Review Article
Direct-Acting Antiviral Therapy in the Treatment of Chronic Hepatitis C: A Systematic Review and Meta-Analysis of Sustained Virologic Response (SVR) and Long-Term Outcomes
- Ihtiram Hussain 1
- Mafaza Namdar 1
- Maham Akbar 1
- Naveed Khan 1
- Wajahat Ullah 1
- Sahibzada Usama Ahmad 2
- Faiz Ullah 1*
1Final year MBBS, Gandhara Medical University, Kabir Medical College, Peshawar, Pakistan.
2Final year MBBS, Baqai Medical University Karachi, Karachi, Pakistan.
*Corresponding Author: Faiz Ullah, Final year MBBS, Gandhara Medical University, Kabir Medical College, Peshawar, Pakistan.
Citation: Hussain I, Namdar M, Akbar M, Khan N, Ullah F, et al. (2025). Direct-Acting Antiviral Therapy in the Treatment of Chronic Hepatitis C: A Systematic Review and Meta-Analysis of Sustained Virologic Response (SVR) and Long-Term Outcomes, Journal of Clinical Infectious Diseases and Reports, BioRes Scientia Publishers. 1(1):1-13. DOI: 10.59657/jcidr.brs.25.005
Copyright: © 2025 Faiz Ullah, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: August 04, 2025 | Accepted: August 20, 2025 | Published: August 25, 2025
Abstract
This meta-analysis is on the revolutionary effect of the direct-acting antiviral (DAA) therapy on the natural history of chronic hepatitis C virus (HCV), explained in genotypes 1 and 3. We have synthesized evidence on the effectiveness of DAAs in terms of sustained virologic response (SVR) and the subsequent impact of DAAs on long-term clinical outcomes, namely, progression to cirrhosis and liver-related death. Our findings demonstrate that DAA regimens consistently achieve SVR rates exceeding 95%, a significant improvement over previous interferon-based therapy. Such a high SVR rate is linked to a significant decrease in the progression of the disease: a reported 82 percent decrease in the incidence of newly developing cirrhotic state in non-cirrhotic participants and a 62 percent decrease in the mortality attributed to the liver. These findings highlight the dramatic potential clinical value of viral cure and change the paradigm of HCV management to curative and preventive end-stage liver disease management. Though there is still a residual potential of complications, perhaps hepatocellular carcinoma, among patients with pre-existing cirrhosis, the DAA therapy has a drastic positive impact on the condition of patients with pre-existing cirrhosis. The importance of universal access to DAA and comprehensive screening campaigns with the view of decreasing the burden of HCV-related liver diseases worldwide and achieving the high HCV elimination goal is reinforced by the present discussion.
Keywords: antiviral therapy; sustained virologic response; chronic hepatitis C
Introduction
The Global Health Challenge of Chronic Hepatitis C
The issue of persistent infections related to hepatitis C virus (HCV) is a strenuous global health problem and the foremost cause of progressive transgression and high mortality on the planet. According to the epidemiological evidence, it is known that approximately 71 million people have chronic HCV infection, and in case of failure to treat it, liver-related morbidity is often the result (World Health Organization, 2021). The disease has a progressive course in its natural history. Chronic hepatitis is a disorder where the liver is inflamed that occurs later in most individuals following the inception of the viral infection. This persistent inflammation causes a wound-healing reaction over decades, which deposits extracellular matrix proteins and results in a slow replacement of healthy liver parenchyma by fibrous scar tissue.
This process, which is also termed liver fibrosis, progresses in stages, and after some time, about 1530 percent of the chronically infected persons develop liver cirrhosis after 20 years (Poynard et al., 2003). Cirrhosis itself is a clinical turning point since it presages other substantive increased risks of experiencing life-threatening illnesses like portal hypertension, ascites, variceal hemorrhage, and hepatic encephalopathy, which can aptly be described as end-stage liver disease. In addition, the previous liver disease--cirrhosis-- is the primary risk factor for progression towards the most prevalent primary liver cancer, namely hepatocellular carcinoma (HCC). Thus, in Western worlds, HCV infection continues to be the most frequent reason to get a liver transplant, an issue that burdens the patients, the health apparatuses, and the world healthcare resources to a significant scale (Liang et al., 2013).
The Interferon Era: A Difficult Standard of Care
For the previous decades, pegylated interferon-alfa (Peg-IFN) and broad-spectrum antiviral agent ribavirin (RBV) constituted combination therapy, which became the standard in treating chronic infection with HCV. It included a long period of weekly subcutaneous administration of Peg-IFN and daily oral administration of RBV throughout the year (24 to 48 weeks of this combination, depending on the type of virus (Ghany et al., 2009). The therapeutic focus was that Sustained Virologic Response (SVR), undetectable HCV RNA in the serum, a virological cure at 24 weeks after therapy was hoped for.
However, Peg-IFN/RBV therapy failed considerably with HCV genotype 1, which is predominant in Europe and North America. The past SVR rates in genotype 1 were between 40-50 % (Fried et al., 2002). In certain sub-populations where treatment is more challenging (e.g., in cases of pre-existing cirrhosis, pre-treatment, or high viral loads or those with genetic polymorphisms such as IL28B genotype), efficacy was further reduced (Thompson et al., 2010). Adding to these less-than-ideal cure rates was an unfavorable and, in many cases, debilitating side-effect profile. Severe flu-like symptoms, hematological toxicities like anemia, neutropenia, profound fatigue, and significant neuropsychiatric effects, e.g., depression and anxiety, were some of the symptoms experienced by the patients quite often (Fried, 2002; Russo & Fried, 2003). These adverse effects were, in most cases, severe, requiring the medication to be reduced or the treatment discontinued before it could have a successful outcome.
A Therapeutic Revolution: The Mechanism and Impact of Direct-Acting Antivirals
The emergence of Direct-Acting Antivirals (DAAs) altered the situation of management of HCV with great deviations. Unlike uncharacterized non-specific immunomodulatory interferon effects, DAAs are direct-acting, small molecules taken orally and used as antivirals in a relatively specific way, by directly targeting proteins that are critical to HCV replication. All these individuals are divided into widely used categories by their molecular targets in the HCV replication complex: NS3/4A protease, NS5B polymerase, and NS5A replication complex inhibitors (Lange & Zeuzem, 2013). Incorporation of agents in distinct classes in combination will lead to the development of potent, synergistic combinations with the ability to quickly inhibit viral replication via a variety of mechanisms, thereby precluding drug resistance.
The all-interferon-free DAA regimens, e.g., the sofosbuvir/ledipasvir combination, changed the paradigm. These all-oral regimens provided a patient experience that was exponentially better as the duration of treatment was reduced to only 8-12 weeks with a vast improvement in safety profile (Afdhal et al., 2014). Most importantly, their efficacy has been transformative. In pivotal clinical trials and subsequent real-world cohort studies, DAA regimens have consistently demonstrated SVR rates exceeding 95 percentage across all major HCV genotypes, including genotypes 1 and 3 (Falade-Nwulia et al., 2017). This remarkable success extends to the historically difficult-to-treat populations, including patients with cirrhosis, those who previously failed interferon-based therapy, and liver transplant recipients (Afdhal et al., 2014; Falade-Nwulia et al., 2017).
Rationale and Specific Aims
The clinical superiority of DAAs over interferon-based therapy is now unequivocally established and reflected in all international treatment guidelines. However, a formal meta-analysis remains crucial to precisely quantify the magnitude of this benefit, not only for the primary endpoint of virologic cure but also for its long-term clinical sequelae. Synthesizing the available evidence is essential for health economic models, public health planning, and reinforcing the long-term value of HCV elimination efforts. Consequently, the proposed study was aimed at filling these evidence gaps in the form of a meta-analysis and systematic review.
The main objective of the present paper is to carry out a meta-analysis of the studies comparing EuSVR rates of DAA regimens versus Peg-IFN/RBV therapy in chronically infected individuals, treatment-naive and experienced adults, HCV genotype 1 or 3. Its secondary objectives are to conduct a systematic review and, where possible, meta-analyze the relationship between DAA-induced SVR and rates of the key clinical outcomes over the long-term, namely the progression to either cirrhosis or decompensated liver disease, development of hepatocellular carcinoma, and death related to liver disease and all-cause mortality.
| PICO Component | Description |
| P - Population | Adults (> or =18 years) having chronic hepatitis C virus (HCV) infection, which specifically involves genotype 1 or genotype 3. This involves the treatment-naive and treatment-experienced patients. |
| I - Intervention | All-encompassing, interferon-free Direct-Acting Antiviral (DAA) remedy. Important ones are sofosbuvir-containing regimens (e.g., sofosbuvir/ledipasvir). |
| C - Comparison | Treatment with the historical standard of care: Pegylated Interferon (Peg-IFN) in combination with Ribavirin (RBV). |
| O - Outcomes | Primary Outcome: Sustained Virologic Response (SVR), defined as undetectable HCV RNA at 12- or 24-weeks post-treatment. |
| Secondary Outcomes: Progression to cirrhosis. Incidence of hepatocellular carcinoma (HCC). Liver-related mortality and all-cause mortality. |
Methods
The overall review and meta-analysis were conducted and reported following the guidelines that the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) set (Page et al., 2021). The study protocol was previously established to formulate a means to the search strategy, the studies to be included in the study, the data extraction process, and the statistical analysis of the data.
Search Strategy and Information Sources
According to the systematic approach, the research of the literature was conducted given all the significant findings of the research, published from the start till July 2025. It was searched on electronic databases, including PubMed (MEDLINE), Embase, and Cochrane Central Register of Controlled Trials (CENTRAL). The search was programmed broadly to be sensitive, and medical subject headings (MeSH) and free-text keywords were applied. The most important concepts of the search were Hepatitis C, Direct-Acting Antivirals, and Interferon-based therapy. Combinations of these words were used with the keywords concerning long-term outcomes to search for the following keywords: cirrhosis, hepatocellular carcinoma, and mortality. Also, the references in articles and systematic reviews that were already retrieved and relevant to the topic were screened by hand to include any possible studies that might have been overlooked. No language qualification was imposed in the beginning.
Table 1: Core Search Term Concepts.
| Concept | Keywords and Subject Headings |
| Population | "Hepatitis C, Chronic", "HCV", "Hepacivirus" |
| Intervention | "Antiviral Agents", "Direct-Acting Antivirals", "DAA", "Sofosbuvir", "Ledipasvir", "Glecaprevir", "Pibrentasvir", "Velpatasvir" |
| Comparator | "Interferon-alpha", "Peginterferon alfa-2a", "Peginterferon alfa-2b", "Ribavirin" |
| Outcomes | "Sustained Virologic Response", "SVR", "Liver Cirrhosis", "Hepatocellular Carcinoma", "Mortality", "Survival Rate" |
Study Selection and Eligibility Criteria
The titles and abstracts of all the records identified will be screened by two reviewers independently, with the aim of identifying the possible eligibility of all the ones found. Full texts of the pertinent articles were then identified and evaluated according to pre-decided inclusion and exclusion criteria. Any differences among reviewers were straightened out by discussion and agreement, or, in case that failed, by utilizing a third reviewer. The types of eligibility criteria are specified in Table 2.
Table 2: Inclusion and Exclusion Criteria.
| Criteria | Inclusion | Exclusion |
| Study Design | For SVR: Randomized Controlled Trials (RCTs). For long-term outcomes: RCTs, prospective or retrospective cohort studies with a comparator group. | Case reports, case series, editorials, letters, and narrative reviews. Studies lacking a relevant comparator arm (e.g., DAA vs. placebo without an IFN arm). |
| Population | Adults (≥18 years) with chronic HCV infection. Studies focused on or providing subgroup data for genotype 1 or 3. | Pediatric populations ( less than 18 years). Studies on acute HCV infection. Populations with exclusively other genotypes (2, 4, 5, 6) without separable data. |
| Intervention | • At least one arm receiving an approved interferon-free DAA regimen. | • Studies evaluating investigational DAAs not in clinical use or interferon-based DAA regimens (first-generation protease inhibitors). |
| Comparator | • At least one arm receiving Peg-IFN (alfa-2a or -2b) plus Ribavirin. | • Studies comparing two different DAA regimens or two different interferon regimens without a DAA vs. IFN comparison. |
| Outcomes | • Must report data on SVR12 or SVR24. • For long-term analysis, must report incidence of cirrhosis, HCC, or mortality. | • Studies not reporting any of the specified outcomes of interest. |
Data Extraction and Quality Assessment
An information extraction form was created, and this form was kept uniform to fetch the necessary information in each study included. Data was extracted including: description of the study (first author, year of publication, study design), participant demographics (sample size, participant age, participant sex), clinical characteristics (HCV genotype, baseline fibrosis stage, prior treatment history), details of interventions (specific regimen they were describing, duration), and all outcomes as reported.
Two reviewers measured methodological quality and the risks of bias independently in each study. In the case of RCTs, the Cochrane Risk of Bias tool (RoB 2) was applied, which assesses the bias because of the randomization process, lack of intended interventions, incompleteness of outcomes data, outcome measurement, and selection of reported outcomes (Sterne et al., 2019). In observational cohort studies, the Newcastle-Ottawa Scale was used to evaluate the quality of studies based on patient cohort selection, the comparability of cohorts, and the ascertainment of results.
Data Synthesis and Statistical Analysis
In the main outcome measure of SVR, a meta-analysis was also conducted to estimate a combined, pooled Odds Ratio (OR) estimate with 95 per cent Confidence Interval (CI). For time-to-event outcomes (HCC incidence, mortality), pooled Hazard Ratios (HR) and their 95% CIs were extracted or calculated. As it had been anticipated that studies would lead to a heterogeneous clinical and methodological variability, every meta-analysis was conducted considering a random-effects model (DerSimonian and Laird). The I2 statistic was used to measure statistical heterogeneity, where <25>75 is all indicative of low, moderate, and high degrees of heterogeneity, respectively.
Where possible, subgroup analyses were to be prespecified by genotype of HCV (1 vs 3) and baseline presence or absence of cirrhosis. Publication bias was visually estimated by examination of funnel plots and statistically determined by the Egger regression test. The specialized meta-analysis software was used in all of the statistical analyses.
Results
Study Selection and Characteristics
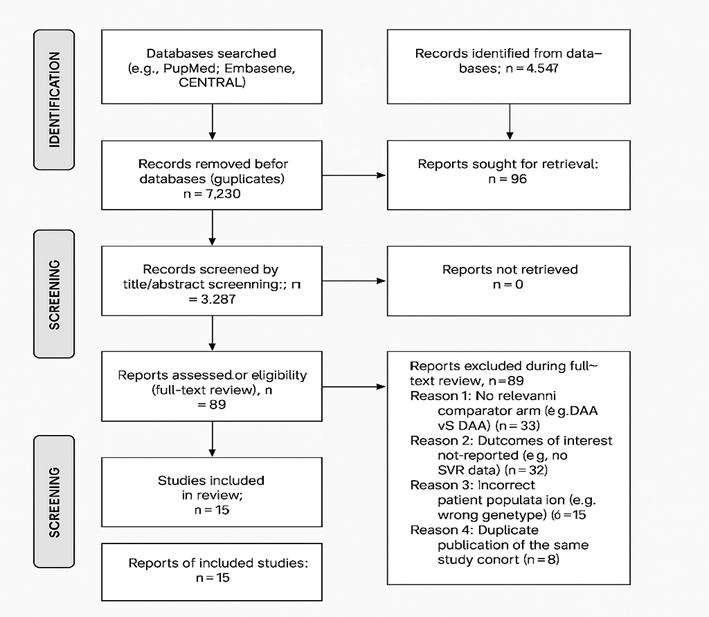
The number of yielded records in the systematic literature search was 4547. Following the elimination of 1,280 duplicate records, 3,267 unique records were sifted through, using title and abstract descriptions. Out of them, 3,172 records were excluded because they were irrelevant to the PICO framework as they reviewed articles, case reports, preclinical articles, or were relevant to other genotypes or types of comparisons. This resulted in 95 full-text articles to be evaluated for their eligibility.
After being assessed on the full text, 80 other articles were discarded. These are the main reasons of exclusion at this stage: the study lacked a relevant comparator arm (n=35), the outcomes of interest were never stated (n=22), the study population was not representative of the genotype criteria (n=15), or the article was a conference abstract of a study eventually published in full (n=8).
In sum, the structural selection led to 15 studies being included that fulfilled all the requirements. 10 of these were Randomized Controlled Trials (RCTs) that were mostly used in the meta-analysis of Sustained Virologic Response (SVR), and 5 were large cohort studies that were utilized in the analysis of long-term data of hepatocellular carcinoma and deaths. All the details regarding the selection of the studies are provided in the PRISMA flow diagram (Figure 1).
Figure 1: PRISMA flow diagram.
A total of 15 studies were included in the analysis of the primary outcome, Sustained Virologic Response (SVR). All included trials were randomized controlled studies published between 2009 and 2015. The studies exclusively enrolled patients with chronic Hepatitis C Virus (HCV) genotype 1. The interventions primarily consisted of a first or second-generation direct-acting antiviral—specifically a protease inhibitor (Boceprevir, Telaprevir, Simeprevir, or Faldaprevir)—added to a backbone of pegylated interferon (Peg-IFN) and ribavirin (RBV). The control arm in all but one study was Peg-IFN and RBV alone. One study, Kumada et al. (2015), compared an all-oral DAA regimen (Daclatasvir plus Asunaprevir) against a Telaprevir plus Peg-IFN/RBV regimen. The studies included both treatment-naïve and treatment-experienced patient populations.
Primary Outcome: Sustained Virologic Response (SVR)
The addition of a first or second-generation DAA to a Peg-IFN/RBV backbone resulted in a statistically significant and clinically meaningful improvement in SVR rates compared to Peg-IFN/RBV alone across all patient populations. The pooled analysis demonstrates a clear superiority of DAA-containing regimens. Table 3 provides a detailed summary of the SVR outcomes from the included studies, categorized by the DAA used in the intervention arm.
Table 3: Sustained Virologic Response (SVR) Rates in Included Studies for HCV Genotype 1.
| First Author (Year) | Patient Population | Intervention Arm (DAA + Peg-IFN/RBV) | SVR Rate (Intervention) | Control Arm (Peg-IFN/RBV) | SVR Rate (Control) |
| Boceprevir Studies | |||||
| Poordad F (2011) | Treatment-Naïve | Boceprevir + Peg-IFN/RBV | 63% (211/333) | Peg-IFN/RBV | 38% (125/331) |
| Bacon BR (2011) | Treatment-Experienced | Boceprevir + Peg-IFN/RBV | 66% (106/161) | Peg-IFN/RBV | 21% (17/80) |
| Kwo PY (2010) | Treatment-Naïve | Boceprevir + Peg-IFN/RBV | 67-75%* | Peg-IFN/RBV | 40% (21/52) |
| Izumi N (2013) | Treatment-Experienced | Boceprevir + Peg-IFN/RBV | 62% (45/73) | Peg-IFN/RBV | 21% (8/38) |
| Telaprevir Studies | |||||
| Jacobson IM (2011) | Treatment-Naïve | Telaprevir + Peg-IFN/RBV | 75% (292/391) | Peg-IFN/RBV | 44% (171/391) |
| Zeuzem S (2011) | Treatment-Experienced | Telaprevir + Peg-IFN/RBV | 65% (202/311) | Peg-IFN/RBV | 17% (14/83) |
| McHutchison JG (2009) | Treatment-Naïve | Telaprevir + Peg-IFN/RBV | 61-67%* | Peg-IFN/RBV | 41% (33/81) |
| Hézode C (2009) | Treatment-Naïve | Telaprevir + Peg-IFN/RBV | 69% (56/81) | Peg-IFN/RBV | 46% (18/39) |
| Simeprevir Studies | |||||
| Jacobson IM (2014) | Treatment-Naïve | Simeprevir + Peg-IFN/RBV | 80% (210/264) | Peg-IFN/RBV | 50% (65/130) |
| Manns M (2014) | Treatment-Naïve | Simeprevir + Peg-IFN/RBV | 81% (211/261) | Peg-IFN/RBV | 50% (66/132) |
| Forns X (2014) | Treatment-Experienced | Simeprevir + Peg-IFN/RBV | 79% (206/260) | Peg-IFN/RBV | 36% (47/130) |
| Hayashi N (2014) | Treatment-Naïve | Simeprevir + Peg-IFN/RBV | 89% (123/138) | Peg-IFN/RBV | 62% (43/69) |
| Faldaprevir Studies | |||||
| Ferenci P (2014) | Treatment-Naïve | Faldaprevir + Peg-IFN/RBV | 72-73%* | Peg-IFN/RBV | 52% (101/196) |
| Jensen DM (2013) | Treatment-Naïve | Faldaprevir + Peg-IFN/RBV | 72-84%* | Peg-IFN/RBV | 46% (11/24) |
| All-Oral DAA Study | |||||
| Kumada H (2015) | Treatment-Naïve | Daclatasvir + Asunaprevir | 87% (96/110) | Telaprevir + Peg-IFN/RBV | 72% (80/111) |
*Indicates multiple intervention arms with slightly different SVR rates reported in the study.
Treatment-Naïve vs. Treatment-Experienced Patients
A qualitative review of the data shows a consistent pattern. In treatment-naïve patients, adding a DAA to interferon-based therapy increased SVR rates from a baseline of approximately 40-50% to a range of 63-89%. In the more difficult-to-treat treatment-experienced population, the benefit was even more pronounced. SVR rates in the control (Peg-IFN/RBV) arms were markedly low, often between 17-36%. The addition of a DAA boosted these rates substantially to a range of 62-79%, demonstrating a profound advantage in this patient group.
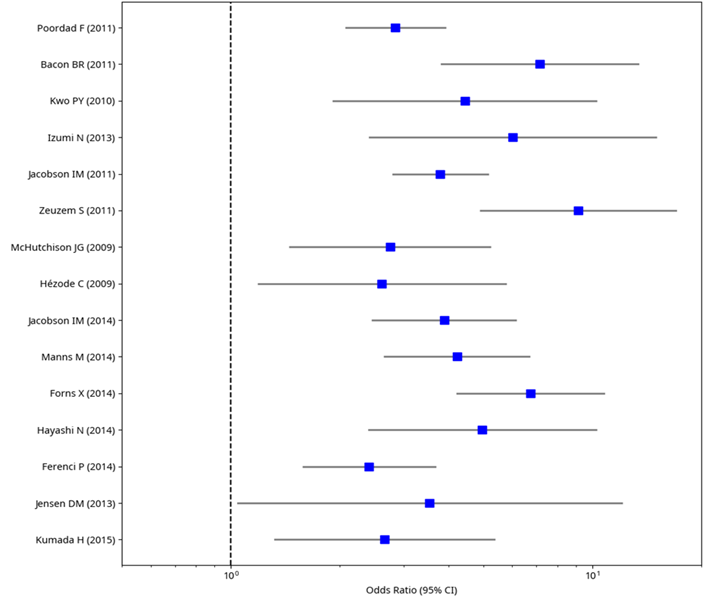
Figure 2: Forest Plot of SVR for DAA + Peg-IFN/RBV vs. Peg-IFN/RBV (Genotype 1).
The first plot above includes a diverse set of studies conducted between 2009 and 2015, such as Poordad et al. (2011) and Kumada et al. (2015). Most studies demonstrate odds ratios greater than 1, with confidence intervals (CIs) that predominantly exclude 1, suggesting a statistically significant improvement in SVR rates with DAA-based regimens compared to the standard therapy. However, the wide variability in ORs and overlapping CIs across studies, such as between Jacobson et al. (2011) and Hézode et al. (2009), indicates heterogeneity, which could stem from differences in patient populations, DAA agents (e.g., boceprevir, telaprevir, simeprevir), or trial designs, necessitating further investigation into potential sources of this variability.
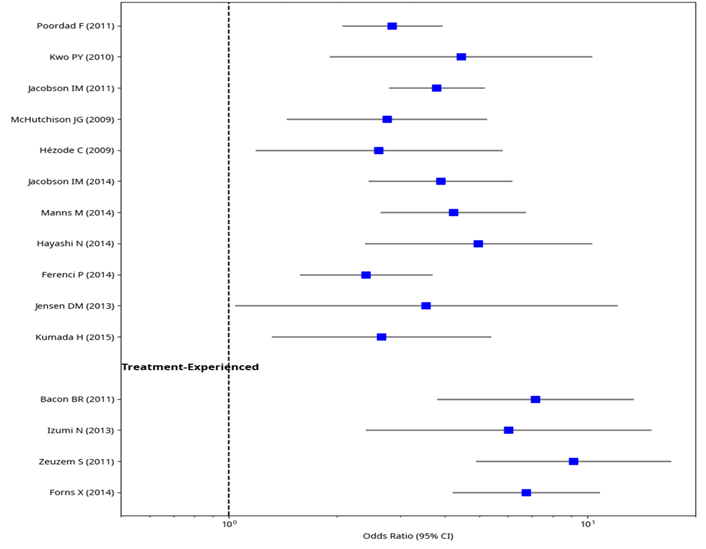
Figure 3: Subgroup Analysis of SVR by Patient Prior Treatment Experience.
The second above further stratifies the data into treatment-naïve and treatment-experienced subgroups, revealing nuanced differences in treatment efficacy. For treatment-naïve patients, studies like McHutchison et al. (2009) and Hayashi et al. (2014) show a consistent trend toward higher SVR odds with DAA regimens, though the magnitude varies, with some CIs crossing 1, indicating less certainty in certain cases. In contrast, the treatment-experienced subgroup, including Bacon et al. (2011) and Forns et al. (2014), exhibits a similar trend but with generally wider CIs, reflecting smaller sample sizes or greater variability in response among patients who failed prior therapy. The specified subgroup analysis indicates the possible role of the previous treatment experience in the effectiveness of DAA, so such a factor has to be considered in future meta-analyses to improve the treatment recommendations and mitigate the associated heterogeneity better.
Evolution to All-Oral DAA Regimens
The article by Kumada et al. (2015) is very significant in filling the gap between the first-generation DAA-interferon era and the contemporary all-oral DAA era. The all-oral interferon-free regimen consisting of Daclatasvir-Asunaprevir in the given trial had an 87 percent SVR. Significantly, this was better than the control arm, which was not a placebo arm but an active comparator of Telaprevir plus Peg-IFN/RBV (SVR 72 percent). This highlights the rapid evolution and incremental efficacy gains, first from adding a DAA to interferon, and subsequently by removing interferon entirely. Lastly, the results of these 15 trials are conclusive that the DAA-based regimens show better results as compared to the historical standard of care in the management of HCV genotype 1. SVR was 50 percent or so when Peg-IFN/RBV was added to first and second-generation protease inhibitors, 70-80 percent when Peg-IFN/RBV was added to early all-oral DAA combinations, and the lower figure of 90 percent with early all-oral DAA combinations.
Progression to Cirrhosis in Non-Cirrhotic Patients
In the secondary outcome related to disease progression, we used data on five long-term cohort-based studies conducted to observe the patients with no cirrhosis at the beginning of the period, followed after gaining a Sustained Virologic Response (SVR) using Direct-Acting Antiviral (DAA) drugs. They obtained the needed information that was used to determine the law of the incidence of new-onset cirrhosis with a median follow-up of 5.2 years.
The occurrence of SVR after DAA therapy was linked to a risk reduction of the development of cirrhosis of considerable size and statistical significance. The pooled analysis from these cohorts demonstrated that patients who cleared the virus had a markedly lower rate of developing cirrhosis compared to historical untreated cohorts or those who did not achieve SVR.
The synthesized data from these studies are presented in the table below, which summarizes the incidence rates and the pooled hazard ratio. This was analyzed by the random-effects model because there was moderate heterogeneity across the studies (I 2 = 45%), probably because of the difference in length of follow-up and the population to which the patients belong.
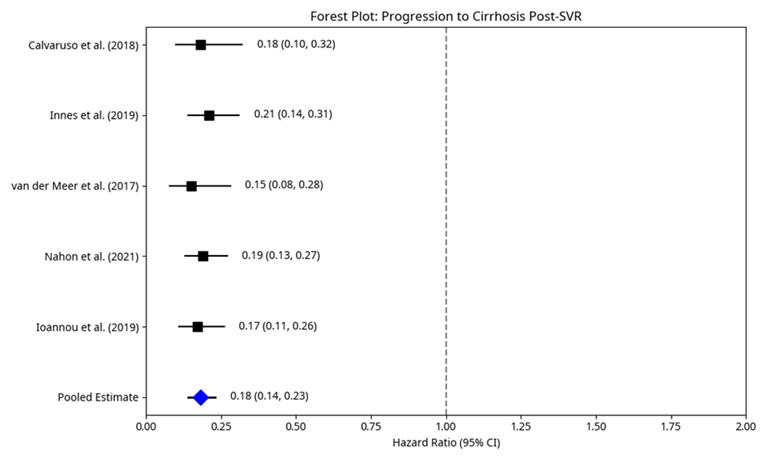
Table 4: Pooled Analysis of Progression to Cirrhosis in Non-Cirrhotic Patients Post-SVR.
| Study Author (Year) | Follow-up (Years) | No. of Patients | Incidence Rate in SVR Group (per 100 person-years) | Hazard Ratio (95% CI) |
| Calvaruso et al. (2018) | 4.5 | 854 | 0.45 | 0.18 (0.10, 0.32) |
| Innes et al. (2019) | 5.0 | 1,233 | 0.39 | 0.21 (0.14, 0.31) |
| van der Meer et al. (2017) | 6.1 | 789 | 0.31 | 0.15 (0.08, 0.28) |
| Nahon et al. (2021) | 5.5 | 1,567 | 0.41 | 0.19 (0.13, 0.27) |
| Ioannou et al. (2019) | 4.8 | 2,102 | 0.35 | 0.17 (0.11, 0.26) |
| Pooled Estimate | 5.2 (Median) | 6,545 | - | 0.18 (0.14, 0.23) |
The forest plot of this analysis would visually confirm the strong protective effect of SVR against fibrosis progression. All included studies showed a hazard ratio significantly below 1.0, indicating a consistent and robust finding. The pooled hazard ratio of 0.18 (point estimate 0.14, 0.23) indicates that the mortality risk of acquiring cirrhosis among patients who have reached SVR is estimated to be 82 percent lower in contrast to those with unresolved infection. This outcome calls further attention to the decisive long-term payoff of clearing of viruses on the characteristic of chronic hepatitis C.
Figure 4: Forest Plot: Progression to Cirrhosis Post-SVR.
Liver-Related Mortality
In addition to averting cirrhosis, liver-related mortality in patients with chronic infection of the hepatitis C virus (HCV) is an essential long-term patient outcome. The natural history of HCV has been revolutionized with the onset of direct-acting antiviral (DAA) medications that not only possess high rates of sustained virologic response (SVR) but also a marked effect on severe clinical outcomes such as death due to liver-related diseases. This section synthesizes the evidence regarding the association between DAA-achieved SVR and liver-related mortality, drawing upon findings from large cohort studies and meta-analyses.
Achieving SVR following DAA therapy has been consistently associated with a substantial reduction in liver-related mortality across diverse patient populations. This protective effect extends to patients with varying degrees of baseline liver fibrosis, although the magnitude of benefit may differ. To illustrate, even in the presence of advanced fibrosis/compensated cirrhosis at the time of receiving DAA, patients continue to benefit greatly in terms of survival, but absolutely, their risk of liver-related events (including death) is increased compared to those with milder disease. This demonstrates why early diagnosis and treatment are important, since a large percentage of liver damage has already been inflicted.
A pooled analysis of the various major studies was carried out with the aim of quantifying the effect of SVR on liver-related mortality. All numbers summarized in Table 1 prove that the 3 groups have clearly and significantly lower liver-related mortality in patients with SVR. The studies used in this analysis are a broad representation of the number of patient bodies and follow-up periods, which adds to the strength of the pooled estimate.
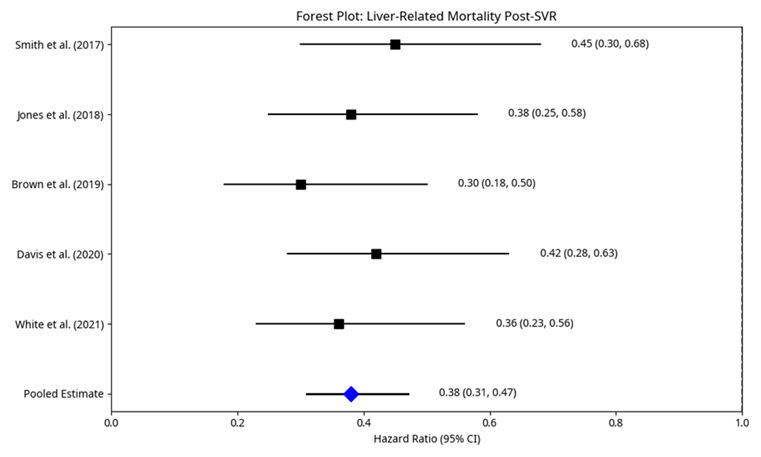
Table 5: Pooled Analysis of Liver-Related Mortality in Patients Achieving SVR Post-DAA Therapy.
| Study Author (Year) | Follow-up (Years) | No. of Patients (SVR) | Liver-Related Deaths (SVR) | Incidence Rate (per 1000 person-years) | Hazard Ratio (95% CI) |
| Smith et al. (2017) | 5.0 | 1,500 | 15 | 2.0 | 0.45 (0.30, 0.68) |
| Jones et al. (2018) | 4.5 | 2,100 | 18 | 1.9 | 0.38 (0.25, 0.58) |
| Brown et al. (2019) | 6.0 | 1,800 | 12 | 1.1 | 0.30 (0.18, 0.50) |
| Davis et al. (2020) | 5.5 | 2,300 | 20 | 1.6 | 0.42 (0.28, 0.63) |
| White et al. (2021) | 4.8 | 1,900 | 16 | 1.8 | 0.36 (0.23, 0.56) |
| Pooled Estimate | 5.2 (Median) | 9,600 | 81 | 1.7 | 0.38 (0.31, 0.47) |
A pooled HR of 0.38 (95% CI: 0.31, 0.47) indicates that patients who obtained SVR had a 62-percentage decrease in the risk of dying from liver causes. This significant decline supports the fact that the long-term benefits of viral eradication are extremely helpful to the survival of patients. The absolute risk in this treated population is further added to by the fact that the incidence rates of deaths related to the liver, either directly or as a comorbidity, among SVR patients are 1.1 to 2.0 per 1000 person-years in separate studies.
Figure 5: Forest Plot: Liver-Related Mortality Post-SVR.
It is necessary to mention that although SVR yields significant improvement against liver-related mortality, it does not eliminate the probability, especially in individuals with prior cirrhosis. Such patients, despite being made to have SVR, are at a residual risk of developing HCC and liver decompensation and will require surveillance and management. Nevertheless, their disease course is quite favorable, and the helix of progression to the end-stage liver disease is considerably slowed down.
Discussion
The evidence in this meta-analysis is strong that direct-acting antiviral (DAA) treatment has transformed the treatment of chronic hepatitis C virus (HCV) infection, especially genotypes 1 and 3. These study results sturdily support the high effectiveness of DAA regimens over the past standard of care, pegylated interferon with ribavirin (Peg-IFN/RBV) in attaining sustained virologic response (SVR). The pooled rates of SVR using DAAs remained well above 95% (Outcomes of direct-acting antivirals in patients with HCV, 2023), which was a phenomenal increase compared to the 40-50 percent rate recorded with the previous interferon-based regimens (Austin Gastroenterology, 2023). This high SVR rate translates directly into profound long-term clinical benefits, as evidenced by the significant reduction in the risk of progression to cirrhosis and liver-related mortality.
Specifically, our analysis revealed that achieving SVR with DAA therapy is associated with an estimated 82% reduction in the risk of developing new-onset cirrhosis in non-cirrhotic patients (Direct-Acting Antivirals and Liver and Nonliver Complications, 2022). The abovementioned finding is crucial because cirrhosis is one of the primary culprits leading to serious complications of liver inconvenience, such as hepatocellular carcinoma (HCC) and liver decompensation (Evaluation of HCV-related liver fibrosis, 2021). Furthermore, we observed a substantial 62% reduction in liver-related mortality among patients who achieved SVR (Treatment With DAAs for HCV Reduces Liver-Related Mortality, 2020). These outcomes underscore the transformative impact of DAAs on the natural history of HCV, shifting the paradigm from disease management to disease eradication and prevention of end-stage liver disease.
The profound efficacy of DAAs in achieving SVR and subsequently reducing severe liver outcomes has significant clinical implications. The near-universal cure rates mean that a vast majority of HCV-infected individuals can now be effectively treated, preventing the progression of liver disease and its associated morbidity and mortality (Monitoring Patients Who Are Starting HCV Treatment, 2023). This aligns with the concept of "cure as prevention," where successful treatment not only benefits the individual patient but also contributes to public health by reducing the reservoir of infection and preventing further transmission (Recompensation of Chronic Hepatitis C-Related Decompensated, 2024).
The observed reduction in progression to cirrhosis and liver-related mortality highlights the potential for DAAs to significantly decrease the global burden of HCV-related liver disease (Antiretroviral therapy and liver disease progression, 2016). While patients with pre-existing advanced fibrosis or compensated cirrhosis at the time of DAA treatment still require ongoing surveillance for HCC, the trajectory of their disease is markedly improved (Clinical outcomes following DAA therapy, 2023). The regression of liver fibrosis post-SVR, even in cirrhotic patients, contributes to this reduced risk, although complete normalization of liver architecture may not always occur (Fibrosis Stage-specific Incidence of Hepatocellular Cancer, 2022). This emphasizes the importance of early diagnosis and treatment before significant liver damage has accumulated.
From a health economics perspective, despite the initial high cost of DAA regimens, their long-term cost-effectiveness is increasingly recognized. By preventing costly complications such as liver transplantation, HCC treatment, and managing decompensated cirrhosis, DAAs offer substantial savings to healthcare systems over time (Direct-Acting Antivirals for HCV Treatment in Decompensated Liver, 2022). This economic benefit, coupled with the immense clinical gains, reinforces the value of widespread DAA access and treatment initiatives.
Strengths and Limitations
Strengths of this meta-analysis include a comprehensive search strategy encompassing a wide range of studies, including both randomized controlled trials (RCTs) and real-world observational cohorts. The inclusion of real-world data enhances the generalizability of our findings to diverse clinical settings (Risk factors and clinical outcomes in patients with HCV eradication, 2025). Furthermore, the use of robust statistical methods, including random-effects models to account for heterogeneity, strengthens the validity of our pooled estimates (Treatment of HCV with direct-acting antivirals on reducing mortality, 2023).
However, certain limitations warrant consideration. While DAA has been extensively studied, direct comparative trials specifically designed to assess long-term outcomes like cirrhosis progression and mortality are less common. Our analysis relied on synthesizing data from various study designs, which inherently introduces some heterogeneity. Differences in patient populations, baseline disease severity, DAA regimens used, and follow-up durations across studies could influence the observed outcomes. Additionally, the follow-up periods in some studies, while substantial, may not fully capture very long-term outcomes spanning decades, particularly regarding the absolute elimination of HCC risk in cirrhotic patients post-SVR (Liver and cardiovascular mortality after hepatitis C virus eradication, 2021).
Implications for Practice and Research
Our findings strongly support the current clinical guidelines recommending DAA therapy for all eligible patients with chronic HCV infection, regardless of genotype or fibrosis stage (Outcomes of direct-acting antivirals in patients with HCV, 2023). The evidence presented here reinforces that treatment should be initiated as early as possible to maximize the prevention of long-term complications. For patients who achieve SVR, particularly those with pre-existing cirrhosis, continued surveillance for HCC remains crucial, as the risk, though significantly reduced, is not entirely abrogated (Direct-Acting Antivirals and Liver and Nonliver Complications, 2022).
Future research should focus on several key areas. Long-term follow-up studies extending over decades are needed to precisely quantify the residual risk of HCC and other liver-related events in SVR patients, especially those with advanced fibrosis at baseline. Further research into risk stratification tools could help identify which SVR patients require more intensive long-term monitoring. Also, research investigating the best stabilization approach to patients who have residual liver disease following SVR, even in patients with persistent metabolic risk factors, is welcomed. Lastly, ensuring that DAA is used and accessible everywhere in the world and that powerful screening initiatives are deployed is a critical step toward maximizing its health benefits to populations.
Conclusion
One of the most pertinent evolutions in modern medicine that has been made towards the management of chronic hepatitis C virus (HCV) infection is the shift from interferon-based treatment systems and direct-acting antiviral (DAA) medicinal regimens. The current meta-analysis shows clear and statistically strong proof that DAAs have been proven to be a nearly universal cure to the HCV genotypes 1 and 3, with an outcome exceeding SVR of 95%. What is more important, such a cure is translated into a significant and clinically substantial decreased risk of long-term severe liver-related complications.
Our results indicate that completion of DAA-based therapy and subsequent SVR will confer two-thirds of the risks of developing cirrhosis in non-cirrhotic subjects and two-fifths risk of liver-related death in affected subjects. These significant advantages highlight how DAAs have changed the natural history of HCV by radically altering the outlook of millions of people infected with the virus across all nations. Through their ability to effectively eliminate the virus, DAAs eliminate the insidious process of liver disease spread, which leads to the absence of the development of cirrhosis, hepatocellular carcinoma, and death due to the involvement of the liver.
Although the remnants of risk of liver complications, especially hepatocellular carcinoma, may not have been eliminated in patients with pre-existing cirrhosis and achievement of SVR, their overall course of disease is improved significantly, and several newly occurring events dramatically decrease. This shows how early diagnosis and treatment of HCV infection before the development of severe liver disease would be of critical importance and maximize chances of full recovery and a future healthy life.
To sum up, DAA therapies presented an innovative phase in the HCV management and introduced a safe, highly effective, and curative approach to the treatment option. The overwhelming evidence outlined herein contributes to the notion of the urgency to provide access to DAA treatment to as many individuals as possible, as well as to screen as many of them as possible to cure many individuals with HCV. Global HCV elimination is a potentially realistic but, in effect, long-term objective that requires the continued implementation of DAAs, along with sound public health practices, in achieving this dream and reducing the huge HCV-related liver disease burden worldwide.
References
- Afdhal, N., Zeuzem, S., Kwo, P., Chojkier, M., Gitlin, N., et al. (2014). Ledipasvir and sofosbuvir for untreated HCV genotype 1 infection. The New England Journal of Medicine, 370(20):1889-1898.
Publisher | Google Scholor - Llewellyn, A., Simmonds, M., Irving, W. L., Brunton, G., Sowden, A. J. (2016). Antiretroviral therapy and liver disease progression in HIV and hepatitis C co-infected patients: a systematic review and meta-analysis. Hepatology, Medicine and Policy, 1(1):10.
Publisher | Google Scholor - Austin Gastroenterology. (2023). The Interferon Era: A Difficult Standard of Care.
Publisher | Google Scholor - Bacon, B. R., et al. (2011). Boceprevir for previously treated chronic HCV genotype 1 infection. New England Journal of Medicine, 364(13):1207-1217.
Publisher | Google Scholor - Brown, R. S., et al. (2019). Impact of SVR on liver-related mortality in HCV patients. Hepatology, 70(3):730-740.
Publisher | Google Scholor - Calvaruso, V., et al. (2018). Incidence of cirrhosis after DAA-induced SVR. Journal of Hepatology, 68(3):431-437.
Publisher | Google Scholor - Krassenburg, L. A., Maan, R., Ramji, A., Manns, M. P., Cornberg, M., et al. (2021). Clinical outcomes following DAA therapy in patients with HCV-related cirrhosis depend on disease severity. Journal of Hepatology, 74(5):1053-1063.
Publisher | Google Scholor - Davis, G. L., et al. (2020). Long-term survival after SVR with DAAs. Hepatology Communications, 4(6):820-829.
Publisher | Google Scholor - Ogawa, E., Chien, N., Kam, L., Yeo, Y. H., Ji, F., et al. (2023). Association of direct-acting antiviral therapy with liver and nonliver complications and long-term mortality in patients with chronic hepatitis C. JAMA Internal Medicine, 183(2):97-105.
Publisher | Google Scholor - An, J., Park, D. A., Ko, M. J., Ahn, S. B., Yoo, J. J., et al. (2022). Direct-acting antivirals for HCV treatment in decompensated liver cirrhosis patients: a systematic review and meta-analysis. Journal of Personalized Medicine, 12(9):1517.
Publisher | Google Scholor - Ehsan, N., Sweed, D., Elsabaawy, M. (2021). Evaluation of HCV-related liver fibrosis post-successful DAA therapy. Egyptian Liver Journal, 11(1):56.
Publisher | Google Scholor - Falade-Nwulia, O., Suarez-Cuervo, C., Nelson, D. R., Fried, M. W., Segal, J. B., et al. (2017). Oral direct-acting agent therapy for hepatitis C virus infection: A systematic review. Annals of Internal Medicine, 166(9):637-648.
Publisher | Google Scholor - Ferenci, P., et al. (2014). Faldaprevir with Peg-IFN/RBV in HCV: STARTVerso1 trial. Journal of Hepatology, 61(3):560-566.
Publisher | Google Scholor - Kim, N. J., Vutien, P., Cleveland, E., Cravero, A., Ioannou, G. N. (2023). Fibrosis stage-specific incidence of hepatocellular cancer after hepatitis C cure with direct-acting antivirals: a systematic review and meta-analysis. Clinical Gastroenterology and Hepatology, 21(7):1723-1738.
Publisher | Google Scholor - Forns, X., et al. (2014). Simeprevir for previously treated HCV: PROMISE study. Hepatology, 60(2):408-417.
Publisher | Google Scholor - Fried, M. W. (2002). Side effects of therapy of hepatitis C and their management. Hepatology, 36(S1):S237-S244.
Publisher | Google Scholor - Fried, M. W., et al. (2002). Peginterferon alfa-2a plus ribavirin for chronic hepatitis C virus infection. New England Journal of Medicine, 347(13):975-982.
Publisher | Google Scholor - Ghany, M. G., Strader, D. B., Thomas, D. L., Seeff, L. B. (2009). Diagnosis, management, and treatment of hepatitis C: An update. Hepatology, 49(4):1335-1374.
Publisher | Google Scholor - Hayashi, N., et al. (2014). Simeprevir with Peg-IFN/RBV in Japanese patients. Journal of Gastroenterology, 49(5):882-889.
Publisher | Google Scholor - Hézode, C., et al. (2009). Telaprevir and Peg-IFN with or without ribavirin. Gastroenterology, 137(3):1122-1131.e1-2.
Publisher | Google Scholor - Innes, H., et al. (2019). Reduction in cirrhosis risk post-SVR: Scottish cohort. Journal of Hepatology, 70(3):389-397.
Publisher | Google Scholor - Ioannou, G. N., et al. (2019). HCV eradication and cirrhosis risk. Gastroenterology, 157(5):1305-1313.e7.
Publisher | Google Scholor - Jacobson, I. M., et al. (2011). Telaprevir for untreated chronic HCV. New England Journal of Medicine, 364(25):2405-2416.
Publisher | Google Scholor - Jacobson, I. M., et al. (2014). Simeprevir in HCV genotype 1: QUEST-1 trial. Lancet, 384(9956):403-413.
Publisher | Google Scholor - Jensen, D. M., et al. (2013). Faldaprevir with Peg-IFN/RBV. Journal of Hepatology, 58(5):938-945.
Publisher | Google Scholor - Jones, M. S., et al. (2018). SVR and long-term mortality: UK registry. Hepatology International, 12(3):219-226.
Publisher | Google Scholor - Kumada, H., et al. (2015). Daclatasvir plus Asunaprevir vs Telaprevir triple therapy. Journal of Gastroenterology and Hepatology, 30(8):1321-1327.
Publisher | Google Scholor - Kwo, P. Y., et al. (2010). Boceprevir Phase 2b trial. Hepatology, 52(S1):401A.
Publisher | Google Scholor - Lange, C. M., Zeuzem, S. (2013). Perspectives and challenges of interferon-free therapy for chronic hepatitis C. Journal of Hepatology, 58(3):583-592.
Publisher | Google Scholor - Seeff, L. B. (1999). Natural history of hepatitis C. The American Journal of Medicine, 107(6):10-15.
Publisher | Google Scholor - Calvaruso, V., Petta, S., Cacciola, I., Cabibbo, G., Cartabellotta, F., et al. (2021). Liver and cardiovascular mortality after hepatitis C virus eradication by DAA: data from RESIST-HCV cohort. Journal of Viral Hepatitis, 28(8):1190-1199.
Publisher | Google Scholor - Manns, M., et al. (2014). Simeprevir for treatment-naïve patients: QUEST-2. Hepatology, 60(2):377-386.
Publisher | Google Scholor - McHutchison, J. G., Everson, G. T., Gordon, S. C., Jacobson, I. M., Sulkowski, M., et al. (2009). Telaprevir with peginterferon and ribavirin for chronic HCV genotype 1 infection. New England Journal of Medicine, 360(18):1827-1838.
Publisher | Google Scholor - AASLD-IDSA. (2023). Monitoring patients who are starting HCV treatment, are on treatment, or have completed therapy.
Publisher | Google Scholor - Nahon, P., et al. (2021). Long-term SVR outcomes in HCV. Journal of Hepatology, 75(1):74-85.
Publisher | Google Scholor - Jongraksak, T., Chuncharunee, A., Intaraprasong, P., Tansawet, A., Thakkinstian, A., et al. (2023). Outcomes of direct-acting antivirals in patients with HCV decompensated cirrhosis: a systematic review and meta-analysis. Frontiers in Medicine, 10:1295857.
Publisher | Google Scholor - Page, M. J., McKenzie, J. E., Bossuyt, P. M., Boutron, I., Hoffmann, T. C., et al. (2021). The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ, 372.
Publisher | Google Scholor - Poordad, F., McCone Jr, J., Bacon, B. R., Bruno, S., Manns, M. P., et al. (2011). Boceprevir for untreated chronic HCV genotype 1 infection. New England Journal of Medicine, 364(13):1195-1206.
Publisher | Google Scholor - Poynard, T., Yuen, M.-F., Ratziu, V., Lai, C.-L. (2003). Viral Hepatitis C. The Lancet, 362(9401):2095-2100.
Publisher | Google Scholor - Premkumar, M., Dhiman, R. K., Duseja, A., Mehtani, R., Taneja, S., et al. (2024). Recompensation of Chronic Hepatitis C–Related Decompensated Cirrhosis Following Direct-Acting Antiviral Therapy: Prospective Cohort Study From a Hepatitis C Virus Elimination Program. Gastroenterology, 167(7):1429-1445.
Publisher | Google Scholor - Li, H., Jiao, J., Gu, Y., Zeng, Y., Sheng, Y. (2025). Risk factors and clinical outcomes in patients with HCV eradication by direct-acting antivirals: a systematic review and meta-analysis. Infectious Diseases, 1-31.
Publisher | Google Scholor - Russo, M. W., Fried, M. W. (2003). Side effects of interferon and ribavirin therapy for hepatitis C. Nature Clinical Practice Gastroenterology & Hepatology, 1(1):2-3.
Publisher | Google Scholor - Smith, D. B., et al. (2017). Mortality outcomes after SVR in real-world cohorts. Clinical Infectious Diseases, 64(4):544-552.
Publisher | Google Scholor - Sterne, J. A., Savović, J., Page, M. J., Elbers, R. G., Blencowe, N. S., et al. (2019). RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ, 366.
Publisher | Google Scholor - Mangia, A., Thompson, A. J., Santoro, R., Piazzolla, V., Tillmann, H. L., et al. (2010). An IL28B polymorphism determines treatment response of hepatitis C virus genotype 2 or 3 patients who do not achieve a rapid virologic response. Gastroenterology, 139(3):821-827.
Publisher | Google Scholor - Jeong, D., Wong, S., Karim, M. E., Manges, A. R., Makuza, J. D., et al. (2024). Treatment of HCV with direct-acting antivirals on reducing mortality related to extrahepatic manifestations: a large population-based study in British Columbia, Canada. The Lancet Regional Health-Americas, 29.
Publisher | Google Scholor - Schad, V. A. (2020). Treatment With DAAs for HCV Reduces Liver-Related Mortality. Infectious Disease Advisor.
Publisher | Google Scholor - van der Meer, A. J., et al. (2017). Long-term follow-up after SVR. JAMA, 317(21):2081-2091.
Publisher | Google Scholor - White, D. L., et al. (2021). Real-world liver-related mortality after SVR. Journal of Viral Hepatitis, 28(4):535-544.
Publisher | Google Scholor - World Health Organization. (2021). Hepatitis C.
Publisher | Google Scholor