Research Article
Enhancing Compliance with Central Venous Catheterization Guidelines: A Quality Improvement Audit in A Teaching Hospital
- Muhammad Shaheryar Bashir 1*
- Shahana Ghazal 2
- Muhammad Talha Mahmood 1
- Abdullah Khalil 1
- Aiman Nazir 1
- Muhammad Khurram 3
1Intern General Medicine, Department of General Medicine, Tertiary Care Hospital, Rawalpindi, Pakistan.
2Resident Physician General Medicine, Department of General Medicine, Tertiary Care Hospital, Rawalpindi, Pakistan.
3Professor & Head of Department, Department of General Medicine, Tertiary Care Hospital, Rawalpindi, Pakistan.
*Corresponding Author: Muhammad Shaheryar Bashir, Intern General Medicine, Department of General Medicine, Tertiary Care Hospital, Rawalpindi, Pakistan.
Citation: Bashir MS, Ghazal S, Mahmood MT, Khalil A, Nazir A, et al. (2026). Enhancing Compliance with Central Venous Catheterization Guidelines: A Quality Improvement Audit in A Teaching Hospital. International Clinical and Medical Case Reports, BioRes Scientia Publishers. 5(2):1-7. DOI: 10.59657/2837-5998.brs.26.053
Copyright: © 2026 Muhammad Shaheryar Bashir, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: July 10, 2025 | Accepted: January 13, 2026 | Published: January 19, 2026
Abstract
Introduction: Central venous catheter (CVC) insertion is a critical yet high-risk procedure in medical care. This audit aimed to evaluate and enhance compliance with standardized guidelines for CVC insertion among postgraduate residents at a tertiary care hospital in Rawalpindi, Pakistan, to minimize central line-associated bloodstream infections (CLABSI).
Methods: A cross-sectional observational study was conducted in Medical and Nephrology units over two audit cycles spanning seven months. Practices of 34 postgraduate residents were evaluated using a structured checklist based on the guidelines from John Hopkins Medicine and the American Society of Anesthesiologists. A targeted intervention, including educational materials, was introduced post-initial audit, and compliance rates were reassessed through a re-audit.
Results: Initial compliance rates with key procedural practices were below satisfactory thresholds (<80%) in 26.4% of cases, particularly in areas like aseptic techniques and full barrier precautions. Post-intervention, re-audit results demonstrated significant improvements, with 94% of residents achieving adequate compliance scores (>80%). Improvements were particularly notable in the use of full barrier precautions (47.1% to 82.3%; p=0.002) and sterile draping (58.8% to 85.3%; p=0.0001).
Conclusions: Structured interventions, such as education and checklist-based guidance, significantly enhance compliance with standardized CVC practices. Regular audits and continuous education programs are recommended to sustain these improvements and reduce CLABSI risks in resource-constrained settings.
Keywords: catheter; guidelines; hopkins; sterile; aseptic
Introduction
Central venous catheterization (CVC) is a very common procedure performed across medical and surgical wards as well as intensive care units. It provides relatively extended vascular access for critically ill patients, in order to the administer intricate life-saving medications, blood products and parenteral nutrition [1].
Major vascular catheterization provides a risk of easy accessibility and dissemination of catheter related infections as well as venous thromboembolism. Therefore, it’s crucial to ensure following standardized practices while insertion and management of CVC in order to minimize the infection risks and procedural complications. The aim of these central line insertion guidelines is to address the primary concerns related to predisposition of Central line associated blood stream infections (CLABSI). These guidelines are evidence based and gathered from pre-existing data associated with CVC insertion [2].
The most common used sites for central venous catheterization are internal jugular and subclavian veins as compared to femoral veins. Catheterization of these vessels enables healthcare professionals to monitor hemodynamic parameters while ensuring lower risks of CLABSI and thromboembolism. Femoral vein is less preferred due to advantage of invasive hemodynamic monitoring and low risk of local infection and thromboembolic phenomena [3].
CVC can be inserted using Landmark guided technique and ultrasound guided techniques. Following informed consent, the aseptic technique for CVC insertion includes performing appropriate hand hygiene and ensuring personal protective measures, establishing and maintaining sterile field, preparation of the site using chlorhexidine, and draping the patient in a sterile manner from head to toe. Additionally, the catheter is prepared by pre-flushing and clamping all unused lumens, and the patient is placed in the Trendelenburg position. Throughout the procedure, maintaining a firm grasp on the guide wire is essential, which is subsequently removed post-procedure. It is followed by flushing and aspirating blood from all lumens, applying sterile caps, and confirming venous placement. Procedure is ended with cleaning the catheter site with chlorhexidine, and application of a sterile dressing [4].
Hence, formal training and knowledge of standardized practices of CVC insertion is essential for health care professionals in order to prevent CLABSI [5]. Our audit assesses the current practices of doctors working at a tertiary care hospital to analyze their background knowledge of standard practices to prevent CLABSI during insertion of CVC.
Rationale
Limited audits and quality improvement projects have been carried out in A Tertiary Care Hospital of Rawalpindi, Pakistan so the information obtained from this audit helps improve patient care by improving residents’ practice of CVC insertion.
Aim
This study was aimed to audit and re-audit residents’ practices of central venous line insertion in medical and nephrology units of A Tertiary Care Hospital of Rawalpindi, Pakistan and to assess the adherence of residents to checklist and practice guidelines of CVC insertion implemented by John Hopkins Hospital and American Society of Anesthesiologists.
Audit Hypothesis
The practices of residents working in medicine and nephrology units of A Tertiary Care Hospital of Rawalpindi, Pakistan are not in complete compliance with standard guidelines for CVC insertion.
Re-Audit Hypothesis
The practices of residents working in medicine and nephrology units of A Tertiary Care Hospital of Rawalpindi, Pakistan are improved in response to complete compliance with standard guidelines for CVC insertion.
Materials and Methods
Study Design: Cross sectional Direct Observational Study
Study Setting: Indoor wards of Medical Unit- 2 and Nephrology Unit of a Tertiary Care Hospital, Rawalpindi
Study Population
Post Graduate Residents working at Medical Unit-II and Nephrology Department of a Tertiary Care Hospital, Rawalpindi
Study Duration
7 months (1st cycle of audit from December 2023 to February 2024 & 2nd cycle of audit, data analysis and results compilation from April 2024 to June 2024).
Sample Size: 34
Sampling Technique: Convenience Sampling
Data Collection Technique: Direct observation and interview
Data Collection Tool: Structured Performa containing two parts, was used to gather data relevant to the study.
Inclusion Criteria: Post Graduate Residents working in the Medical and Nephrology units of a Tertiary Care Hospital, Rawalpindi who have passed atleast 5 supervised CVCs in either internal jugular, subclavian or femoral vein.
Exclusion Criteria: Post graduate residents with less than 6 months experience after induction were excluded from the study.
Methodology
This audit was conducted as a cross sectional direct observational study and two-phase quality improvement project in the Medical and Nephrology Units of a Tertiary Care Hospital of Rawalpindi from December 2023 to February 2024.
After taking informed consent from patients and residents, CVC insertion in 34 patients by 34 individual residents was observed. Observers were given a purposely designed observational tool made from John Hopkins Medicine checklist and ASA practice guidelines for central line insertion, for assessment of residents’ practices.
First part contained questions regarding the demographic details of residents such as age, gender, year of post graduate training, and parent department, and data related to the procedure such as date and time of procedure, need of CVC discussion during rounds, site of CVC insertion, catheter type and type of procedure (Landmark guided CVC or Ultrasound guided CVC insertion). Second part included direct observational checklist based on checklist provided for prevention of intravascular catheter-associated bloodstream infections to audit the practices of residents during CVC insertion that included: adequate hand hygiene before insertion, adherence to aseptic techniques, using sterile personal protective equipment and sterile full body drape of patient, choosing the best insertion site to minimize infections based on patient characteristics.
The parameters observed to be done completely were scored "1" and the items not done were scored "0". The cumulative percentage of performed practices according to checklist, was satisfactory if it was 80% or more and unsatisfactory if it was less than 80%.
After initial audit, participants were given pamphlets with checklist incorporating John Hopkins Medicine checklist and ASA practice guidelines for CVC insertion. Re audit was performed one month after the audit, including same participants who participated in initial audit. The results of audit and re-audit were analyzed using SPSS version 25. Mean +/- SD was calculated for quantitative variables and Number (N) percentage was calculated for qualitative variables. Z- Test was applied on proportions of parameters and test scores to calculate Z –score and P value (<0>
Field of work
The field of work encompassed a duration of 6 months from March 2024 to August 2024. It started by introduction of auditors with the participants during the official working hours at Medical Unit- 2 and Nephrology Wards of a Tertiary Care Hospital, Rawalpindi and a briefing about research purpose and their participation procedure. After informed consent, the audit was performed by interview and direct observation of residents during CVC insertion utilizing Performa.
The duration of each session varied from 20-40 minutes. In the first phase, audit was performed, the standard guidelines were provided to residents in the form of brochures, that included John Hopkins checklist along with American Society of Anesthesiologists practice guidelines for insertion of CVC. After one month, re-auditing was done in the same setting with the same participants.
Data Analysis
Data analysis was performed using SPSS version 25. Descriptive statistics were presented in the form of frequencies and percentages for qualitative variables, along with means and standard deviations for quantitative variables. P-value <0>
Results of Audit and Re-Audit
Among the 34 participants, 44% of the participants belonged to Nephrology Department and 56% of participants belonged to Department of Internal Medicine.
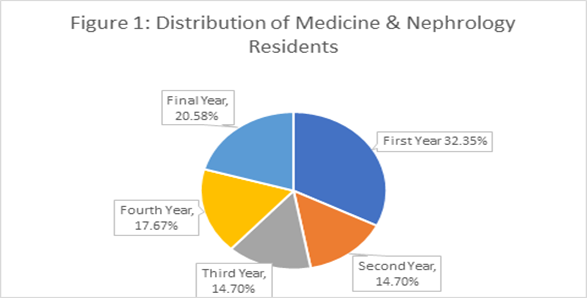
32.3% residents were in their first year, 14.7% in second, 14.7 in third year, 17.6% in fourth year and 17.6% in 5th/Final year of training. (Figure 1).
Figure 1: Distribution of Medicine and Nephrology Residents by Year of Postgraduate Training.
This figure illustrates the proportional distribution of postgraduate residents across different years of training within the Medicine and Nephrology units.
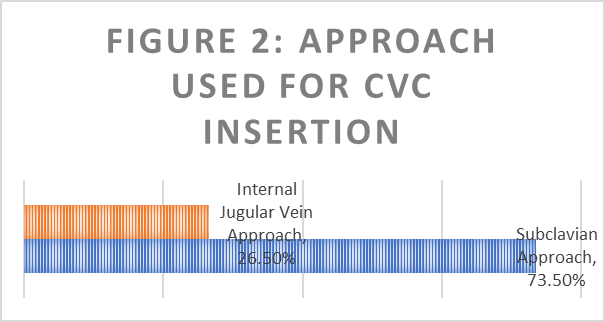
47% of the participants were male and 53% were female. Participants were aged between 27 and 34 years old, the median age at the time of audit was 29 years. Landmark guided CVC insertion was performed in Subclavian Vein (73.5%) and Internal Jugular Vein (26.5%). (Figure 2).
Figure 2: Approach Used for Central Venous Catheter (CVC) Insertion.
This figure highlights the proportions of CVC insertions performed via the subclavian approach versus the internal jugular vein approach.
Following comparative tabulation (Table-1) elicits the difference of results in re-audit to evaluate adherence to standard practices of CVC insertion after intervention.
Table-1: Comparison of CVC insertion practices: 1st cycle of Audit versus Re-Audit.
| Item | Parameters | Audit | Re-Audit | p-value |
| Indication of central line placement | Need discussed with consultants during round | 88.2% | 100% | 0.03 |
| Knowing optimal site for insertion | Selection of site based on clinical need, insertion site that is not contaminated or liable to be contaminated (not contaminated or liable to contamination (e.g., around inguinal area, infected or burned skin, site near to tracheostomy or near open surgical wound) and preferring upper body insertion site to minimize the risk of CLABSI) | 82.4% | 85.2% | 0.74 |
| Adequate Hand Hygiene | Handwashing with soap and water / scrubbing with chlorhexidine before procedure | 82.4% | 97% | 0.03 |
| Taking informed consent from patients or next of kin if patient doesn’t have capacity to make decisions | Yes | 88.2% | 100% | 0.03 |
| Adequate personal protective equipment | Using cap, mask, sterile gown/gloves, eye protection during procedure | 47.1% | 82.3% | 0.002 |
| Place patient in Trendelenburg position or place supine and flat if Femoral CVC | Yes (for Trendelenburg unless contraindicated (e.g., increased ICP) | 82.4% | 94% | 0.13 |
| Sterile procedure site | Scrub back and forth with Chlorhexidine with friction for 30 seconds and let air dry | 85.3% | 97% | 0.08 |
| Sterile technique to drape from head to toe | Yes | 58.8% | 85.3% | 0.0001 |
| Using local anesthetic around insertion site and/or sedation when needed | Yes | 94% | 100% | 0.15 |
| Maintaining a sterile field | Patient full body drape, sterile tray, ultrasound probe and all equipment for the procedure | 85.3% | 97% | 0.08 |
| Prepare catheter by pre-Flushing and clamping all lumens not in use during procedure | Yes | 82.4% | 85.3% | 0.74 |
| Ensure grasp on guide wire is maintained throughout procedure and removed post procedure. | Yes | 91% | 94% | 0.64 |
| Aspirate blood from all lumens, flush, and apply sterile caps | Yes | 85.3% | 94% | 0.23 |
| Ensure Venous Placement of Catheter | Either by assessing for appropriate fluid column response (the fluid and blood column should fall if venous, rise if arterial), Transducing CVP, Obtaining a blood gas or Fluoroscopy | 91% | 100% | 0.07 |
| Clean site with chlorhexidine, apply sterile dressing, and apply sterile caps on all hubs. | Yes | 88.2% | 97% | 0.16 |
| Order chest X-Ray to verify placement in case of (Internal Jugular and subclavian vein) | Yes | 88.2% | 91% | 0.69 |
| Documentation at the end of procedure | Yes | 85.3% | 97% | 0.08 |
| Practices Scoring (Figure 3) | Adequate Practices (% score =/> 80) | 73.5% | 94% | 0.02 |
| Inadequate Practices (% score less than80) | 26.5% | 6% |
Figure 3: Comparison of Practices Scoring: Pre-Audit vs. Post-Audit Compliance Rates.
This figure compares the percentage of residents demonstrating adequate and inadequate compliance with CVC insertion practices before and after the intervention.
Discussion
To our knowledge, this is the first audit and quality improvement project involving central line insertion practices among residents being done in tertiary care hospital of Rawalpindi. Our quality improvement project aimed to enhance the adherence to best practices in central venous catheter (CVC) insertion among postgraduate residents through an audit and re-audit cycle. Central line insertion practices play a pivotal role in acquiring preventable central line associated blood stream infections (CLABSI) which cause significant mortality in patients [6].
In order to achieve optimal infection control, implementation of “bundle” concept is necessary that includes five basic evidence-based interventions: adequate hand hygiene, draping CVC site with chlorhexidine, optimal barrier precautions for central line insertion, selection of best insertion site suited to patient needs, and prompt catheter removal [7].
A study done in Thailand established that incorporation of bundle practices during CVC insertion is not only effective in decreasing rate of CLABSI but also successful in maintaining reduced incidence of CLABSI in hospital settings with limited resources [8].
Developed countries such as United States (US) has been greatly successful in decreasing CLABSI rates by employing bundle practices over the past 10 years [6]. John Hopkins Medicine in United States recommends similar practices for prevention of CLABSI and formulated a checklist for healthcare professionals for safe insertion of CVC [9].
Survey findings in the guidelines for CVC insertion published by American society of Anesthesiologists (ASA) describe criteria of selection of optimal site for CVC insertion which is based on clinical need, not contaminated or liable to contamination (e.g., around inguinal area, infected or burned skin, site near to tracheostomy or near open surgical wound) and preferring upper body insertion site to minimize the risk of CLABSI in adults [10].
We determined a strong need for auditing the practices of central line insertion in residents because prevention of CLABSI is more cost effective and manageable than treatment of CLABSI in tertiary care hospitals of Pakistan. A study done in tertiary care NICU in Karachi established a significant reduction of CLABSI after introduction of CLABSI Prevention Package (CPP) [11].
Another study done in our tertiary care hospital demonstrated a significant level of contamination specially by Acinetobacter species of healthcare equipment also pointing towards importance of using sterile field and equipment during central line insertion.
In our study, we assessed the practices of residents in compliance with the John Hopkins Medicine and ASA practice guidelines for CVC insertion. During 1st cycle of audit, several inadequate critical practices, such as the use of full barrier precautions, sterile drape of patient from head to toe were found among residents working in the Medical and Nephrology Units of a Tertiary Care Hospital, Rawalpindi, with compliance rates below 80%.
26.4% of the residents had collective inadequate practices of safe CVC insertion. Following targeted intervention, including the introduction of a brochure containing John Hopkins checklist along with ASA guidelines for insertion of CVC, the re-audit results demonstrated significant improvements in these areas, with use of full barrier precautions, sterile drape of patient from head-to-toe compliance exceeding the 80% threshold and only 5.8% of the residents demonstrating collective inadequate practices (below 80% of total score).
The observed improvements are clinically significant (p value less than 0.05), suggesting a likely reduction in CLABSI due to better adherence to aseptic techniques. Our findings are consistent with those reported in similar studies, which have shown that education and procedural checklists can markedly enhance adherence to best practices.
Despite the positive outcomes, there are several limitations of this study such as relatively small sample size and short time duration between audit and re-audit that can affect the generalizability of our study. Additionally, reliance on self-reported compliance could introduce bias. Future studies should consider larger, multicenter designs and objective measures of compliance.
The immediate impact of our project is the enhanced safety and quality of CVC insertions within our institution. To sustain these improvements, we recommend regular audits, continuous education programs, and the integration of the checklist into the electronic health record system. Further research should explore the long-term effects of these interventions and identify additional strategies for quality improvement.
Conclusion
Our audit found that many of the residents adopted inadequate practices because of lack of proper training and institutional guidelines for CVC insertion. Our re-audit elaborated an improvement in the practices of residents following intervention with educational material. Our study underscores the importance of structured quality improvement initiatives in enhancing clinical practices and patient outcomes.
Abbreviations
CVC: Central Venous Catheter; CLABSI: Central Line-Associated Bloodstream Infection; ASA: American Society of Anesthesiologists
Declarations
Development of Observational Tool
The structured observational checklist used in this study was specifically developed for the purposes of this audit and has not been published elsewhere.
Ethics Approval and Consent to Participate
This study was conducted in compliance with the ethical principles outlined in the Declaration of Helsinki. Approval for the study was obtained from the Ethical Review Board (ERB) of the Department of Medicine Unit-2, Rawalpindi Medical University, Rawalpindi. Informed consent was obtained from all participants, and they were provided the option to withdraw from the study at any time without repercussions.
Consent for Publication
Not applicable, as the manuscript does not involve identifiable personal data.
Availability of Data and Materials
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Competing Interests
The authors declare that they have no competing interests.
Funding
No specific funding was received for this study.
Authors' Contributions
Dr. Muhammad Shaheryar Bashir conceptualized and designed the study, coordinated the audit process, and drafted the manuscript.
Shahana Ghazal contributed to data collection and analysis.
Muhammad Talha Mahmood, Abdullah Khalil, and Aiman Nazir participated in the audit implementation and data entry.
Muhammad Khurram supervised the study, provided critical revisions, and ensured academic integrity. All authors reviewed and approved the final manuscript.
Acknowledgements
The authors would like to thank the residents and staff of the Medical and Nephrology Units of the Tertiary Care Hospital, Rawalpindi, for their cooperation and participation in this quality improvement audit. Special thanks to the Ethical Review Board for their guidance and approval.
Availability of Data and Materials
The datasets generated and/or analyzed during the current study are not publicly available due to ethical constraints and patient confidentiality concerns. However, they are available from the corresponding author upon reasonable request.
References
- Panepinto R, Harris J, Wellette J. (2021). A review of best practices related to intravenous line management for nurses. Nursing Clinics, 56(3):389-399.
Publisher | Google Scholor - Timsit JF, Baleine J, Bernard L, Calvino-Gunther S, Darmon M, Dellamonica J, Desruennes E, Leone M, Lepape A, Leroy O, Lucet JC. (2020). Expert consensus-based clinical practice guidelines management of intravascular catheters in the intensive care unit. Annals of Intensive Care, 10:1-26
Publisher | Google Scholor - Steele R, Irvin CB. (2001). Central line mechanical complication rate in emergency medicine patients. Academic Emergency Medicine, 8(2):204-207.
Publisher | Google Scholor - Almahmoud RS, Alfarhan MA, Alanazi WM, Alhamidy FK, Balkhy HH, et al. (2020). Assessment knowledge and practices of central line insertion and maintenance in adult intensive care units at a tertiary care hospital in Saudi Arabia. Journal of Infection and Public Health, 13(11):1694-1698.
Publisher | Google Scholor - Latif A, Halim MS, Pronovost PJ. (2015 ). Eliminating infections in the ICU: CLABSI. Current Infectious Disease Reports, 17:1-9.
Publisher | Google Scholor - Marschall J, Mermel LA, Classen D, Arias KM, Podgorny K, et al. (2008). Strategies to prevent central line–associated bloodstream infections in acute care hospitals. Infection Control & Hospital Epidemiology, 29(S1):S22-30.
Publisher | Google Scholor - Apisarnthanarak A, Ratz D, Greene MT, Khawcharoenporn T, Weber DJ, et al. (2017). National survey of practices to prevent health care-associated infections in Thailand: the role of prevention bundles. American journal of infection control, 45(7):805-810.
Publisher | Google Scholor - Hopkins Medicine. Central Line Insertion Checklist. Baltimore: Johns Hopkins Medicine.
Publisher | Google Scholor - Apfelbaum JL, Rupp SM, Tung A, Connis RT, Domino KB, et al. (2020). Practice guidelines for central venous access 2020 an updated report by the American Society of Anesthesiologists Task Force on central venous access. Anesthesiology, 132(1):8-43.
Publisher | Google Scholor - Hussain AS, Ahmed AM, Arbab S, Ariff S, Ali R, et al. (2021). CLABSI reduction using evidence based interventions and nurse empowerment: a quality improvement initiative from a tertiary care NICU in Pakistan. Archives of Disease in Childhood, 106(4):394-400.
Publisher | Google Scholor