Research Article
Predictive Value of Tumor Burden for Oncologic and Functional Outcomes After Post-Chemotherapy Retroperitoneal Lymph Node Dissection
- Molgado-Garza Víctor Manuel ID 1*
- Madero-Morales Pedro Antonio ID 1*
- González-Garza Raquel ID 1
- Cárdenas-Álvarez Héctor Alejandro 1
- Menchaca-Martínez Oziel 2
- Gutiérrez-González Adrián ID 1
1 Service of Urology, University Hospital ‘José Eleuterio González,’ Autonomous University of Nuevo León, México.
2 School of Medicine, Autonomous University of Nuevo León, México.
*Corresponding Author: Madero-Morales Pedro Antonio, Service of Urology, University Hospital ‘José Eleuterio González,’ Autonomous University of Nuevo León, México.
Citation: Molgado-Garza VM, Madero-Morales PA, González-Garza, R, Cárdenas-Álvarez HA, Menchaca-Martínez O, et al. (2026). Predictive Value of Tumor Burden for Oncologic and Functional Outcomes After Post-Chemotherapy Retroperitoneal Lymph Node Dissection, International Clinical and Medical Case Reports, BioRes Scientia Publishers. 5(1):1-9, DOI: 10.59657/2837-5998.brs.26.059
Copyright: © 2026 Pedro Antonio Madero Morales, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: December 18, 2025 | Accepted: January 05, 2026 | Published: January 16, 2026
Abstract
Objective: Post-chemotherapy retroperitoneal lymph node dissection (PC-RPLND) for residual masses is a technically demanding but potentially curative procedure. Our purpose is to describe the oncologic and functional outcomes of patients undergoing PC-RPLND at a referral center in Northeastern Mexico.
Methods: A retrospective, correlational, and analytical study was conducted on 21 patients treated between January 2020 and April 2025. Demographic, surgical, and histopathological variables were analyzed using χ², Mann-Whitney U, Fisher’s exact tests, and Spearman’s correlations.
Results: The mean age was 25.3 years (range 16-36). The most frequent clinical stage at surgery was IIIB (57.2%). Standard PC-RPLND was performed in 66.7%, salvage in 23.8%, and desperation in 9.5%. The open approach predominated (85.7%) with a mean operative time of 304 minutes. Complications occurred in 33.3%, mostly Clavien I, with no perioperative mortality. Complete remission was achieved in 76.2%, while recurrence occurred in 23.8%. Teratoma (55%) and necrosis (35%) were the most frequent histologies. The presence of viable malignancy increased the risk of recurrence by 31-fold (p < 0.001). Tumor size and volume correlated with greater vascular involvement (r = 0.73, p = 0.002) and complication severity (r = 0.56, p = 0.007). Masses larger than 10 cm were associated with a 90% rate of retrograde ejaculation.
Conclusions: PC-RPLND provides adequate oncologic control with acceptable morbidity. Tumor size and viable malignancy remain the strongest predictors of recurrence and ejaculatory dysfunction in patients with post-chemotherapy residual masses.
Keywords: tumor; oncologic outcomes; post-chemotherapy; retroperitoneal lymph node dissection; 21 patients; teratoma; necrosis; histologies; vascular involvement; retrograde ejaculation; dysfunction
Introduction
Since the introduction of platinum-based chemotherapy for germ cell tumors (GCTs), the role of retroperitoneal lymph node dissection (RPLND) has shifted toward managing post-chemotherapy residual masses rather than serving as a primary treatment modality [1]. Persistent elevation of tumor markers after orchiectomy indicates retroperitoneal disease, which is usually managed with systemic chemotherapy [2]. Histologically, 6-10% of residual masses contain viable tumor, 50% teratoma, and 40% fibrosis or necrosis. When recurrence occurs more than two years after chemotherapy, 54-88% of patients may have viable tumor and 12-28% teratoma, the latter carrying up to a 20% risk of somatic malignant transformation into adenocarcinoma [1-3].
Treatment algorithms following systemic therapy can be complex due to the limited experience many surgeons have with RPLND. Lowrance et al. showed that few urologists are high-volume surgeons, and most U.S. residents finish training with two or fewer cases as primary surgeon; only 10% perform more than nine procedures as first assistant, reflecting limited exposure to this technically demanding operation [4]. Centralized care at high-volume centers is crucial: Wells et al. reported an average of six post-chemotherapy nerve-sparing RPLNDs per year, but treatment at referral centers significantly reduced mortality and perioperative complications (6% to 0.8%), recurrence rates (16% to 3%), and improved complete resection rates [5-8].
Different clinical contexts define the indication for surgery: standard RPLND follows first-line chemotherapy with normalized tumor markers; salvage RPLND occurs after a second regimen with subsequent marker normalization; desperation RPLND is performed after two or more chemotherapy cycles with persistently elevated markers, representing chemoresistance; and redo RPLND refers to recurrence after prior surgery, as classified by Indiana University [9].
According to the latest European Association of Urology (EAU) guidelines, restaging imaging for residual masses should occur 3-4 weeks after completion of the final chemotherapy cycle, and RPLND should be performed within 6-8 weeks thereafter [10]. A contrast-enhanced CT within six weeks and tumor markers measured within 10 days before surgery are recommended. The procedure is technically challenging due to chemotherapy-induced desmoplastic reaction, particularly in salvage, desperation, or redo cases, which carry higher complication rates [11].
Given the young age of most patients, nerve-sparing retroperitoneal lymph node dissection (NS-RPLND) is preferred to preserve ejaculatory function. This requires meticulous identification of sympathetic roots and careful dissection along major vessels. Historically, NS-RPLND involves removal of lymphatic chains from the infrarenal to the interiliac level at the aortic bifurcation, giving rise to modified templates based on the laterality of the primary testicular tumor [12].
Additional procedures are frequently necessary to achieve complete resection, reported in 23-45% of large series [12]. Radical nephrectomy is the most common ancillary procedure (5-31%), particularly on the left side due to proximity to the renal hilum. Moreover, vascular interventions are required in up to 15% of cases, with aortic resection or replacement performed in up to 40% [13]. Meta-analyses have identified predictors for adjunctive surgery, the most significant being residual masses larger than 10 cm in diameter [10].
Oncologic outcomes correlate strongly with histology. Patients with fibrosis or teratoma generally have excellent prognoses, whereas viable malignancy predicts worse survival. Cancer-specific and radiographic progression-free survival reach 95% in patients with fibrosis, 85-95% in those with teratoma, and only 50-70% in those with viable malignancy, reflecting resistance to systemic therapy [12].
From a functional standpoint, RPLND aims to achieve oncologic control while preserving quality of life. Although spermatogenesis remains unaffected, ejaculation mechanisms can be compromised. Surgical modifications, such as nerve-sparing techniques and lateralized templates based on primary tumor side, have significantly improved outcomes. Meticulous dissection to avoid traction and nerve injury (neurapraxia) allows preservation of antegrade ejaculation in 75-87% of patients [13].
Raja et al. demonstrated that nerve preservation is feasible in only 30% of post-chemotherapy RPLNDs, with 57.8% of these patients maintaining antegrade ejaculation after a mean follow-up of 6.8 months [14]. With the advent of minimally invasive approaches, including laparoscopic and robot-assisted techniques, retrospective series report antegrades ejaculation rates as high as 86.5% [15]. However, such outcomes depend heavily on patient selection, as desmoplastic reactions are particularly intense in seminomatous disease, often limiting the feasibility of nerve-sparing procedures.
In summary, post-chemotherapy RPLND remains an essential procedure in the management of residual retroperitoneal masses in testicular cancer. While the procedure is technically demanding and carries a risk of vascular and functional complications, its role in achieving long-term oncologic control is well established. Centralization to experienced, high-volume centers, adherence to standardized surgical templates, and nerve-sparing techniques are critical to optimizing outcomes. Tumor histology, size, and response to chemotherapy remain key determinants of prognosis and surgical complexity.
Materials and Methods
A correlational, retrospective, cross-sectional, and analytical study was conducted to describe the oncologic and functional outcomes of patients undergoing post-chemotherapy retroperitoneal lymph node dissection (PC-RPLND) at a referral hospital in Northeastern Mexico. Case selection was performed through the review of medical records available in the Service of Urology of the University Hospital “José Eleuterio González” of the Autonomous University of Nuevo León, corresponding to the period between January 2020 and April 2025. The present study was reviewed and approved by the hospital’s ethics committee under approval registration number UR:25-00003.
Patients included were males with histopathologically confirmed diagnosis of testicular cancer; retroperitoneal disease treated with at least one cycle of platinum-based or second-line chemotherapy; and had undergone post-chemotherapy retroperitoneal lymph node dissection (PC-RPLND), regardless of the surgical approach (open or laparoscopic). Only medical records containing complete information regarding oncologic management before, during, and after surgery were analyzed. Exclusion criteria comprised patients with testicular cancer and metastatic disease to solid organs, those who underwent primary RPLND without prior platinum-based chemotherapy, and those with unresectable residual masses.
Descriptive and inferential statistics were used to evaluate demographic, surgical, pathological, and outcome variables. Quantitative variables such as age, tumor diameter, surgical time, and hospital stay were summarized as means with standard deviations or medians with interquartile ranges, according to data distribution. Categorical variables, including clinical stage, type of lymphadenectomy (standard, salvage, or desperation), surgical approach (open or laparoscopic), histological findings, and complication grades, were expressed as absolute and relative frequencies.
Comparisons between categorical variables were made using the Chi-square test, and when expected frequencies were below five, the Fisher’s exact test was applied. The Mann–Whitney U test was used to compare continuous variables between independent groups, such as tumor diameter, volume, and surgical time in patients with versus without retrograde ejaculation or recurrence. For comparisons involving more than two groups, the Kruskal-Wallis test was performed.
To explore associations between continuous variables, including tumor diameter, volume, vascular involvement, and Clavien–Dindo classification, Spearman’s rank correlation coefficient (ρ) was calculated. Correlation strength was interpreted as weak (ρ < 0.4), moderate (ρ = 0.4-0.7), or strong (ρ > 0.7).
For binary outcomes such as recurrence, remission, and retrograde ejaculation, 2×2 contingency tables were constructed, and relative risks (RR) and odds ratios (OR) were estimated with 95% confidence intervals (95% CI). Given the small sample size and presence of perfect separation in some categorical outcomes, Firth’s penalized logistic regression was used in exploratory multivariate models to obtain bias-reduced and robust OR estimates.
All analyses were two-tailed, and a p value < 0.05 was considered statistically significant. Statistical analyses were conducted using IBM SPSS Statistics, version 25 (IBM Corp., Armonk, NY, USA) and GraphPad Prism, version 10 (GraphPad Software, San Diego, CA, USA) for graphical representation of data.
Results
A total of 21 patients were included in the study, with a mean age of 25.3 years (range 16–36). One patient aged 3 years was excluded. The most frequent clinical stage at the time of RPLND was IIIB, accounting for 57.2% of cases. According to the Indiana classification, 14 (66.7%) procedures were standard RPLND, 5 (23.8%) were salvage RPLND, 2 (9.5%) were desperation RPLND, and no “redo” cases were performed. The open approach was predominant (85.7%, n = 18), while the laparoscopic approach was used in 14.3% (n = 3), with a mean operative time of 304.4 minutes (±100.2) (Table 1).
Table 1: Descriptive summary of demographic, clinical, and surgical variables in 21 patients who underwent post-chemotherapy RPLND. Most patients were clinical stage IIIB, with a mean age of 25.3 years, predominance of the open approach, and a mean operative time of 304 minutes.
| Variable | Parameter | N (%) |
| Age (years) | Mean (± S.D.) | 25.3 (± 6.4) |
| Min. | 16 | |
| Max. | 36 | |
| Clinical Stage | IB | 1 (4.7%) |
| IIB | 6 (28.6%) | |
| IIIB | 12 (57.2%) | |
| IIIC | 2 (9.5%) | |
| Histology | Necrosis | 7 (35%) |
| Teratoma | 11 (55%) | |
| Choriocarcinoma | 1 (5%) | |
| Viable Malignancy | 2 (5%) | |
| Mean Mass Diameter (cm) | Mean (± S.D.) | 7.2 (± 4.9) |
| Mean Mass Volume (cm) | Mean (± S.D.) | 216.4 (± 461.2) |
| Surgical Approach | Open | 18 (85.7%) |
| Laparoscopic | 3 (14.3%) | |
| Surgical Time (minutes) | Mean (± S.D.) | 304.4 (± 100.2) |
| Standard Line | 14 (66.67%) | |
| Salvatage Line | 5 (23.8%) | |
| Desperation Line | 2 (9.5%) | |
| *Re-Do* Line | 0 (0%) | |
| Remission | 16 (76.2%) | |
| Recurrence | 5 (23.8%) | |
| Resectability | R0 | 15 (71.4%) |
| R1 | 2 (9.5%) | |
| R2 | 4 (19.05%) |
S.D. (Standard Deviation).
The overall rate of perioperative and early postoperative complications (within 30 days) was 33.3% (n = 7), most of which were Clavien–Dindo grade I (66.7%), with only three complications classified as Clavien–Dindo IIIa or higher. No intraoperative or procedure-related deaths were reported. Among additional procedures performed, four patients underwent cavorrhaphy due to vena cava injury, one pulmonary metastasectomy, one mesenteric defect closure, one right double-J stent placement, one appendectomy, and one left nephrectomy. No additional procedures were required in 53.2% (n = 11) of the remaining patients. The mean hospital stay was 2.4 days, and only one patient experienced complications beyond 30 days. Complete remission was achieved in 76.2% (n = 16), while recurrence occurred in 23.8% (n = 5). Regarding resectability, 71.4% (n = 15) achieved R0 status, 9.5% (n = 2) R1, and 19.0% (n = 4) R2. Comparison of perioperative complications between open and laparoscopic approaches showed no significant difference (p = 0.5211).
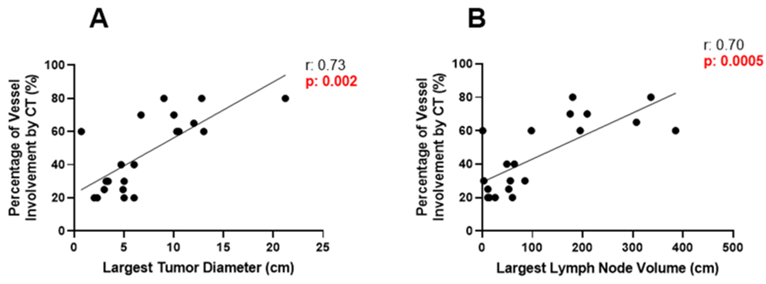
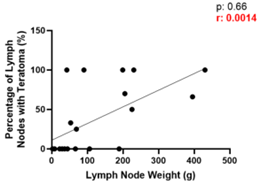
The mean diameter of residual masses was 7.2 cm (±4.9), and the mean volume was 216.4 cm³ (±461.2). The most common histopathological findings were teratoma in 55% (n = 11), necrosis in 35% (n = 7), viable malignancy in 5% (n = 1), and choriocarcinoma in 5% (n = 1) (Table 2). Moderate-to-strong positive correlations were found between maximum tumor diameter (r = 0.73, p = 0.002) and volume (r = 0.70, p = 0.0005) with the percentage of vascular involvement observed on CT imaging, indicating that larger tumors are associated with greater vascular compromise (Figure 1A and 1B). A moderate positive correlation was also found between lymph node weight and the percentage of nodes containing teratoma (r = 0.66, p = 0.0014), suggesting that larger nodal masses are more likely to contain teratomatous elements (Figure 2).
Table 2: Summary of perioperative outcomes and complications classified by Clavien–Dindo. The overall complication rate was 33.3%, mostly grade I. Mean hospital stay was 2.4 days, and retrograde ejaculation occurred in 47.6% of cases.
| Variable | Parameter | N (%) |
| Additional Procedure during RPLND | Cavorraphy | 4 (19.05%) |
| Pulmonary Metastasectomy | 1 (4.76%) | |
| Closure of Mesenteric Gap | 1 (4.76%) | |
| Left Nephrectomy | 1 (4.76%) | |
| Right Ureteral Catheter Placement | 1 (4.76%) | |
| Appendicectomy | 1 (4.76%) | |
| No Additional Procedure | 11 (52.38%) | |
| Perioperative Complications | 7 (33.3%) | |
| Complications within first 30 days | 3 (14.2%) | |
| Complications Further than 30 days | 1 (4.76%) | |
| Clavien-Dindo Classification | I | 14 (66.7%) |
| II | 4 (19.02%) | |
| IIIa | 3 (14.28%) | |
| Retrograde Ejaculation | Yes | 10 (47.62%) |
| No | 11 (52.38%) | |
| Mean Hospital Stay (days) | Mean (±S.D.) | 2.4 (±2) |
RPLND (Retroperitoneal Lymph Mode Dissection), S.D. (Standard Deviation).
Figure 1: (A) Moderate-to-strong positive correlation between maximum tumor diameter and percentage of vascular involvement on CT (r = 0.73, p = 0.002). (B) Strong positive correlation between the largest lymph node volume and vascular involvement (r = 0.70, p = 0.0005). These findings indicate that larger or more voluminous retroperitoneal masses are associated with greater vascular compromise.
Figure 2: Moderate positive correlation between lymph node weight and percentage of nodes containing teratoma (r = 0.66, p = 0.0014). The larger the mass, the higher the likelihood of being a teratoma.
Patients without viable malignancy were 11 times more likely to achieve complete remission compared with those who presented viable malignant components (Table 3). Although the confidence interval was wide due to the small sample size, the association remained statistically significant (p < 0 xss=removed>0% viable malignancy had an approximately 31-fold higher risk of recurrence compared with those with 0% viable malignancy (Table 4). The presence of teratoma was not associated with a higher or lower probability of recurrence or remission (RR = 0.8, OR = 0.4, p = 0.6108).
Table 3: Patients without viable malignancy were significantly more likely to achieve complete remission (p = 0.0001), with a high odds ratio (OR = 363).
| Measure | Value | CI 95% | P value |
| Fisher’s exact test | - | - | 0.0001 |
| Relative Risk (RR) | 11.65 | 0.82-165.70 | - |
| Odds Ratio (OR) | 363.0 | 6.41-∞ | - |
CI (Confidence Interval).
Table 4: Presence of viable malignancy was strongly associated with recurrence (p less than 0.0001), with a 31-fold higher relative risk compared to patients without viable malignancy.
| Measure | Value | CI 95% | P value |
| Fisher’s exact test | - | - | < 0.0001 |
| Relative Risk (RR) | 31.17 | 2.01-483.37 | - |
| Odds Ratio (OR) | 0.0027 | 0.000-0.156 | - |
CI (Confidence Interval).
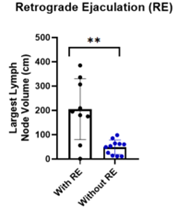
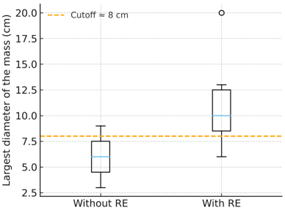
The volume of the largest lymph node was significantly higher in patients who developed retrograde ejaculation (p = 0.0097) (Figure 3). The median tumor diameter in patients without retrograde ejaculation was 4.8 cm, compared with 10.2 cm in those who developed this condition (p = 0.016) (Figure 4). Exploratory subgroup analysis showed that patients with residual masses between 8 and 10 cm accounted for most cases of retrograde ejaculation, suggesting a clinical threshold beyond which the risk of postoperative ejaculatory dysfunction increases markedly. Indeed, 90% of patients with retrograde ejaculation had retroperitoneal masses larger than 10 cm (Table 5).
Figure 3: Patients with retrograde ejaculation had a significantly larger lymph node volume compared to those without (p = 0.0097).
Figure 4: Median tumor diameter was significantly larger in patients with retrograde ejaculation (10.2 cm) compared to those without (4.8 cm; p = 0.016).
Table 5: Patients with residual masses ≥8 cm showed an increased likelihood of developing retrograde ejaculation, with a progressive rise in risk beyond 10 cm (90% of cases).
| Diameter Cutoff Point | Patients with RE (%) | Sensitivity | Specificity |
| ≥ 6 cm | 62% | 0.8 | 0.88 |
| ≥ 8 cm | 75% | 0.7 | 0.7 |
| ≥ 10 cm | 90% | 0.6 | 0.85 |
RE (Retrograde Ejaculation).
The correlation between surgical variables and postoperative complications according to the Clavien–Dindo classification was assessed using Spearman’s rank correlation. No significant association was found between operative time and Clavien grade (ρ = 0.33, p = 0.14) or between the number of resected lymph nodes and Clavien grade (ρ = 0.13, p = 0.57). In contrast, significant positive correlations were observed between lymph node weight and Clavien grade (ρ = 0.55, p = 0.009), maximum tumor diameter and Clavien grade (ρ = 0.56, p = 0.007), and tumor volume and Clavien grade (ρ = 0.56, p = 0.009). These findings suggest that larger tumor size is associated with greater postoperative complication severity (Table 6).
Table 6: Significant positive correlations were found between node weight, largest mass diameter, and tumor volume with Clavien grade, indicating that larger masses are associated with more severe postoperative complications.
| Variable correlated with Clavien-Dindo | P (Spearman) | P-value | CI 95% |
| Surgical Tim (min) | 0.3325 | 0.1409 | -0.1293 a 0.6757 |
| Nodes Weight (g) | 0.5538 | 0.0092 | 0.1471 a 0.8003 |
| Number of Nodes #) | 0.1297 | 0.5752 | -0.3321 a 0.5414 |
| Largest Mass Diameter (cm) | 0.5641 | 0.0077 | 0.1618 a 0.8056 |
| Largest Mass Volume (cm3) | 0.5567 | 0.0088 | 0.1513-0.8018 |
Discussion
RPLND, in any of its modalities, is a highly demanding procedure for both the patient and the urologist, which explains why it is infrequently performed in most training centers. Lowrance et al. reported that urology residents in the United States complete their training having performed two or fewer RPLNDs as the primary surgeon, and only 10% perform at least nine as first assistant [5]. In Mexico, few studies have reported the incidence of patients undergoing RPLND. The largest national cohort to date, published by Piñón-Solís et al., included 346 patients over 15 years, conducted at the largest referral center in the country [4]. Our series of 21 patients (22 including one excluded pediatric case) over a five-year period, while smaller, reflects real-world experience and demonstrates that comparable outcomes can be achieved at a regional reference center. Although long-term follow-up data were not available for all patients, only one death was recorded during the five-year period. Complete remission was achieved in 76.2% of patients, with a recurrence rate of 23.8%, slightly higher but comparable to that reported by the National Cancer Institute of Mexico (24.2%). The presence of viable malignancy remained the strongest predictor of recurrence, as noted in our results section [4].
As previously mentioned, patients who receive chemotherapy frequently develop desmoplastic changes near retroperitoneal masses, making dissection technically difficult and increasing the likelihood of complications or additional procedures. In our study, three intraoperative complications were recorded, with the remainder occurring within the first 30 postoperative days, yielding an overall complication rate of 33.3%. This rate correlated directly with retroperitoneal mass size and the percentage of major vessel involvement on CT imaging. Vascular complications were the most common (19.05%, n = 4), emphasizing the need for preparation when tumor volume exceeds 100 grams or diameter surpasses 5 cm. Winter et al. reported a 20% probability of vascular injury when tumor diameter is >5 cm; although our cohort was smaller, our findings are consistent with those data [12]. In our series, three patients (14.3%) required resection of an additional organ or structure, approximately half the incidence reported by Ehrlich et al., who observed this in up to one-third of cases [16]. When comparing open versus laparoscopic surgery, perioperative complications were 0% in the laparoscopic group, with only one event within 30 days; however, the sample size was too small for meaningful statistical comparison. Nazzani et al. analyzed 151 laparoscopic RPLNDs, reporting a 9% complication rate, although the median tumor size in that study was only 25 mm, suggesting that minimally invasive approaches may be less suitable for larger-volume disease [17].
Unlike other series, the most frequent histology in our cohort was teratoma (55%), followed by necrosis (35%) and viable malignancy (10%). Interestingly, patients with teratoma tended to have larger tumor volumes, showing a moderate-to-strong correlation. The identification of teratomatous features on imaging should raise clinical suspicion for teratoma, which may have surgical and prognostic implications. Although teratoma presence has not been shown to impact overall survival or recurrence risk, the patient in our series with the largest mass (21.2 cm) had teratoma and experienced recurrence within 12 months. Larger sample sizes are required to determine whether tumor volume influences recurrence risk in teratoma cases. Beck et al. reported that 27 patients with teratomas larger than 10 cm had a 25% recurrence rate at five years, underscoring the need for further studies to validate these findings [18].
Given the proximity of the sympathetic plexus and its L1-L4 roots to the retroperitoneal great vessels, urological complications such as retrograde ejaculation are not uncommon. A strength of our study is that it quantified the incidence of this outcome and proposed a practical threshold diameter above which retrograde ejaculation risk significantly increases, providing valuable information for preoperative counseling and surgical planning. Retrograde ejaculation was observed in 47.6% of patients, with the risk increasing sharply beyond an 8 cm mass diameter and reaching 90% when masses exceeded 10 cm. This finding aligns closely with Raja et al., who reported a 57.8% incidence, suggesting that the feasibility of nerve-sparing RPLND is largely dependent on tumor volume [14].
We acknowledge several limitations. The retrospective nature of this study restricts causal inference and external validation of our findings. However, the analysis highlights clinically relevant variables for surgical decision-making in post-chemotherapy RPLND. Although our sample size is limited, the procedure’s technical demands make it difficult to match the case volume of national reference centers such as the National Cancer Institute. Nonetheless, our outcomes demonstrate that comparable oncologic and functional results can be achieved in a high-volume academic training hospital in northeastern Mexico.
Conclusion
Post-chemotherapy RPLND is a complex surgical procedure typically performed in high-volume centers (HVCs) by urologists with specialized experience. Nevertheless, it has been shown to achieve favorable oncologic and functional outcomes that contribute to improved patient survival. Although the procedure is not free of complications, adequate preparation and multidisciplinary support are essential for their management.
It can be concluded that teratomas, being large-volume tumors, represent the histology most frequently associated with perioperative and postoperative complications, higher likelihood of vascular involvement, and the need for additional surgical procedures. Masses larger than 8 cm carry a high risk of retrograde ejaculation, making nerve preservation challenging in these patients. Moreover, the presence of any percentage of viable malignancy significantly increases the risk of recurrence compared with patients whose specimens show only necrosis or teratoma.
Our results reinforce the importance of careful patient selection, meticulous preoperative planning, and the centralization of care in high-volume centers, where optimal surgical and oncologic outcomes can be achieved. Although our sample size was limited, the findings highlight tumor volume and residual histology as key predictors of both oncologic and functional outcomes.
Declarations
Author Contribution Statement
Conception and Design: MGVM, MMPA, GGA
Data Acquisition: MGVM, MMPA, CAHA, MMO
Data Analysis and Interpretation: GGR
Drafting the Manuscript: MGVM, MMPA
Clinical Revision of The Manuscript for Scientific and Factual Context: MGVM, MMPA, GGR, GGA
Statistical Analysis: GGR
Supervision: MMPA, GGA
Conflicts of Interest
The authors declare no conflicts of interest.
Ethical Approval
The present study was approved by the Institutional Ethics Committee (approval no. UR:25-00003) and all participants provided written informed consent before enrollment.
Financial Support
Urology Department’s own resources.
References
- Stephenson, A. J., Sheinfeld, J. (2004). The Role of Retroperitoneal Lymph Node Dissection in The Management of Testicular Cancer. In Urologic Oncology: Seminars and Original Investigations. 22(3):225-233.
Publisher | Google Scholor - Stephenson AJ, Gilligan TD. (2021). Neoplasms of The Testis. In: Partin AW, Dmochowski RR, Kavoussi LR, Peters CA, editors. Campbell-Walsh-Wein Urology. Philadelphia: Elsevier; 1680-1710.
Publisher | Google Scholor - Hartmann, J. T., Candelaria, M., Kuczyk, M. A., Schmoll, H. J., Bokemeyer, C. (1997). Comparison of Histological Results from The Resection of Residual Masses at Different Sites After Chemotherapy for Metastatic Non-Seminomatous Germ Cell Tumours. European Journal of Cancer, 33(6):843-847.
Publisher | Google Scholor - Piñón-Solis, E. O., Jiménez-Ríos, M. A., Scavuzzo, A., Martínez-Cervera, P. F. (2021). Resultado oncológico de linfadenectomía retroperitoneal por tumor residual posquimioterapia en cáncer germinal. Análisis retrospectivo de 15 años. Cirugía y Cirujanos, 89(6): 703-709.
Publisher | Google Scholor - Lowrance, W. T., Cookson, M. S., Clark, P. E., Smith, J. A., Chang, S. S. (2007). Assessing retroperitoneal lymphadenectomy experience in United States urological residency programs. The Journal of Urology, 178(2):500-503.
Publisher | Google Scholor - European Association of Urology. EAU Guidelines on Testicular Cancer 2025.
Publisher | Google Scholor - Wells, H., Hayes, M. C., O'Brien, T., Fowler, S. (2017). Contemporary retroperitoneal lymph node dissection (RPLND) for testis cancer in the UK-a national study. BJU International, 119(1):91-99.
Publisher | Google Scholor - Capitanio, U., Jeldres, C., Perrotte, P., Isbarn, H., Crépel, M., et al. (2009). Population-based study of perioperative mortality after retroperitoneal lymphadenectomy for nonseminomatous testicular germ cell tumors. Urology, 74(2):373-377.
Publisher | Google Scholor - Fléchon, A., Tavernier, E., Boyle, H., Meeus, P., Rivoire, M., et al. (2010). Long‐term oncological outcome after post‐chemotherapy retroperitoneal lymph node dissection in men with metastatic nonseminomatous germ cell tumour. BJU International, 106(6):779-785.
Publisher | Google Scholor - Riggs S, Gaston K, Clark P. (2021). Surgery of Testicular Tumors. In: Partin AW, Dmochowski RR, Kavoussi LR, Peters CA, editors. Campbell-Walsh-Wein Urology. Philadelphia: Elsevier: 1711-1733.
Publisher | Google Scholor - Fitzgerald, J. P., Ercole, B., Parekh, D. J. (2010). Management of post-chemotherapy residual mass in patients with metastatic nonseminomatous germ cell tumors of the testis. Indian Journal of Urology, 26(1):98-101.
Publisher | Google Scholor - Winter, C., Pfister, D., Busch, J., Bingöl, C., Ranft, U., et al. (2012). Residual tumor size and IGCCCG risk classification predict additional vascular procedures in patients with germ cell tumors and residual tumor resection: a multicenter analysis of the German Testicular Cancer Study Group. European Urology, 61(2):403-409.
Publisher | Google Scholor - Heidenreich, A., Pfister, D. (2012). Retroperitoneal lymphadenectomy and resection for testicular cancer: an update on best practice. Therapeutic Advances in Urology, 4(4):187-205.
Publisher | Google Scholor - Raja, A., Malik, K., Kathiresan, N., Radhakrishnan, V. (2021). Nerve-sparing Postchemotherapy Retroperitoneal Lymph Node Dissection (PC RPLND) for nonseminomatous germ cell tumour: experience from a tertiary cancer centre. Indian Journal of Surgical Oncology, 12(2):374-377.
Publisher | Google Scholor - Vasudeo, V., Khanna, A., Pratihar, S. K., Jaipuria, J., Chakraborty, A., et al. (2023). Robot-assisted retroperitoneal lymph node dissection for post-chemotherapy residual mass in testicular cancer: Long-term experience from a tertiary care centre. Journal of Minimal Access Surgery, 19(2):288-295.
Publisher | Google Scholor - Ehrlich, Y., Brames, M. J., Beck, S. D., Foster, R. S., Einhorn, L. H. (2010). Long-term follow-up of cisplatin combination chemotherapy in patients with disseminated nonseminomatous germ cell tumors: is a postchemotherapy retroperitoneal lymph node dissection needed after complete remission? Journal of Clinical Oncology, 28(4):531-536.
Publisher | Google Scholor - Nazzani, S., Stagni, S., Biasoni, D., Catanzaro, M., Macchi, A., et al. (2023). Laparoscopic retroperitoneal lymph-node dissection in metastatic nonseminomatous germ-cell tumors. European Journal of Surgical Oncology, 49(1):257-262.
Publisher | Google Scholor - Beck, S. D., Foster, R. S., Bihrle, R., Einhorn, L. H., Donohue, J. P. (2009). Long-term outcome for patients with high volume retroperitoneal teratoma undergoing post-chemotherapy surgery. The Journal of Urology, 181(6):2526-2532.
Publisher | Google Scholor