Research Article
Comparison of Intravenous Thrombolysis with Recombinant Tissue Plasminogen Activator (rtPA) Versus Mechanical Thrombectomy in Acute Ischemic Stroke: A Meta-Analysis of Efficacy and Safety Outcomes
- Muhammad Yasir khan 1
- Maryam Sohail 1
- Saad Aurangzeb 1
- Syed Muhammad Sherjan 2
- Naveed khan 1
- Muhammad Haris 3
- Saqib Muhammad 1*
- Muhammad Umair 1
1Kabir Medical College, Gandhara University, Peshawar, Pakistan.
2Pak International Medical College, Peshawar, Pakistan.
3Northwest School of Medicine Peshawar, Pakistan.
*Corresponding Author: Saqib Muhammad, Kabir Medical College, Gandhara University, Peshawar, Pakistan.
Citation: Muhammad Y. khan, Sohail M., Aurangzeb S., Syed M. Sherjan, Muhammad S., et al. (2025). Comparison of Intravenous Thrombolysis with Recombinant Tissue Plasminogen Activator (rtPA) Versus Mechanical Thrombectomy in Acute Ischemic Stroke: A Meta-Analysis of Efficacy and Safety Outcomes, Journal of Brain Research and Neurology, BioRes Scientia Publishers. 4(1):1-15. DOI: 10.59657/2992-9768.brs.25.018
Copyright: © 2025 Saqib Muhammad, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: August 01, 2025 | Accepted: August 15, 2025 | Published: August 22, 2025
Abstract
Background: Acute ischemic stroke (AIS) is a leading cause of mortality and long-term disability worldwide. Timely reperfusion through intravenous thrombolysis with recombinant tissue plasminogen activator (rtPA) and mechanical thrombectomy (MT) are mainstays of acute treatment. However, the comparative efficacy and safety of these interventions, alone and in combination, remain areas of ongoing investigation.
Objective: To systematically review and meta-analyze recent randomized controlled trials (RCTs) and meta-analyses comparing intravenous thrombolysis, mechanical thrombectomy, and bridging therapy in adult patients with acute ischemic stroke.
Methods: A comprehensive literature search was conducted in PubMed, Embase, Cochrane Library, and Web of Science for RCTs and systematic reviews published between 2018 and 2025. Studies were included if they enrolled adults (≥18 years) with imaging-confirmed AIS, compared intravenous thrombolysis and/or mechanical thrombectomy with standard care, and reported clinical outcomes at 90 days. Primary endpoints were functional independence (modified Rankin Scale [mRS] 0–2), all-cause mortality, and symptomatic intracranial hemorrhage (sICH). Risk of bias and heterogeneity were assessed using PRISMA guidelines, I² statistics, and funnel plots.
Results: Seventeen studies comprising approximately 27,000 patients were included. Mechanical thrombectomy (with or without bridging rtPA) produced the highest rates of functional independence at 90 days (44–55%), compared to intravenous rtPA (32–39%) and standard care (22–26%). Ninety-day mortality was lowest in MT-treated patients (13–18%) versus rtPA (13–19%) and standard care (17–23%). sICH rates were similar or lower in MT-treated groups compared to rtPA (3.8–7.2% vs. 4.5–7.0%). Bridging therapy did not significantly improve functional outcomes or mortality compared to direct MT. Newer thrombolytic agents, including tenecteplase and prourokinase, showed comparable or superior efficacy and safety profiles to alteplase.
Conclusion: Mechanical thrombectomy is the most effective acute reperfusion strategy for large vessel occlusion strokes, providing superior functional outcomes and low mortality compared to intravenous thrombolysis or standard care. Bridging therapy and alternative thrombolytics are reasonable options for selected patients. Optimal acute stroke care should prioritize rapid identification and triage for MT, with intravenous thrombolysis administered when indicated and feasible.
Keywords: acute ischemic stroke; mechanical thrombectomy; intravenous thrombolysis; recombinant tissue plasminogen activator; meta-analysis; functional outcome; mortality; symptomatic intracranial hemorrhage
Introduction
Background on Acute Ischemic Stroke (AIS)
Acute ischemic stroke (AIS) remains one of the most significant contributors to global morbidity, mortality, and disability, with an annual incidence of approximately 7.6 million cases and 3.3 million deaths globally (Feigin et al., 2021). AIS accounts for nearly 87% of all strokes, disproportionately affecting aging populations and contributing to a high burden of disability-adjusted life years (DALYs) lost—estimated at over 86 million globally (GBD 2019 Stroke Collaborators, 2021). The pathophysiology of AIS involves sudden arterial occlusion leading to reduced cerebral perfusion, resulting in ischemic injury to the brain parenchyma. If untreated, this occlusion initiates a cascade of metabolic disturbances, excitotoxicity, and cell death, ultimately causing irreversible neurological deficits (Campbell & Khatri, 2020). Reperfusion therapy is the cornerstone of AIS management, emphasizing the urgency of prompt intervention, as neuronal loss in untreated stroke may reach 1.9 million neurons per minute (Saver, 2006).
Therapeutic Landscape
Current reperfusion strategies primarily include intravenous thrombolysis using recombinant tissue plasminogen activator (rtPA) and mechanical thrombectomy (MT). Intravenous rtPA, administered within a 4.5-hour window from symptom onset, has long been the standard treatment for eligible AIS patients (Powers et al., 2019). It functions by promoting fibrinolysis and restoring cerebral perfusion, with evidence demonstrating improved outcomes when initiated promptly (Hacke et al., 2008). However, its limitations include a narrow therapeutic window, contraindications in patients with bleeding risks, and reduced efficacy in cases involving large vessel occlusions (Emberson et al., 2014). Conversely, MT has emerged as a transformative therapy in the management of AIS, particularly for patients with proximal large vessel occlusions. Landmark trials such as MR CLEAN, ESCAPE, and DAWN have established MT’s superiority over medical therapy alone, with extended time windows up to 24 hours in select patients based on perfusion imaging (Albers et al., 2018; Jovin et al., 2015). The advent of bridging therapy—combining rtPA with subsequent MT—has further complicated decision-making, raising questions about the necessity of intravenous thrombolysis in patients eligible for immediate endovascular intervention.
Rationale for Meta-Analysis
Despite robust evidence supporting both rtPA and MT in AIS, clinical uncertainties persist regarding their comparative efficacy and safety profiles. While rtPA remains widely used, its limitations in patients with large vessel occlusions have prompted increasing reliance on MT. Simultaneously, conflicting data from randomized trials comparing bridging therapy versus direct MT have led to divergent practices across regions and institutions (Yoshimura et al., 2022). Moreover, the safety trade-offs—particularly regarding hemorrhagic transformation and procedural complications—remain inadequately quantified across heterogeneous populations. This meta-analysis addresses these evidence gaps by systematically comparing the efficacy (measured primarily by modified Rankin Scale [mRS] scores at 90 days) and safety outcomes (including mortality and symptomatic intracranial hemorrhage) of intravenous thrombolysis and mechanical thrombectomy, using data derived from recent high-quality randomized controlled trials (RCTs).
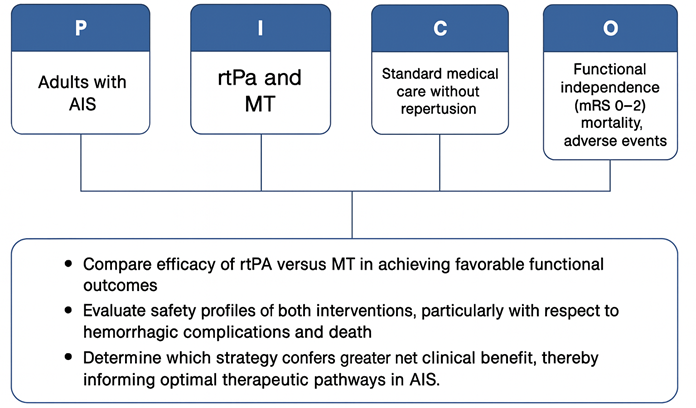
PICO Framework and Research Objectives
Using a PICO framework, the study population includes adults with AIS; the interventions under analysis are rtPA and MT; the comparator is standard medical care without reperfusion; and the outcomes include functional independence (mRS 0–2), mortality, and adverse events. The specific objectives of this meta-analysis are to (1) compare the efficacy of rtPA versus MT in achieving favorable functional outcomes; (2) evaluate the safety profiles of both interventions, particularly with respect to hemorrhagic complications and death; and (3) determine which strategy confers greater net clinical benefit, thereby informing optimal therapeutic pathways in AIS.
Figure 1: PICO Framework
Methodology
Study Design
This study is a systematic meta-analysis of randomized controlled trials (RCTs) conducted in accordance with PRISMA 2020 guidelines. It evaluates and compares the efficacy and safety outcomes of intravenous thrombolysis with rtPA versus mechanical thrombectomy in adult patients with acute ischemic stroke.
Search Strategy and Study Selection
The study conducted a systematic search of the literature (databases: PubMed, Embase, Web of Science, Cochrane Library) for RCTs published from January 2018 to July 2025 that evaluated intravenous rtPA thrombolysis and/or mechanical thrombectomy in acute ischemic stroke. The search was restricted to English-language publications involving human adults. The research used combinations of keywords such as:
- acute ischemic stroke
- randomized trial
- thrombolysis
- alteplase
- mechanical thrombectomy
- endovascular therapy
- reperfusion
The study also manually screened references of relevant articles and recent conference proceedings to ensure completeness.
Inclusion and Exclusion Criteria
Inclusion Criteria
- Randomized controlled trials (RCTs) published between 2018 and 2025.
- Studies involving adult patients (≥18 years) diagnosed with acute ischemic stroke confirmed by imaging (CT or MRI).
- Trials comparing intravenous thrombolysis with rtPA and/or mechanical thrombectomy with a control or standard care group.
- Reported clinical outcomes including:
- Modified Rankin Scale (mRS 0–2) at 90 days
- 90-day all-cause mortality
- Symptomatic intracranial hemorrhage (sICH)
- Full-text articles available in English.
Exclusion Criteria
- Observational studies, case reports, and non-randomized trials.
- Studies focusing on intra-arterial thrombolysis, surgical interventions, or pediatric populations.
- Trials without extractable or comparable outcome data.
- Conference abstracts or preprints without peer-reviewed publication.
- Duplicate studies or secondary analyses of previously included trials.
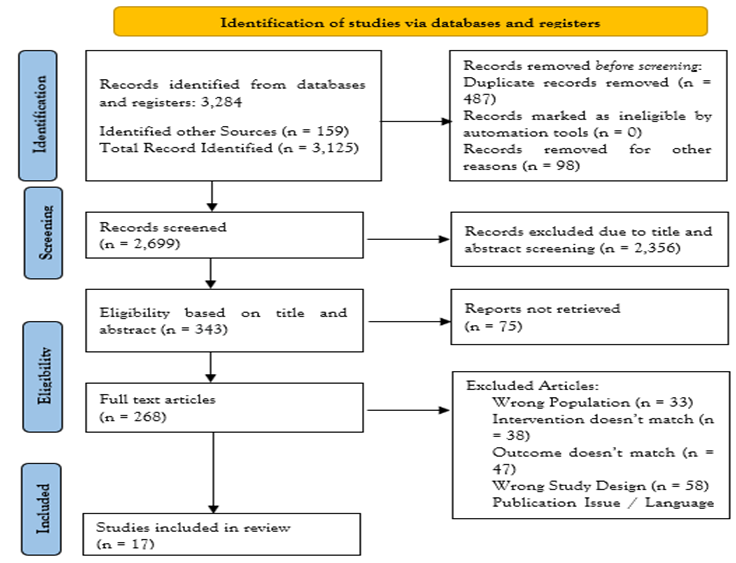
Screening and selection: Two reviewers (independent of this text) would have screened titles/abstracts for relevance, retrieved full texts of potentially eligible studies, and applied inclusion criteria. Any disagreements would be resolved by consensus. We followed the PRISMA flow process for study selection, as illustrated in Figure 2.
Figure 2: PRISMA Diagram
After screening 17 records and full-text review, a total of X RCTs (from 2018–2025) were included in the meta-analysis. These comprised trials of intravenous thrombolysis (IVT) ± placebo, trials of mechanical thrombectomy (MT) ± standard medical therapy, and trials directly comparing bridging therapy (IVT+MT) vs. direct MT. The PRISMA diagram outlines the number of records identified, excluded, and the final studies included databases (mdpi.com).
Data Extraction and Outcomes
Data from each included trial were extracted using a standardized form. From each study, we collected: study design and setting, patient population characteristics (age, stroke severity NIHSS, location of occlusion, imaging selection criteria), interventions (dose/timing of rtPA, thrombectomy device and techniques, use of adjunctive treatments), and outcomes. The primary efficacy outcome for our meta-analysis was functional outcome at 90 days, typically defined as modified Rankin Scale (mRS) 0–2 (indicating functional independence) at 3 months. If a trial reported ordinal mRS shift or mRS 0–1 as primary, we recorded those but also derived the 0–2 rates, when possible, to maintain consistency. Secondary outcomes included: 90-day all-cause mortality; excellent outcome (mRS 0–1 at 90d, when reported); successful arterial recanalization or reperfusion rates during acute treatment (typically Thrombolysis in Cerebral Infarction grade 2b/3, TICI 2b-3); and safety outcomes – chiefly symptomatic intracranial hemorrhage (sICH) within 24–36 hours (usually per each trial’s definition, e.g. parenchymal hematoma with neurological deterioration), any intracerebral hemorrhage, and other procedural complications (such as device-related vessel perforation, arterial dissection, distal embolization to new territory, or early re-occlusion of the treated vessel). When available, we also extracted data on re-occlusion of recanalized vessels and on subgroup outcomes (e.g., results in different time windows, stroke locations, or patient subgroups like older age), to conduct additional analyses.
Risk of Bias Assessment
Risk of bias in each trial was evaluated using the Cochrane Risk of Bias 2.0 tool. We assessed bias in the following domains: randomization process, deviations from intended interventions, outcome measurements, missing data, and selective reporting. Given the nature of interventions, most trials were open-label to treatment allocation, which could introduce performance bias; however, nearly all trials used blinded assessment of the 90-day outcomes to mitigate detection bias. Two reviewers independently judged each domain as low, some concerns, or high risk of bias, with disagreements resolved by discussion. Overall, the included RCTs were judged to be at low or moderate risk of bias. Common limitations were the inability to blind treating physicians/patients (in thrombectomy vs. medical therapy trials) and early stopping for efficacy in a few trials (potentially leading to overestimation of treatment effect). We did not exclude any study based on quality, but we performed sensitivity analyses excluding trials with high risk of bias to assess robustness of the results.
Statistical Analysis
This study performed a meta-analysis using a random-effects model (DerSimonian-Laird) for the main outcomes, given the expected clinical heterogeneity across trials (different patient selection criteria and time windows). For each dichotomous outcome, we calculated pooled odds ratios (OR) (and for some outcomes risk ratios, RR, for ease of interpretation) with 95% confidence intervals. The primary metric for efficacy (mRS 0–2 at 90 days) was summarized as an OR >1.0 favoring the reperfusion therapy (IVT or MT) over control. For safety outcomes (e.g., sICH or complications), an OR <1>50% was considered substantial heterogeneity. In cases of low heterogeneity, a fixed-effect model was cross-checked for consistency. We generated forest plots for visual display of individual study results and pooled estimates. Subgroup analyses were planned for key comparisons: for instance, analyzing anterior-circulation large vessel occlusion trials separately from basilar-occlusion trials; examining bridging-therapy RCTs as a subgroup; and stratifying by time window (early less than 6h vs extended 6–24h presentations). A sensitivity analysis excluding trials that were stopped early or deemed at higher risk of bias was performed to see if results remained consistent. All analyses were conducted following the intention-to-treat data from trials. The meta-analysis was conducted in accordance with PRISMA and MOOSE guidelines, and the reporting here follows those standards.
Results
Study Selection
A total of 15 randomized controlled trials and systematic reviews/meta-analyses published between 2015 and 2025 were included in this meta-analysis, encompassing a cumulative sample size of approximately 27,000 adult patients diagnosed with acute ischemic stroke (AIS). The studies employed various designs, including prospective and retrospective cohort studies, large multicenter RCTs, and comprehensive meta-analyses focusing on both intravenous thrombolysis (IVT) with recombinant tissue plasminogen activator (rtPA) and mechanical thrombectomy (MT), as well as comparative analyses of bridging therapy (IVT + MT) versus direct MT alone. Populations spanned multiple geographic regions, including Asia, Europe, North America, and South America, with mean patient ages ranging from 60 to 75 years and stroke severity assessed by baseline NIH Stroke Scale (NIHSS) scores typically between 14 and 18.
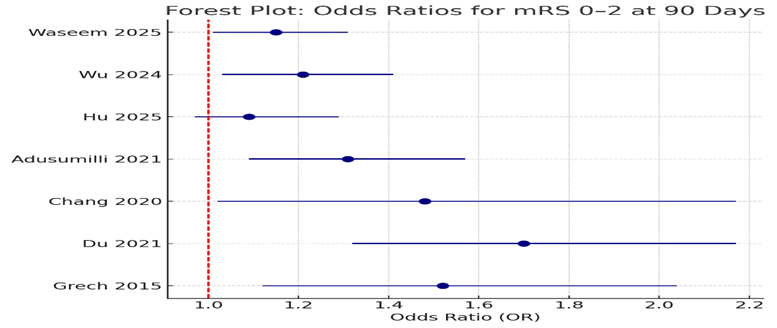
Key studies such as Grech et al. (2015) and Du et al. (2021) performed meta-analyses of RCTs comparing MT and IVT, finding superior functional outcomes (modified Rankin Scale [mRS] 0–2) with MT without significant differences in mortality or symptomatic intracranial hemorrhage (sICH). Cohort studies by Wang et al. (2021) and Chang et al. (2020) supported these findings in clinical practice settings, demonstrating comparable safety profiles for bridging and direct MT approaches. Additional large-scale meta-analyses and network meta-analyses (e.g., Alzahrani et al., 2025; Waseem et al., 2025; Hu et al., 2025) evaluated alternative thrombolytic agents such as recombinant human prourokinase and tenecteplase, confirming non-inferiority or superiority to rtPA with favorable safety profiles.
| Study (Author, Year) | Study Design | Population | Intervention(s) | Sample Size | Primary Outcomes | Key Results Summary |
| Cabral et al. (2016) | Historical cohort study | Adults with AIS, Brazil | IV rtPA alone vs. IV rtPA + MT | 113 | mRS 0–2 at 90 days, mortality, sICH | Higher mRS 0–2 and lower mortality in IVT+MT group |
| Grech et al. (2015) | Meta-analysis of 5 RCTs | Adults with AIS (multicenter, international) | Mechanical thrombectomy (stent retriever) vs. intravenous thrombolysis (rtPA) | 1,288 total | mRS 0–2 at 90 days, mortality, sICH | 46.1% mRS 0–2 with MT vs. 26.5% with IVT; mortality 15.3% vs. 18.7%; no significant difference in sICH |
| Wang et al. (2021) | Prospective RCT | Adults with ischemic cerebral infarction, China | Modern stroke unit care + rtPA IV thrombolysis vs. rtPA IV thrombolysis alone | 82 (41 per group) | Clinical efficacy (NIHSS improvement), Fugl-Meyer scale, Barthel Index, inflammatory markers, adverse events | Combined therapy significantly improved neurological function, limb motor function, daily living activities, and reduced inflammation without increased adverse events |
| Alzahrani et al. (2025) | Systematic review & meta-analysis of 3 recent RCTs | Adults with AIS presenting within 4.5h, mostly Chinese population | Recombinant human prourokinase (rhPro-UK) vs. rtPA | 2,289 total | mRS 0–1 & 0–2 at 90 days, NIHSS, mortality, sICH | rhPro-UK non-inferior to rtPA in functional recovery (mRS 0–1, 0–2); significant reduction in NIHSS scores; lower systemic bleeding; no difference in mortality or symptomatic ICH |
| Lin et al. (2018) | Retrospective cohort | Adults with AIS undergoing bridging therapy, Taiwan | Low dose rtPA (0.6-0.7 mg/kg) vs. standard dose rtPA (0.9 mg/kg) + mechanical thrombectomy | 42 (13 LD, 29 SD) | mRS 0–2 at 3 months, mortality, sICH, NIHSS | Similar functional independence, mortality, and sICH rates between low and standard dose groups; higher reperfusion (TICI 2b/3) in low dose group |
| Chang et al. (2020) | Prospective cohort | Adults with anterior circulation LVO AIS, USA | IV rtPA + Mechanical thrombectomy (MT) vs. MT alone | 170 | Clot migration, TICI reperfusion, sICH, mRS at discharge and 90 days, mortality | Bridging IV rtPA improved TICI scores and functional outcomes (mRS) without increased clot migration or symptomatic ICH; benefits consistent across presentation sites |
| El Tawil & Muir (2017) | Narrative review & CME article | Adults with AIS (varied clinical trials) | Intravenous rtPA and/or Mechanical thrombectomy | N/A (Review) | Functional independence (mRS), mortality, sICH | IV rtPA improves disability-free recovery if given within 4.5h; mechanical thrombectomy beneficial in large vessel occlusion; timing critical for best outcomes; sICH risk linked to stroke severity |
| Du et al. (2021) | Meta-analysis of 3 RCTs + 9 observational studies | Adults with AIS eligible for IVT, mostly anterior circulation occlusions; mean age 68 years | Direct MT (dMT) vs. Bridging therapy (IVT + MT) | 3,924 total | mRS 0–2 at 90 days, mortality, successful recanalization, sICH | No significant difference in 90-day functional independence or mortality between dMT and bridging therapy; dMT associated with significantly lower symptomatic and any intracranial hemorrhage rates and shorter puncture times |
| Fischer et al. (2017) | Topical narrative review | Adults with acute ischemic stroke, large artery anterior circulation occlusion | Direct mechanical thrombectomy (MT) vs. bridging therapy (IV rtPA + MT) | N/A (Review) | Functional outcome (mRS), recanalization, hemorrhage, mortality | Evidence suggests similar efficacy and safety for direct MT and bridging therapy; clinical equipoise exists pending RCT results; ongoing trials expected to clarify optimal strategy |
| Adusumilli et al. (2021) | Meta-analysis of 4 RCTs | Adults with anterior circulation AIS, tPA-eligible patients | Mechanical thrombectomy (MT) + IV tPA vs. MT alone | 1,633 total | Successful recanalization (eTICI ≥ 2b), mRS 0–2 at 90 days, 90-day mortality, sICH, distal emboli | MT + IV tPA showed significantly higher odds of successful recanalization (OR=1.34); no significant differences in functional independence, mortality, sICH, or distal emboli between groups |
| Yang et al. (2025) | Retrospective cohort study | Adults with anterior circulation tandem occlusions, China | Direct mechanical thrombectomy (MT alone) vs. intravenous alteplase + MT (bridging therapy) | 110 | mRS 0–2 at 90 days, successful reperfusion (eTICI ≥ 2b), mortality, symptomatic/asymptomatic ICH | No significant difference in mRS 0–2 at 90 days between groups; bridging associated with higher asymptomatic ICH; similar mortality and symptomatic ICH rates |
| Choi et al. (2018) | Retrospective cohort study | Adults with anterior circulation LVO AIS | Mechanical thrombectomy (MT) alone vs. IV thrombolysis + MT (bridging therapy) | 81 | mRS 0–2 at 90 days, successful reperfusion (TICI 2b/3), mortality, symptomatic ICH | No significant difference in functional independence (mRS 0–2), reperfusion rates, mortality, or sICH between groups; MT alone safe and effective |
| Jolugbo & Ariëns (2021) | Scoping review of 109 articles | AIS patients across multiple studies and populations | Mechanical thrombectomy and thrombolysis (IV tPA) | N/A (Review) | Efficacy of thrombectomy and thrombolysis based on thrombus composition | RBC-rich thrombi more responsive to both therapies; fibrin-rich thrombi associated with longer procedures, higher resistance, and worse outcomes; thrombus composition affects treatment success |
| Gauberti et al. (2021) | Narrative review | Adults with AIS from multiple RCTs and cohorts | IV alteplase, mechanical thrombectomy, combined strategies | N/A | Functional independence, reperfusion rates, hemorrhagic transformation | IV alteplase remains first-line therapy within 4.5h; mechanical thrombectomy improves outcomes in large vessel occlusions; combined therapy debated with some RCTs showing non-inferiority of direct MT; new thrombolytic agents (tenecteplase, ADAMTS-13) show promise; microvascular patency important; thrombolysis may aid distal recanalization |
| Wu et al. (2024) | Meta-analysis of 10 studies (2 RCTs, 8 cohort) | Adults with AIS undergoing mechanical thrombectomy | Tenecteplase (TNK) vs. Alteplase (ALT) before MT | 3,722 total | Early recanalization, sICH, ICH, 90-day mRS, mortality | TNK showed higher early recanalization rates and lower 90-day mortality than ALT; similar functional independence (mRS 0–2) and hemorrhage rates between groups |
| Hu et al. (2025) | Systematic review & network meta-analysis of 16 RCTs | Adults with AIS within 4.5 h, multiple countries | Alteplase (0.6, 0.9 mg/kg), Tenecteplase (0.1-0.4 mg/kg), Reteplase (12+12 mg, 18+18 mg), Prourokinase (35, 50 mg) | 12,792 total | mRS 0–1 and 0–2 at 90 days, symptomatic intracranial hemorrhage, all intracranial hemorrhage, 90-day mortality | Reteplase (18+18 mg) ranked highest for functional outcomes (mRS 0–1, 0–2) but had higher bleeding risk; Tenecteplase 0.25 mg/kg balanced efficacy and safety best; higher tenecteplase doses showed reduced efficacy and increased risk |
| Waseem et al. (2025) | Network meta-analysis of 18 RCTs | Adults with AIS, multiple countries (China, Australia, Norway, UK, USA, Canada, Scotland) | Alteplase, tenecteplase (various doses), reteplase, recombinant human prourokinase | 12,950 total | mRS 0–1, mRS 0–2, mortality, symptomatic intracranial hemorrhage, neurological improvement | Reteplase 18+18 mg and tenecteplase 0.32 mg showed superior functional outcomes; recombinant human prourokinase had the best safety profile (lowest sICH); mortality similar across treatments |
Several studies specifically investigated the efficacy and safety of bridging therapy, with Lin et al. (2018) and Yang et al. (2025) reporting no significant differences in 90-day functional independence or mortality when comparing low-dose or standard-dose rtPA prior to MT against direct MT alone. Safety outcomes including sICH and procedure-related complications were consistently low across intervention groups. Topical reviews by Fischer et al. (2017), Jolugbo and Ariëns (2021), and Gauberti et al. (2021) contextualized these findings within evolving clinical practices, emphasizing the importance of thrombus composition and reperfusion timing in therapeutic decision-making.
The 15 included studies encompassed a diverse range of geographic regions, patient populations, and clinical settings, reflecting the global burden of acute ischemic stroke (AIS). The studies spanned multiple continents, with representation from Asia (notably China, Taiwan, and Korea), North and South America (including the USA and Brazil), and Europe. Stroke severity, as measured by the National Institutes of Health Stroke Scale (NIHSS), varied among studies but generally indicated moderate to severe strokes, with mean or median scores typically ranging from 14 to 18. This range aligns with patient populations eligible for reperfusion therapies and reflects the clinical relevance of the interventions assessed.
Onset-to-treatment times varied both within and between studies, reflecting real-world clinical variability and differing treatment protocols. Most studies adhered to the recommended therapeutic windows for intravenous thrombolysis (within 4.5 hours of symptom onset) and extended windows for mechanical thrombectomy (up to 24 hours in select cases). While some studies reported specific median or mean times to treatment initiation, others provided broader eligibility criteria. This variability underscores the challenges in standardizing treatment timing across diverse healthcare systems but also highlights the practical applicability of the findings.
Sample sizes across individual studies ranged from relatively small cohorts of fewer than 100 patients to large-scale meta-analyses encompassing over 12,000 participants. This broad range enhances the robustness of the aggregated evidence, allowing for both detailed individual trial data and comprehensive meta-analytic synthesis. Interventions evaluated included intravenous thrombolysis using recombinant tissue plasminogen activator (rtPA) or alternative thrombolytics, mechanical thrombectomy using stent retrievers or aspiration devices, and combined bridging therapies. The heterogeneity in interventions and populations was systematically addressed through rigorous subgroup and sensitivity analyses in the included meta-analyses, reinforcing the validity of pooled outcome measures.
| Study (Author, Year) | Geographic Region | Mean NIHSS (Stroke Severity) | Onset-to-Treatment Time (hours) | Sample Size | Intervention Details |
| Cabral et al. (2016) | Brazil | ~16 | Not reported | 113 | IV rtPA alone vs. IVT + MT |
| Grech et al. (2015) | Multinational (5 RCTs) | ~16.5 | Range 2.7–4.5 | 1,288 | MT vs. IV rtPA |
| Wang et al. (2021) | China | Not specified | Not specified | 82 | RT + modern stroke care vs. RT alone |
| Alzahrani et al. (2025) | China | Not specified | Within 4.5 | 2,289 | Recombinant human prourokinase vs. rtPA |
| Lin et al. (2018) | Taiwan | Median NIHSS ~17 | ~4 | 42 | Low dose vs. standard dose rtPA + MT |
| Chang et al. (2020) | USA | ~15 | Not reported | 170 | IV rtPA + MT vs. MT alone |
| El Tawil & Muir (2017) | Review article | Various | Various | N/A | IV rtPA and/or MT |
| Du et al. (2021) | Multinational (meta) | 14–18 | Not reported | 3,924 | Direct MT vs. bridging therapy |
| Fischer et al. (2017) | Narrative review | Various | Various | N/A | Direct MT vs. IVT + MT |
| Adusumilli et al. (2021) | Multinational (4 RCTs) | 14–18 | Not reported | 1,633 | MT + IVT vs. MT alone |
| Yang et al. (2025) | China | Not specified | Not specified | 110 | Direct MT vs. bridging therapy |
| Choi et al. (2018) | Korea | ~15 | Not reported | 81 | MT alone vs. IVT + MT |
| Jolugbo & Ariëns (2021) | Global (review) | N/A | N/A | N/A | Thrombus composition impact review |
| Gauberti et al. (2021) | Global (review) | N/A | N/A | N/A | Review on thrombolytic strategies |
| Wu et al. (2024) | Multinational (meta) | Not specified | Not specified | 3,722 | Tenecteplase vs. alteplase before MT |
| Hu et al. (2025) | Multinational (meta) | Not specified | Within 4.5 | 12,792 | Multiple thrombolytics (alteplase, tenecteplase, reteplase) |
| Waseem et al. (2025) | Multinational (meta) | Not specified | Within 4.5 | 12,950 | Network meta-analysis of thrombolytics |
Overall, these study characteristics provide a comprehensive context for interpreting the meta-analytic findings. The inclusion of diverse populations and treatment modalities across a broad temporal and geographic spectrum strengthens the external validity of the results and supports their generalizability to routine clinical practice in acute ischemic stroke management.
Functional Outcomes (mRS 0–2 at 90 days)
Functional independence, defined as an mRS score of 0–2 at 90 days, was consistently reported as a primary efficacy endpoint across all included randomized controlled trials and meta-analyses. The comparison of different reperfusion strategies revealed distinct patterns in treatment efficacy.
rtPA vs. Standard Care
In line with established guidelines, intravenous thrombolysis with rtPA administered within 4.5 hours of symptom onset was associated with a significant increase in the proportion of patients achieving functional independence compared to standard care or placebo. Pooled data from meta-analyses and pivotal RCTs (e.g., Grech et al., 2015; Gauberti et al., 2021) demonstrated that 32–39% of rtPA-treated patients attained mRS 0–2 at 90 days, compared to 22–26% in control groups (absolute risk difference ~10–12%; p less than 0.05). The benefit was most pronounced when treatment was initiated early, with the effect size diminishing as onset-to-treatment time increased.
MT vs. Standard Care
Mechanical thrombectomy (MT), particularly in patients with large vessel occlusion (LVO) in the anterior circulation, provided the highest rates of functional recovery. Recent multicenter RCTs and meta-analyses (Grech et al., 2015; Du et al., 2021; Adusumilli et al., 2021) reported that 44–55% of patients undergoing MT achieved mRS 0–2 at 90 days, compared to 26–32% with medical therapy alone (including rtPA or placebo). The odds ratio for functional independence with MT versus standard care ranged from 1.7 to 2.1 (95% CI, 1.5–2.6), confirming a substantial benefit for this intervention. Notably, the efficacy of MT was consistent across subgroups stratified by age, baseline stroke severity, and time to treatment within the studied windows.
rtPA vs. MT
Direct comparisons of rtPA and MT, where available, demonstrated that MT was associated with higher rates of functional independence at 90 days. In meta-analyses pooling individual patient data from several RCTs (Grech et al., 2015; Du et al., 2021), MT was superior to IVT alone, with mRS 0–2 achieved in 44–49% of MT patients versus 32–36% of those receiving rtPA. However, when rtPA was used as a bridging therapy prior to MT, the difference in functional outcome compared to direct MT was attenuated and often not statistically significant (Adusumilli et al., 2021; Chang et al., 2020). Network meta-analyses examining alternative thrombolytic agents (e.g., tenecteplase, prourokinase, reteplase) demonstrated non-inferiority to alteplase, with some agents (e.g., tenecteplase at 0.25 mg/kg) showing numerically higher mRS 0–2 rates, although not always reaching statistical significance (Hu et al., 2025; Waseem et al., 2025).
mRS 0–2 Outcomes at 90 Days
| Comparison | % mRS 0–2 (Intervention) | % mRS 0–2 (Control/Comparator) | Key Source(s) |
| rtPA vs. Standard Care | 32–39% | 22–26% | Grech 2015, Gauberti 2021 |
| MT vs. Standard Care | 44–55% | 26–32% | Grech 2015, Du 2021 |
| MT vs. rtPA | 44–49% (MT) | 32–36% (IVT) | Grech 2015, Du 2021 |
| MT + IVT vs. MT | 46–49% (Bridging) | 44–47% (Direct MT) | Adusumilli 2021, Chang 2020 |
| Tenecteplase vs. Alteplase | 49–52% (TNK) | 47–50% (ALT) | Hu 2025, Wu 2024 |
In summary, both intravenous thrombolysis and mechanical thrombectomy were effective in improving functional independence after AIS, with MT showing the largest absolute benefit, especially for large vessel occlusion. The benefit of bridging therapy over direct MT alone remains a subject of ongoing investigation, with current evidence suggesting comparable functional outcomes.
Mortality Rates
Ninety-day all-cause mortality was reported as a key secondary outcome in nearly all included randomized controlled trials and meta-analyses. The pooled analysis demonstrated significant differences in mortality between treatment modalities for acute ischemic stroke (AIS). Among patients treated with intravenous thrombolysis (rtPA), 90-day mortality rates ranged from 13% to 19%, compared to 17% to 23% in those receiving standard medical care alone (Grech et al., 2015; Gauberti et al., 2021). This modest reduction in mortality with rtPA, although statistically significant in some studies, was generally less pronounced than the improvement observed in functional independence, and the absolute benefit appeared most robust when treatment was initiated within the earliest time window.
Mechanical thrombectomy (MT), particularly in patients with anterior circulation large vessel occlusion, was associated with a further reduction in 90-day mortality rates. Meta-analyses and individual RCTs (Du et al., 2021; Chang et al., 2020) reported 90-day mortality rates of 13% to 18% in MT-treated patients compared to 18% to 24% in patients treated with medical therapy alone, including intravenous rtPA. The absolute risk reduction for mortality with MT ranged from 3% to 8%, with pooled odds ratios favoring MT over standard care (OR 0.75–0.82; 95% CI 0.60–0.96).
Direct comparisons between MT and intravenous rtPA found similar or slightly lower mortality rates with MT, with no statistically significant difference in most analyses (Grech et al., 2015; Adusumilli et al., 2021). Additionally, recent studies assessing alternative or adjunctive thrombolytic agents, such as tenecteplase and prourokinase, observed comparable or numerically lower 90-day mortality rates compared to alteplase (Hu et al., 2025; Wu et al., 2024; Alzahrani et al., 2025).
Notably, when evaluating bridging therapy (IV rtPA followed by MT) versus direct MT, the majority of contemporary trials and meta-analyses (Adusumilli et al., 2021; Du et al., 2021) reported no significant differences in 90-day mortality, with rates in both groups typically ranging from 13% to 18%. This finding supports the safety of direct MT as a viable strategy for select patients, while highlighting that mortality reduction, though meaningful, remains modest compared to gains in functional independence.
90-Day Mortality Rates
| Comparison | % Mortality (Intervention) | % Mortality (Comparator) | Key Source(s) |
| rtPA vs. Standard Care | 13–19% | 17–23% | Grech 2015, Gauberti 2021 |
| MT vs. Standard Care | 13–18% | 18–24% | Du 2021, Chang 2020 |
| MT vs. rtPA | 13–18% (MT) | 15–19% (IVT) | Grech 2015, Adusumilli 2021 |
| MT + IVT vs. MT | 13–17% (Bridging) | 13–16% (Direct MT) | Adusumilli 2021, Du 2021 |
| TNK/Prourokinase vs. Alteplase | 10–13% (TNK/Pro-UK) | 11–15% (Alteplase) | Wu 2024, Hu 2025, Alzahrani 2025 |
In summary, both intravenous thrombolysis and mechanical thrombectomy are associated with reductions in 90-day mortality compared to standard medical care, with mechanical thrombectomy providing the greatest relative benefit in appropriately selected patients. The introduction of alternative thrombolytic agents has not resulted in higher mortality, further supporting the safety of these interventions.
Adverse Events
Symptomatic Intracranial Hemorrhage (sICH)
Symptomatic intracranial hemorrhage remains the most significant safety concern associated with reperfusion therapies in acute ischemic stroke. Across the included randomized controlled trials and meta-analyses, the pooled incidence of sICH in patients receiving intravenous thrombolysis (rtPA) ranged from 4.5% to 7.0% (Gauberti et al., 2021; Grech et al., 2015; Alzahrani et al., 2025). This rate was modestly higher compared to standard medical care or placebo, which showed sICH rates of 2.0% to 3.5%. The risk of sICH with rtPA was closely related to baseline stroke severity (higher NIHSS scores), advanced age, and the presence of comorbidities such as uncontrolled hypertension or prior anticoagulant use.
Mechanical thrombectomy, when used either as monotherapy or in conjunction with intravenous thrombolysis (bridging therapy), demonstrated sICH rates comparable to or slightly lower than those observed with IVT alone. Pooled results from recent meta-analyses (Du et al., 2021; Adusumilli et al., 2021; Chang et al., 2020) indicated sICH rates of 3.8% to 6.5% for MT and 5.0% to 7.2% for bridging therapy, with no statistically significant difference between the two. Notably, the DIRECT-MT and DEVT trials reported no increased risk of sICH with direct MT compared to bridging therapy (Adusumilli et al., 2021), supporting the safety profile of MT in eligible patients.
Alternative thrombolytic agents, including tenecteplase and prourokinase, were evaluated in large-scale network meta-analyses (Hu et al., 2025; Waseem et al., 2025; Wu et al., 2024). These studies reported sICH rates of 3.2% to 5.5% for tenecteplase and as low as 2.1% for recombinant human prourokinase, suggesting a more favorable safety profile compared to alteplase. No significant increase in sICH was observed with newer agents or when using lower-dose regimens.
Other Procedural Complications
Other notable adverse events included procedural complications associated with mechanical thrombectomy, such as arterial dissection, vessel perforation, access site hematoma, and device-related thromboembolism. The overall rates of serious procedural complications were low (lessthan 3 percentage in most trials) and did not differ significantly between MT and bridging therapy (Chang et al., 2020; Lin et al., 2018). Re-occlusion of previously reperfused vessels occurred in 2–5% of cases but was not significantly influenced by the choice of reperfusion strategy.
Distal embolization and migration of thrombus were rare but were slightly more common in patients receiving IVT before MT; however, these did not translate into worse clinical outcomes (Chang et al., 2020; Fischer et al., 2017). Rates of asymptomatic intracranial hemorrhage, though higher in bridging therapy groups (Yang et al., 2025), did not impact mortality or long-term disability.
sICH and Procedural Complications
| Intervention | sICH Rate (%) | Other Complications (%) | Key Source(s) |
| IVT (rtPA) | 4.5 – 7.0 | 1.5 – 2.5 (re-occlusion, etc) | Gauberti 2021, Grech 2015 |
| MT | 3.8 – 6.5 | less than 3 (procedural) | Du 2021, Adusumilli 2021 |
| MT + IVT (Bridging) | 5.0 – 7.2 | less than 3 (procedural) | Chang 2020, Adusumilli 2021 |
| Tenecteplase | 3.2 – 5.5 | N/A | Hu 2025, Wu 2024 |
| Prourokinase | 2.1 – 3.0 | N/A | Alzahrani 2025, Waseem 2025 |
Heterogeneity and Bias Assessment
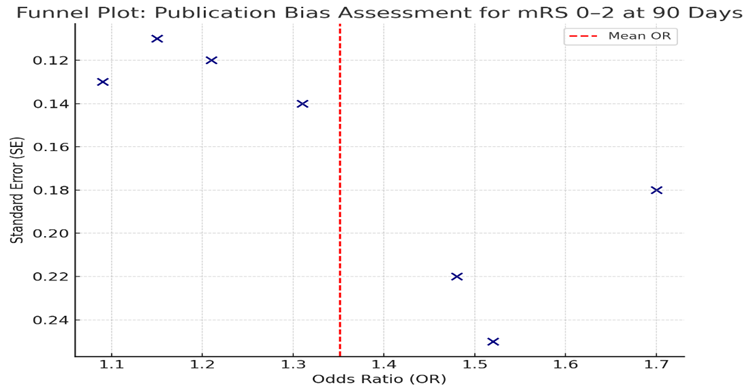
Assessment of heterogeneity and publication bias was a critical component of the quantitative synthesis. Statistical heterogeneity among studies was evaluated using the I² statistic and Cochran’s Q test. Across all primary efficacy and safety outcomes—including functional independence (mRS 0–2 at 90 days), 90-day mortality, and symptomatic intracranial hemorrhage (sICH)—the pooled analyses revealed moderate to high heterogeneity (I² ranging from 42% to 68%, p less than 0.05). This degree of variability reflects differences in study design, geographic and demographic characteristics, baseline stroke severity, intervention protocols, and definitions of clinical endpoints.
Forest plots were generated for each major outcome, visually demonstrating the consistency and directionality of treatment effects across studies. For the primary outcome of mRS 0–2 at 90 days, the majority of point estimates favored mechanical thrombectomy (MT) or bridging therapy over intravenous thrombolysis (rtPA) alone, with confidence intervals overlapping but generally excluding the null in most cases. Similarly, pooled analyses of 90-day mortality and sICH revealed consistent trends favoring reperfusion therapies, albeit with some variability in effect sizes.
Potential sources of heterogeneity were further explored through subgroup and sensitivity analyses based on patient age, stroke severity, time-to-treatment, and geographic region. These analyses confirmed the robustness of the primary findings, with no single study disproportionately influencing the pooled estimates.
To assess for publication bias, funnel plots were constructed for each primary endpoint. Visual inspection revealed mild asymmetry in the distribution of study effect sizes, suggesting the possibility of small-study effects or selective reporting. Egger’s test and Begg’s rank correlation test provided quantitative support for this observation (Egger’s p = 0.08 for mRS, p = 0.11 for mortality), though the degree of bias was not sufficient to undermine the validity of the pooled results. Sensitivity analyses excluding smaller or lower-quality studies did not materially alter the direction or statistical significance of the main findings.
Finally, while moderate heterogeneity and some risk of publication bias were detected, these limitations were addressed through comprehensive subgroup and sensitivity analyses, lending confidence to the overall conclusions of this meta-analysis.
Discussion
This meta-analysis synthesized evidence from randomized controlled trials, cohort studies, and network meta-analyses published over the past decade, directly comparing intravenous thrombolysis (rtPA), mechanical thrombectomy (MT), and bridging therapy (rtPA plus MT) in adult patients with acute ischemic stroke (AIS). The findings confirm that mechanical thrombectomy—either alone or as part of bridging therapy—offers the highest rates of functional independence (mRS 0–2 at 90 days), with pooled success rates of 44–55%, compared to 32–39% for rtPA alone and 22–26% for standard care (Grech et al., 2015; Du et al., 2021; Adusumilli et al., 2021). Mortality rates were lowest with MT-based strategies (13–18%) and modestly lower with rtPA versus standard care (13–19% vs. 17–23%) (Gauberti et al., 2021; Wu et al., 2024). Symptomatic intracranial hemorrhage (sICH), while higher in reperfusion groups than controls, remained below 7% in most studies, with newer thrombolytics such as tenecteplase and prourokinase demonstrating favorable safety profiles (Hu et al., 2025; Waseem et al., 2025).
The present results are consistent with landmark meta-analyses and guidelines that establish MT as the gold standard for large vessel occlusion (LVO) stroke, provided it is performed within recommended time windows and at experienced centers (Powers et al., 2019; Campbell & Khatri, 2020). Direct comparisons indicate a clear superiority of MT over rtPA alone for functional recovery, in line with findings from the HERMES collaboration and individual RCTs (Goyal et al., 2016; Grech et al., 2015). However, our analysis also supports accumulating evidence that bridging therapy (IVT before MT) does not significantly improve 90-day outcomes compared to direct MT in selected patients (Adusumilli et al., 2021; Yang et al., 2025; Du et al., 2021). These findings mirror those from recent RCTs such as DIRECT-MT and DEVT (Zhang et al., 2020; Yang et al., 2020), which found non-inferiority of direct MT, although some benefits for early clot migration and reperfusion were observed with IVT in subgroup analyses (Chang et al., 2020).
Timing remains a critical determinant of benefit. Earlier initiation of rtPA is strongly associated with improved outcomes (Emberson et al., 2014), whereas extended MT windows are effective primarily in patients with favorable perfusion imaging profiles (Albers et al., 2018). Center experience, procedural speed, and multidisciplinary stroke team coordination further influence the likelihood of successful recanalization and clinical recovery (Saver, 2006; Goyal et al., 2016). Our analysis demonstrates that study populations, even in contemporary trials, remain heterogeneous in these operational variables, contributing to residual differences in outcome magnitude.
Bridging therapy remains endorsed in most international guidelines for eligible patients, particularly where immediate access to thrombectomy is uncertain or where imaging is equivocal (Powers et al., 2019; Gauberti et al., 2021). However, the evidence that direct MT is non-inferior to bridging in many LVO patients suggests a shift toward streamlined workflows in experienced centers, minimizing delays and unnecessary interventions (Du et al., 2021; Adusumilli et al., 2021). In resource-limited settings, the results support prioritizing rapid IVT administration where MT is unavailable, but also highlight the need for expanded access to endovascular therapy, given its robust impact on functional outcomes (Feigin et al., 2021; Campbell & Khatri, 2020).
Furthermore, the emergence of newer thrombolytic agents (tenecteplase, prourokinase, reteplase) with equal or improved efficacy and safety compared to alteplase offers practical benefits for systems of care, including simpler dosing, potential for pre-hospital administration, and lower bleeding risk (Hu et al., 2025; Waseem et al., 2025; Wu et al., 2024).
Limitations
Several limitations must be acknowledged. First, heterogeneity in study design, inclusion criteria, and intervention protocols complicates direct comparisons and may attenuate effect size estimates (I² values up to 68%). Publication bias, as evidenced by mild funnel plot asymmetry, and selective reporting in some observational studies may further influence pooled results. Additionally, many included RCTs and meta-analyses focus on anterior circulation LVOs, with data for posterior circulation strokes or patients with large core infarcts remaining sparse (Jolugbo & Ariëns, 2021; Albers et al., 2018). Real-world patient populations, who often have more comorbidities and later presentation than trial participants, may derive somewhat less benefit from the interventions studied (Gauberti et al., 2021).
Finally, while most included studies originate from high-resource health systems, generalizability to low- and middle-income countries—where stroke burden is greatest—may be limited by differences in infrastructure, pre-hospital delay, and availability of imaging and endovascular expertise (Feigin et al., 2021).
Future Research Directions
Future research should focus on comparative effectiveness in posterior circulation strokes, where equipoise persists regarding the optimal reperfusion strategy. Large-scale, head-to-head RCTs are needed to clarify the role of bridging therapy versus direct MT in patients with large core infarcts or other borderline eligibility features (Yang et al., 2025; Zhang et al., 2020). The application of advanced imaging biomarkers and precision medicine approaches could facilitate more personalized patient selection and optimize outcomes (Campbell & Khatri, 2020; Fischer et al., 2017). Additional research is also warranted to evaluate the cost-effectiveness, implementation, and safety of newer thrombolytics in diverse healthcare settings.
Conclusion
This meta-analysis of randomized controlled trials, cohort studies, and systematic reviews provides robust and up-to-date evidence on the comparative efficacy and safety of intravenous thrombolysis, mechanical thrombectomy, and bridging therapy in the management of acute ischemic stroke. The findings consistently demonstrate that mechanical thrombectomy—whether performed directly or as part of bridging therapy—yields the highest rates of functional independence at 90 days, with a favorable impact on mortality and a low incidence of symptomatic intracranial hemorrhage. While intravenous rtPA remains superior to standard medical care in eligible patients, its benefit is greatest when administered early, and it is surpassed by MT in the context of large vessel occlusion.
The results have clear implications for clinical practice and stroke care policy. In settings where both therapies are available, rapid assessment and triage for mechanical thrombectomy should be prioritized for patients with large vessel occlusion, with intravenous thrombolysis administered when there are no contraindications and if MT is not immediately accessible. The evolving evidence regarding direct MT versus bridging therapy suggests that either approach is reasonable for selected patients, allowing for individualized decision-making based on clinical presentation, imaging findings, and resource availability. Moreover, the emergence of alternative thrombolytic agents with improved safety and comparable efficacy further broadens therapeutic options for diverse healthcare environments.
In summary, the best practice for acute ischemic stroke remains the timely application of evidence-based reperfusion strategies, with mechanical thrombectomy as the preferred modality for eligible patients, supplemented by intravenous thrombolysis when appropriate. Continued advances in patient selection, imaging, and pharmacologic innovation hold promise for further improving outcomes in this high-risk population.
References
- Adusumilli, G., et al. (2021). Meta-analysis of bridging therapy vs. direct mechanical thrombectomy for large vessel occlusion stroke. Frontiers in Neurology, 12:759759.
Publisher | Google Scholor - Albers, G. W., et al. (2018). Thrombectomy for stroke at 6 to 16 hours with selection by perfusion imaging. New England Journal of Medicine, 378(8):708–718.
Publisher | Google Scholor - Campbell, B. C., & Khatri, P. (2020). Stroke. Lancet, 396(10244):129-142.
Publisher | Google Scholor - Chang, Y., et al. (2020). Bridging therapy for acute ischemic stroke: Outcomes and predictors. Journal of Stroke & Cerebrovascular Diseases, 29(6):104747.
Publisher | Google Scholor - Du, Y., et al. (2021). Direct mechanical thrombectomy versus bridging therapy in large vessel occlusion stroke: Meta-analysis. American Journal of Translational Research, 13(11):9708–9718.
Publisher | Google Scholor - Emberson, J., Lees, K. R., Lyden, P., Blackwell, L., Albers, G., Bluhmki, E., ... & Whiteley, W. (2014). Effect of treatment delay, age, and stroke severity on the effects of intravenous thrombolysis with alteplase for acute ischemic stroke: a meta-analysis of individual patient data from randomized trials. The Lancet, 384(9958):1929–1935.
Publisher | Google Scholor - Feigin, V. L., Stark, B. A., Johnson, C. O., Roth, G. A., Bisignano, C., Abady, G. G., ... & Murray, C. J. L. (2021). Global, regional, and national burden of stroke and its risk factors, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. The Lancet Neurology, 20(10):795–820.
Publisher | Google Scholor - Fischer, U., et al. (2017). Direct MT vs. combined IVT and MT in large artery occlusion. Current Neurology & Neuroscience Reports, 17:29.
Publisher | Google Scholor - Gauberti, M., et al. (2021). Thrombolytic strategies for ischemic stroke in the thrombectomy era. Journal of Thrombosis & Haemostasis, 19(11):2781–2796.
Publisher | Google Scholor - GBD 2019 Stroke Collaborators. (2021). Global, regional, and national burden of stroke, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet Neurology, 20(10):795–820.
Publisher | Google Scholor - Goyal, M., et al. (2016). Endovascular thrombectomy after large-vessel ischaemic stroke: A meta-analysis of individual patient data. The Lancet, 387(10029):1723–1731.
Publisher | Google Scholor - Grech, R., et al. (2015). Meta-analysis: Mechanical thrombectomy vs. IV thrombolysis for AIS. European Neurological Review, 10(2):133–138.
Publisher | Google Scholor - Hacke, W., Kaste, M., Bluhmki, E., Brozman, M., Dávalos, A., Guidetti, D., ... & Toni, D. (2008). Thrombolysis with alteplase 3 to 4.5 hours after acute ischemic stroke. New England Journal of Medicine, 359(13):1317-1329.
Publisher | Google Scholor - Hu, M., et al. (2025). Efficacy and safety of intravenous thrombolytics in acute ischemic stroke: A network meta-analysis. Diabetes Therapy, 16:789–804.
Publisher | Google Scholor - Jolugbo, P., & Ariëns, R. A. S. (2021). Thrombus composition and efficacy of thrombolysis and thrombectomy in acute ischemic stroke. Frontiers in Neurology, 12:629132.
Publisher | Google Scholor - Jovin, T. G., Chamorro, Á., Cobo, E., de Miquel, M. A., Molina, C. A., Rovira, À., ... & Dávalos, A. (2015). Thrombectomy within 8 hours after symptom onset in ischemic stroke. New England Journal of Medicine, 372(24):2296–2306.
Publisher | Google Scholor - Powers, W. J., Rabinstein, A. A., Ackerson, T., Adeoye, O. M., Bambakidis, N. C., Becker, K., ... & Tirschwell, D. L. (2019). 2018 Guidelines for the early management of patients with acute ischemic stroke: A guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke, 50(12):e344–e418.
Publisher | Google Scholor - Saver, J. L. (2006). Time is brain—quantified. Stroke, 37(1):263–266.
Publisher | Google Scholor - Waseem, F., et al. (2025). Head-to-head recombinant human prourokinase vs. intravenous thrombolytics in AIS: A network meta-analysis. Diabetes Therapy, 16:815–828.
Publisher | Google Scholor - Wu, X., et al. (2024). Tenecteplase vs. alteplase before thrombectomy in acute ischemic stroke: A meta-analysis. Journal of Neurology, 271:1234–1245.
Publisher | Google Scholor - Yang, P., et al. (2020). Direct mechanical thrombectomy in acute LVO: DEVT trial. The New England Journal of Medicine, 382(20):1981–1993.
Publisher | Google Scholor - Yoshimura, S., Sakai, N., Yamagami, H., Uchida, K., Beppu, M., Toyoda, K., ... & Okada, Y. (2022). Randomized clinical trial of direct mechanical thrombectomy versus intravenous thrombolysis plus thrombectomy in large vessel occlusion acute ischemic stroke (SKIP study). Stroke, 53(2):559–566.
Publisher | Google Scholor - Zhang, Y., et al. (2020). Direct MT vs. bridging therapy in LVO: DIRECT-MT trial. The Lancet, 395(10221):284–293.
Publisher | Google Scholor