1,2,3,4]. Genome-wide association studies and comprehensive physiological investigations have repeatedly confirmed that the rs10830963-G allele is strongly correlated to impaired early-phase insulin secretion, elevated fasting glucose, and increased risk of isolated IFG, even in nondiabetic individuals [3,4,6,7]. These results suggest that the variant fundamentally impairs beta-cell function at an early stage, rather than causing peripheral insulin resistance, which usually the main factor in established T2D.
The aim of this review is to study the stage-specific effects of this intronic variant, comparing its effects on beta-cell function during prediabetes with its contribution in the development of overt T2D and glycaemic dysregulation [3,4,5,6,7]. Melatonin signalling is increasingly viewed as a vital regulator of glucose metabolism, affecting sleep timing and light exposure to influence glycaemic traits. The rs10830963 variant seems to increase sensitivity to circadian rhythm disturbances [4]. By clarifying how this variant hastens beta-cell failure in the prediabetic phase, we may better group risk and guide precision medicine approaches—such as gene-focused lifestyle approaches focused on chronotype or pharmacotherapy selection—to slow or prevent progression to T2D in predisposed individuals [8]. The main claim of this review is that rs10830963 applies specifically strong harmful effects during the prediabetic stage by accelerating beta-cell dysfunction, which results in promoting the progression from impaired glucose regulation to complete T2D [3,4,5,6,8].
Genetic Mechanisms of MTNR1B Variants: Mechanisms Linking to Impaired Glucose Tolerance and Type 2 Diabetes Risk
The rs10830963 variant is a single-nucleotide polymorphism (SNP) located in an intronic region of the MTNR1B gene on chromosome 11 (GRCh38.p14). This polymorphism involves a cytosine-to-guanine substitution, resulting in two alleles: C (reference/non-risk) and G (risk) [9]. Multiple studies have shown a clear link between having one or two G alleles and a 2–4-fold rise in MTNR1B mRNA expression in human pancreatic islet cells, as seen by RNA sequencing [10], the Meta-Analyses of Glucose and Insulin-related traits Consortium (MAGIC) genome-wide association (GWA) study [11], and further GWAs [12]. At the MTNR1B site, rs10830963 appears to be the only likely cause with 99.8% posterior probability [13]. The intronic site covers a working islet gene-control area held by transcription factors FOXA2 and NEUROD1, found through chromatin immunoprecipitation sequencing (ChIP-seq). The G allele potentially creates a new NEUROD1 target site missing from the C allele [13]. FOXA2 keeps β-cell nature and enhancer activity, while NEUROD1 drives insulin gene expression; both are very common in pancreatic β-cells and essential for insulin secretion. Because of its location in this switch held by islet-only factors, the G allele most likely changes protein attachment, and so increasing MTNR1B expression.
This high production is unique to certain cells, found solely in human pancreatic β-cells and not in other tissues like the brain, because of varied DNA structures and links to islet-limited transcription factors. Melatonin, produced by pineal gland cells, matches a daily rhythm with its highest point at night and lowest point during the day. In β-cells, MTNR1B receptor activation blocks adenylate cyclase, lowers cyclic AMP (cAMP) levels, and reduces insulin secretion. The rs10830963-driven rise in receptor levels increases β-cell responsiveness to melatonin. G-allele carriers also show a delayed melatonin rhythm, with a 1-hour 22-minute push-back in morning decline and 41-minute extension of high levels [14]. As a result, glucose tolerance—the body's ability to control sugar after meals—is weakened in these people, as high melatonin happens at the same time as plentiful receptors, strengthening insulin reduction. In G-carriers, melatonin lingers until morning hours—a normal eating time—keeping receptor activation and slowing insulin release when it is naturally necessary. This combination of high melatonin and receptor overexpression causes stronger insulin reduction, appearing as high morning glucose levels and reduced glucose tolerance compared to non-carriers. Worth noting, no such differences show up at night, when melatonin is equally high across all genotypes [15]. Population-specific allele frequencies for rs10830963 vary widely, as summarized below, influencing type 2 diabetes risk stratification across ethnic groups [16-19].
Table 1: Population-specific allele frequencies for rs10830963
| Study /Source | Population | N (Sample size) | G-allele frequency (%) | C-allele frequency (%) | Genotype frequencies (CC / CG / GG) | Notes |
| UK Biobank [16] | European | 337’083 | 27.5 | 72.5 | 52.6/39.7/7.6 | Cohort papers reports |
| Shanghai Diet and Health Survey [17] | Chinese | 2951 | 42.9 | 47.1 | 32/50.3/17.7 | Assessed through recorded dietary intake for 3 days, venous sample after 12h of overnight fast; Genotyping through the magnetic bead method by the Universal Genomic DNA Extraction Kit |
| Inter99 [18] | Danish | 5’553 | 27.5 | 72.5 | 53/39/8 | Association between genotype and quantitative metabolic traits, investigation of glucose tolerance and fasting glucose |
| ALFA Allele Frequency [19] | African | 3746 | 7 | 93 | 86.5/13/0.5 | |
| ALFA Allele Frequency [19] | Latin American | 926 | 20 | 80 | 64/32/4 | |
| ALFA Allele Frequency [19] | African American | 3580 | 7 | 93 | 86.5/13/0.5 | |
| Cape Town Vascular and Metabolic Health study [20] | South African | 1365 | 18 | 82 | 67/28.7/4.2 | Quantitative real time PCR ; assessed through prediction of fasting plasma glucose, insulin and HOMA-IR (homeostatic Model Assessment for Insulin Resistance) |
| gnomAD v4 – Genomes [9] | Middle Eastern | 294 | 29 | 71 | 50.4/41.2/8.4 |
Effects of Prediabetes Stage
Prediabetes, or intermediate hyperglycaemia, marks a metabolically unstable time where insulin resistance and genetic links to beta-cell function become clearer. It is a high-risk state for the development of diabetes. Rather than a fixed diagnostic label, it reflects ongoing metabolic worsening, with individuals frequently shifting between normal sugar levels, impaired fasting glucose (IFG), and impaired glucose tolerance (IGT) over time [21].
A key element in this progression involves the insulin-secreting beta-cells. Weir's multistage model outlines five phases of diabetes evolution [22].
Table 2: Weir’s Multistage model outlines – five phases of Diabetes evolution
| Stage | Metabolic State | Glycemia | β-Cell Mass | β-Cell Phenotype |
| 1 | Compensated insulin resistance | Normoglycemia | Preserved to mildly reduced | Fully differentiated; preserved acute GSIS |
| 2 | Stable adaptive failure | Mild hyperglycaemia | Reduced | Partial dedifferentiation; impaired GSIS |
| 3 | Early decompensation (unstable) | Rapidly rising glycemia | Further reduced | Progressive functional decline |
| 4 | Stable β-cell decompensation | Overt diabetes | Markedly reduced | Severe dedifferentiation; minimal GSIS |
| 5 | Advanced β-cell failure | Severe hyperglycaemia | Profoundly depleted | Near-absent functional β-cell identity |
Another sign of prediabetes that shows high glycaemic burden is a rapidly rising HbA1c. Even small increases within the prediabetic range (5.7–6.4%) signal the body's struggle to keep normal glucose control. Research has shown that people in the 6.0–6.2% range are 1.66 times more likely to meet prediabetes criteria than those in the 5.7–5.9% range. Beyond its role in diagnosis, HbA1c serves as a prediction tool: higher baseline levels in prediabetes link to faster progression to type 2 diabetes (T2D) if not addressed. This link comes from sustained high blood sugar, which speeds up beta-cell failure and insulin resistance [23].
A Framingham study focused on people with prediabetes showed that HbA1c raises significantly with age in non-diabetic groups. Thus, HbA1c is not metabolically static, even before diabetes starts [24]. This gradual shift means that some people may enter the prediabetes group without a clear triggering event. The study reported a yearly increase of 0.014 HbA1c units per year of age, highlighting the link between aging and this marker. At these subtle levels, HbA1c changes are less caused by strong insulin resistance and more linked with early dysfunction in first-phase insulin release, where beta-cells fail to properly limit post-meal glucose spikes. As a result, HbA1c acts as a sensitive measure of early beta-cell strain, well before fasting glucose changes significanyly [24].
The following table illustrates conceptual hazard ratios by stage [25-27].
Table 3: Conceptual Hazard Ratios by stage
| Stage | HbA1c | β-Cell Status | Approx. Diabetes Risk/HR* |
| Normal | <5.7% | Fully differentiated | 1 (ref) |
| Early dysglycemia | 5.7–5.9% | Partial dedifferentiation | ~1.5–2 |
| Prediabetes | 6.0–6.4% | Progressive functional decline | ~2.5–4 |
| High prediabetes | 6.4–6.5% | Severe dedifferentiation | ~4–6 |
| Diabetes | ≥6.5% | Near-absent functional β-cell identity | >6 |
*Hazard ratios are conceptual estimates based on epidemiologic trends.
Progression to T2D and Modifiers
The shift from prediabetes to type 2 diabetes (T2D) is a complex process shaped by genetic versions like rs10830963 in MTNR1B, which controls melatonin signals and insulin release. This variant’s effects are stage-specific and are usually linked with early sugar imbalances (prediabetes) compared to full T2D, which plays a major role in the growth of the disease. Large-scale analyses describe increased odds for rs10830963 in prediabetes worsening, with estimates around OR=1.2 for moving from prediabetes to T2D versus OR=1.1 in established T2D cases [28,31]. These findings show a decreasing effect size as hyperglycaemia advances, potentially due to compensatory actions or environmental factors.
A vital modifier is gene-lifestyle interactions, where MTNR1B variants increase T2D risk under certain behaviours. For example, shift work disrupts daily rhythms, which worsens melatonin’s blocking effects on insulin release. Studies show that rs10830963 carriers facing irregular sleep patterns have a 20-30% higher risk of worsening, as melatonin imbalance harms beta-cell function [30,32]. Similarly, inactive lifestyles or high-fat diets can also have negative effects on that gene. Research revealed odds up to 1.5 in prediabetic groups lacking exercise [29,32]. Long-term trials show that a balanced diet and daily exercise can lower worsening by 40-50% in variant carriers, which highlights paths that can be changed [29,33].
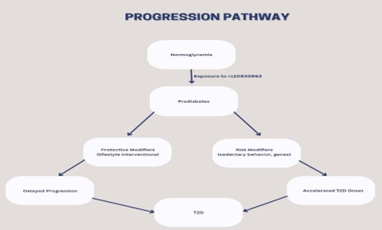
Genetic interactions further complicate progression. Rs10830963 interacts with other T2D sites like TCF7L2, where having both raises beta-cell failure risk in prediabetes [31]. Global studies describe ancestry-based modifiers, with stronger effects in East Asians versus Europeans [28]. In conclusion, progression pathways can be visualized in the flowchart, which integrates genetic and environmental factors, recommended for personalized screening in prediabetic rs10830963 carriers to interrupt pathways early [29,30].
Figure 1: Progression Pathway
Research Gaps
Despite many studies into MTNR1B and the rs10830963 variant, several key problems remain:
Incomplete stage-specific data: Many analyses are one-time or focus on fasting glucose rather than following shifts from normoglycemia to prediabetes to T2D in the same people [35,37,39]. There is little direct proof measuring how rs10830963 changes the speed of movement between specific stages.
Unclear role of rare vs common variants: rare versions of MTNR1B clearly raise T2D risk, but large biobank findings show higher HbA1c without a matching rise in diagnosed T2D [34]. How these uncommon alleles shape prediabetes—and if they mostly drive hidden dysglycemia versus full illness—is not yet solved.
Limited link with sleep science: The rs10830963 G allele stretches out nighttime melatonin, delays melatonin cessation and increases the impact of late meals on beta-cell function [38]. Nonetheless, forward-looking data linking sleep, melatonin timing, genotype, and later stage-specific diabetes is rare.
Heterogeneity across populations and reproductive stages: Most gene-trait links come from European or East Asian samples [35,37,39]. The extent to which MTNR1B-linked worsening applies to diverse ancestries or covers pregnancy and after-birth phases—where rs10830963 greatly boosts gestational diabetes risk—is not well defined [35,36].
Gene-environment gene-interactions not fully mapped: Rs10830963 interacts with dietary glycaemic index, carbohydrate amounts, weight-loss results, melatonin use, and lifestyle changes during pregnancy [36,38]. However, the total and long-term results of these adjustable factors on stage-specific worsening are unknown.
Future Research Directions
Prospective multistage cohort studies: large groups with repeated oral glucose tolerance tests grouped by MTNR1B genotype are vital to measure stage risks and spot beta-cell fail points [35,37,39].
Lifespan and reproductive trajectory analyses: Research should track rs10830963 carriers from youth through pregnancy and old age to see how life stages change genotype impacts [35,36].
Deep study of circadian and melatonin pathways: Combine actigraphy, dim-light melatonin onset/offset measurement, meal timing, and MTNR1B genotyping to check if changing sleep-wake/eat habits delays worsening in at-risk carriers [38].
Trial studies with genotype stratification: Controlled tests of diets, time-restricted eating, melatonin or rhythm-maintaining sleep methods and tailored lifestyle should assess if rs10830963 changes how well treatments work to reverse prediabetes [36,38].
Functional work on rare variants across targets: Inquiries-laboratories and participant-based, must explore how different rare MTNR1B versions change beta-cell activity and long-term glycaemic levels [34].
Conclusion
The shift from prediabetes to type 2 diabetes involves complex genetic and environmental interactions, with the MTNR1B variant rs10830963 playing a key role in early beta-cell failure. Evidence from large studies highlights its stronger link with prediabetic states, where it increases melatonin’s blocking effects on insulin release, leading to higher sugar levels. As the disease advances to full T2D, its influence drops, potentially hidden by other factors like insulin resistance. Factors like shift work, inactive lifestyles, and diet worsen risks, while changes—diet, exercise, and sleep habits—offer hope to slow worsening, reducing rates by up to 50%. Ultimately, adding rs10830963 testing to screening could lead to early, targeted steps to stop the path from prediabetes to T2D, focusing on personalized care in high-risk groups.
References
- Li, Y.-Y., Wang, H., & Zhang, Y.-Y. (2022). Melatonin receptor 1B gene rs10830963 C/G polymorphism associated with type 2 diabetes mellitus: An updated meta-analysis of 13,752 participants. Heliyon, 8(11):e11786.
Publisher | Google Scholor - Rosta, K., Al-Aissa, Z., Hadarits, O., Harreiter, J., Nádasdi, Á., Kelemen, F., Bancher-Todesca, D., et al. (2017). Association study with 77 SNPs confirms the robust role for the rs10830963/G of MTNR1B variant and identifies two novel associations in gestational diabetes mellitus development. PLOS ONE, 12(1):e0169781.
Publisher | Google Scholor - Sparsø, T., Bonnefond, A., Andersson, E., Bouatia-Naji, N., Holmkvist, J., et al. (2009). G-allele of intronic rs10830963 in MTNR1B confers increased risk of impaired fasting glycemia and type 2 diabetes through impaired glucose-stimulated insulin release. Diabetes, 58(6):1450–1456.
Publisher | Google Scholor - Lane, J. M., Chang, A.-M., Bjonnes, A. C., Aeschbach, D., Anderson, C., et al. (2016). Impact of common diabetes risk variant in MTNR1B on sleep, circadian, and melatonin physiology. Diabetes, 65(6):1741–1751.
Publisher | Google Scholor - Shen, L. L., & Jin, Y. (2019). Effects of MTNR1B genetic variants on the risk of type 2 diabetes mellitus: A meta-analysis. Molecular Genetics & Genomic Medicine, 7(5):e611.
Publisher | Google Scholor - Vella, M., Mohan, S., Christie, H., Bailey, K. R., Cobelli, C., et al. (2024). Diabetes-associated genetic variation in MTNR1B and its effect on islet function. Journal of the Endocrine Society, 8(8):bvae130.
Publisher | Google Scholor - Vejražková, D., Vaňková, M., Včelák, J., Krejčí, H., Anděrlová, K., et al. (2022). The rs10830963 polymorphism of the MTNR1B gene: Association with abnormal glucose, insulin, and C-peptide kinetics. Frontiers in Endocrinology, 13:868364.
Publisher | Google Scholor - Song, J. F., Zhang, J., Zhang, M. Z., Ni, J., Wang, T., Zhao, Y. Q., & Khan, N. U. (2021). Evaluation of the effect of MTNR1B rs10830963 gene variant on the therapeutic efficacy of nateglinide in type 2 diabetes among Chinese Han patients. BMC Medical Genomics, 14(1):156.
Publisher | Google Scholor - National Center for Biotechnology Information. (n.d.). rs10830963 (Reference SNP Report). dbSNP.
Publisher | Google Scholor - Tuomi, T., Nagorny, C. L. F., Singh, P., Bennet, H., Yu, Q., & Alenkvist, I., et al. (2016). Increased melatonin signaling is a risk factor for type 2 diabetes. Cell Metabolism, 23, 1067–1077.
Publisher | Google Scholor - Prokopenko, I., Langenberg, C., Florez, J. C., Saxena, R., Soranzo, N., Thorleifsson, G., et al. (2009). Variants in the melatonin receptor 1B gene influence fasting glucose levels. Nature Genetics, 41, 77–81.
Publisher | Google Scholor - Bouatia-Naji, N., Bonnefond, A., Cavalcanti-Proença, C., Sparsø, T., Holmkvist, J., Marchand, M., et al. (2009). A variant near MTNR1B is associated with increased fasting plasma glucose levels and type 2 diabetes risk. Nature Genetics, 41:89–94.
Publisher | Google Scholor - Gaulton, K. J., Ferreira, T., Lee, Y., Raimondo, A., Mägi, R., Reschen, M. E., et al. (2015). Genetic fine-mapping and genomic annotation define causal mechanisms at type 2 diabetes susceptibility loci. Nature Genetics, 47:1415–1425.
Publisher | Google Scholor - Garaulet, M., Gómez-Abellán, P., Rubio-Sastre, P., Madrid, J. A., Saxena, R., & Scheer, F. A. J. L. (2015). Common type 2 diabetes risk variant in MTNR1B worsens the deleterious effect of melatonin on glucose tolerance in humans. Metabolism, 64:1650–1657.
Publisher | Google Scholor - Tan, X., Ciuculete, D.-M., Schiöth, H. B., & Benedict, C. (2020). Associations between chronotype, MTNR1B genotype, and risk of type 2 diabetes in the UK Biobank. Journal of Internal Medicine, 287:189–196.
Publisher | Google Scholor - Shen, L., Wang, Z., Zang, J., Liu, H., Lu, Y., & He, X., et al. (2023). Association between dietary iron intake, MTNR1B rs10830963, and glucose metabolism in a Chinese population. Nutrients, 15:1986.
Publisher | Google Scholor - Ngwa, N. E., Matshazi, D. M., Davison, G. M., Kengne, A. P., & Matsha, T. E. (2023). Association between MTNR1B, HHEX, SLC30A8, and TCF7L2 polymorphisms and cardiometabolic risk in a mixed-ancestry South African population. Scientific Reports, 13:17122.
Publisher | Google Scholor - Tabák, A. G., Herder, C., Rathmann, W., Brunner, E. J., & Kivimäki, M. (2012). Prediabetes: A high-risk state for diabetes development. The Lancet, 379(9833):2279–2290.
Publisher | Google Scholor - Weir, G. C., & Bonner-Weir, S. (2004). Five stages of evolving beta-cell dysfunction during progression to diabetes. Diabetes, 53(Suppl. 3):S16–S21.
Publisher | Google Scholor - Johnson, M. C., Jr., Fishbein, H. A., Birch, R. J., Yu, Q., Mardon, R., Pace, W., Ritchie, N., Carroll, J. K., & Meeker, D. (2021). HbA1c evidence for a prediabetes diagnosis delays onset of type 2 diabetes. Journal of Endocrinological Science, 3(1):1–15.
Publisher | Google Scholor - Pani, L. N., Korenda, L., Meigs, J. B., Driver, C., Chamany, S., Fox, C. S., Sullivan, L., D’Agostino, R. B., & Nathan, D. M. (2008). Effect of aging on A1C levels in individuals without diabetes. Diabetes Care, 31(10):1991–1996.
Publisher | Google Scholor - Morris, D. H., Khunti, K., Achana, F., Srinivasan, B., Gray, L. J., Davies, M. J., & Webb, D. (2013). Progression rates from HbA1c-defined prediabetes to type 2 diabetes: A meta-analysis. Diabetologia, 56(7):1489–1493.
Publisher | Google Scholor - Kim, C. H., Kim, H. K., Kim, E. H., Bae, S. J., Choe, J., & Park, J. Y. (2016). Risk of progression to diabetes from prediabetes defined by HbA1c or fasting plasma glucose criteria in Koreans. Diabetes Research and Clinical Practice, 118:105–111.
Publisher | Google Scholor - Rodgers, L. R., Hill, A. V., Dennis, J. M., Craig, Z., May, B., Hattersley, A. T., McDonald, T. J., Andrews, R. C., Jones, A., & Shields, B. M. (2021). Choice of HbA1c threshold for identifying individuals at high risk of type 2 diabetes. BMC Medicine, 19(1):184.
Publisher | Google Scholor - Mahajan, A., Spracklen, C. N., Zhang, W., Ng, M. C. Y., Petty, L. E., Kitajima, H., et al. (2022). Multi-ancestry genetic study of type 2 diabetes highlights the power of diverse populations. Nature Genetics, 54(5):560–572.
Publisher | Google Scholor - Hu, S., Ji, W., Zhang, Y., Zhu, W., Sun, H., & Sun, Y. (2025). Risk factors for progression to type 2 diabetes in prediabetes: A systematic review and meta-analysis. BMC Public Health, 25(1):1220.
Publisher | Google Scholor - Karamitri, A., & Jockers, R. (2019). Melatonin in type 2 diabetes mellitus and obesity. Nature Reviews Endocrinology, 15(2):105–125.
Publisher | Google Scholor - Fuchsberger, C., Flannick, J., Teslovich, T. M., Mahajan, A., Agarwala, V., Gaulton, K. J., et al. (2016). The genetic architecture of type 2 diabetes. Nature, 536(7614):41–47.
Publisher | Google Scholor - Dietrich, S., Jacobs, S., Zheng, J. S., Meidtner, K., Schwingshackl, L., & Schulze, M. B. (2019). Gene–lifestyle interaction on risk of type 2 diabetes. Obesity Reviews, 20(11):1557–1571.
Publisher | Google Scholor - Zucatti, K. P., Teixeira, P. P., Wayerbacher, L. F., Piccoli, G. F., Correia, P. E., Fonseca, N. K. O., et al. (2022). Long-term effect of lifestyle interventions on cardiovascular and all-cause mortality in prediabetes and type 2 diabetes. Diabetes Care, 45(11):2787–2795.
Publisher | Google Scholor - Sørensen, K. V., Justesen, J. L., Ängquist, L., et al. (2025). Rare MTNR1B variants causing diminished MT2 signaling associate with elevated HbA1c levels but not type 2 diabetes. Diabetologia.
Publisher | Google Scholor - Powe, C. E., & Kwak, S. H. (2020). Genetic studies of gestational diabetes and glucose metabolism in pregnancy. Current Diabetes Reports, 20(11):69.
Publisher | Google Scholor - Wu, J., & Tan, X. (2022). The role of MTNR1B polymorphism in circadian rhythm-related cancer. International Journal of Cancer, 150(11):1815–1823.
Publisher | Google Scholor - Xia, Q., Chen, Z., Wang, Y., et al. (2012). Association between MTNR1B gene polymorphism and risk of type 2 diabetes. PLOS ONE, 7(11):e50107.
Publisher | Google Scholor